Abstract
The issue of treating Multiple Sclerosis (MS) begins with disease-modifying treatments (DMTs) which may cause lymphopenia, dyspnea, and many other adverse effects. Consequently, further identification and evaluation of alternative treatments are crucial to monitoring their long-term outcomes and hopefully, moving toward personalized approaches that can be translated into clinical treatments. In this article, we focused on the novel therapeutic modalities that alter the interaction between the cellular constituents contributing to MS onset and progression. Furthermore, the studies that have been performed to evaluate and optimize drugs’ efficacy, and particularly, to show their limitations and strengths are also presented. The preclinical trials of novel approaches for multiple sclerosis treatment provide promising prospects to cure the disease with pinpoint precision. Considering the fact that not a single treatment could be effective enough to cover all aspects of MS treatment, additional researches and therapies need to be developed in the future. Since the pathophysiology of MS resembles a jigsaw puzzle, researchers need to put a host of pieces together to create a promising window towards MS treatment. Thus, a combination therapy encompassing all these modules is highly likely to succeed in dealing with the disease.
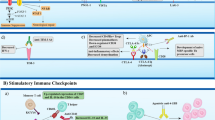
Graphic Abstract
The use of different therapeutic approaches to re-induce self-tolerance in autoreactive cells contributing to MS pathogenesis is presented. A Combination therapy using these tools may help to deal with the clinical disabilities and symptoms of the disease in the future.


Similar content being viewed by others
Abbreviations
- CNS:
-
Central nervous system
- MS:
-
Multiple sclerosis
- RRMS:
-
Relapsing–remitting multiple sclerosis
- PPMS:
-
Primary progressive MS
- SPMS:
-
Secondary progressive MS
- EAE:
-
Experimental autoimmune encephalomyelitis
- TMEV:
-
Theiler’s murine encephalomyelitis virus
- DMT:
-
Disease-modifying treatments
- Th1:
-
T helper 1 (IFNγ producing CD4+ T cells)
- Th1:
-
T helper 17 (IL-17 producing CD4+ T cells)
- Tc1:
-
T cytotoxic 1 (IFNγ producing CD8+ T cells
- Tc17:
-
T cytotoxic 17 (IL-17 producing CD8+ T cells)
- Treg:
-
CD4+CD25+Foxp3+ regulatory T cells
- DNAzymes:
-
Deoxyribozymes
- siRNA:
-
Short interfering RNA or RNA interference (RNAi)
- circRNA:
-
Circular RNA
- ASO:
-
Antisense oligonucleotide
- MBP:
-
Myelin basic protein
- PLP:
-
Myelin proteolipid protein
- MOG:
-
Myelin oligodendrocyte glycoprotein
- i.v.:
-
Intravenous
- i.p.:
-
Intraperitoneal
- i.m.:
-
Intramuscular
- i.c.v.:
-
Intracerebroventricular
- OS:
-
Oxidative stress
- ROS:
-
Reactive oxygen species
- LncRNAs:
-
Long non-coding RNAs
- PBMC:
-
Peripheral blood mononuclear cell
- MBP:
-
Myelin basic protein
References
Aartsma-Rus A, Krieg AM (2017) FDA Approves Eteplirsen for Duchenne muscular dystrophy: the next chapter in the Eteplirsen saga. Nucleic Acid Ther 27(1):1–3
Ahmed Z, Fulton D, Douglas MR (2020) Opicinumab: is it a potential treatment for multiple sclerosis? Ann Transl Med 8(14):892
Andersson A, Isaksson M, Wefer J, Norling A, Flores-Morales A, Rorsman F, Kampe O, Harris RA, Lobell A (2008) Impaired autoimmune T helper 17 cell responses following DNA vaccination against rat experimental autoimmune encephalomyelitis. PloS one 3(11):e3682
Antony JM, Ellestad KK, Hammond R, Imaizumi K, Mallet F, Warren KG, Power C (2007) The human endogenous retrovirus envelope glycoprotein, syncytin-1, regulates neuroinflammation and its receptor expression in multiple sclerosis: a role for endoplasmic reticulum chaperones in astrocytes. J Immunol (Baltimore, Md: 1950) 179(2):1210–1224
Appaiahgari MB, Vrati S (2007) DNAzyme-mediated inhibition of Japanese encephalitis virus replication in mouse brain. Mol Ther 15(9):1593–1599
Bar-Or A, Vollmer T, Antel J, Arnold DL, Bodner CA, Campagnolo D, Gianettoni J, Jalili F, Kachuck N, Lapierre Y et al (2007) Induction of antigen-specific tolerance in multiple sclerosis after immunization with DNA encoding myelin basic protein in a randomized, placebo-controlled phase 1/2 trial. Arch Neurol 64(10):1407–1415
Bar-Or A, Pachner A, Menguy-Vacheron F, Kaplan J, Wiendl H (2014) Teriflunomide and its mechanism of action in multiple sclerosis. Drugs 74(6):659–674
Batista PJ, Chang HY (2013) Long noncoding RNAs: cellular address codes in development and disease. Cell 152(6):1298–1307
Bielekova B (2013) Daclizumab therapy for multiple sclerosis. Neurotherapeutics 10(1):55–67
Bomprezzi R (2015) Dimethyl fumarate in the treatment of relapsing–remitting multiple sclerosis: an overview. Ther Adv Neurol Disord 8(1):20–30
Boneschi FM, Vacchi L, Rovaris M, Capra R, Comi G (2013) Mitoxantrone for multiple sclerosis. Cochrane Database of Syst Rev. https://doi.org/10.1002/14651858.CD002127.pub3
Bourquin C, Iglesias A, Berger T, Wekerle H, Linington C (2000) Myelin oligodendrocyte glycoprotein-DNA vaccination induces antibody-mediated autoaggression in experimental autoimmune encephalomyelitis. Eur J Immunol 30(12):3663–3671
Bouvier-Müller A, Ducongé F (2018) Nucleic acid aptamers for neurodegenerative diseases. Biochimie 145:73–83
Braley TJ, Segal BM, Chervin RD (2015) Hypnotic use and fatigue in multiple sclerosis. Sleep Med 16(1):131–137
Brandstadter R, Katz Sand I (2017) The use of natalizumab for multiple sclerosis. Neuropsychiatr Dis Treat 13:1691–1702
Brazão TF, Johnson JS, Müller J, Heger A, Ponting CP, Tybulewicz VL (2016) Long noncoding RNAs in B-cell development and activation. Blood 128(7):e10–e19
Breaker RR, Joyce GF (1994) A DNA enzyme that cleaves RNA. Chem Biol 1(4):223–229
Bumcrot D, Manoharan M, Koteliansky V, Sah DW (2006) RNAi therapeutics: a potential new class of pharmaceutical drugs. Nat Chem Biol 2(12):711–719
Burnett JC, Rossi JJ (2012) RNA-based therapeutics: current progress and future prospects. Chem Biol 19(1):60–71
Cao Y, Yang L, Jiang W, Wang X, Liao W, Tan G, Liao Y, Qiu Y, Feng D, Tang F et al (2014) Therapeutic evaluation of Epstein-Barr virus-encoded latent membrane protein-1 targeted DNAzyme for treating of nasopharyngeal carcinomas. Mol Ther 22(2):371–377
Cardamone G, Paraboschi EM, Rimoldi V, Duga S, Solda G, Asselta R (2017) The characterization of GSDMB splicing and backsplicing profiles identifies novel isoforms and a circular RNA that are dysregulated in multiple sclerosis. Int J Mol Sci 18(3):576
Chakravarthy M, Aung-Htut MT, Le BT, Veedu RN (2017) Novel Chemically-modified DNAzyme targeting Integrin alpha-4 RNA transcript as a potential molecule to reduce inflammation in multiple sclerosis. Sci Rep 7(1):1613
Chapman JA, Beckey C (2006) Pegaptanib: a novel approach to ocular neovascularization. Ann Pharmacother 40(7–8):1322–1326
Cho EA, Moloney FJ, Cai H, Au-Yeung A, China C, Scolyer RA, Yosufi B, Raftery MJ, Deng JZ, Morton SW et al (2013) Safety and tolerability of an intratumorally injected DNAzyme, Dz13, in patients with nodular basal-cell carcinoma: a phase 1 first-in-human trial (DISCOVER). Lancet (london, England) 381(9880):1835–1843
Clifford DB, De Luca A, Simpson DM, Arendt G, Giovannoni G, Nath A (2010) Natalizumab-associated progressive multifocal leukoencephalopathy in patients with multiple sclerosis: lessons from 28 cases. Lancet Neurol 9(4):438–446
Comi G, Martinelli V, Rodegher M, Moiola L, Bajenaru O, Carra A, Elovaara I, Fazekas F, Hartung HP, Hillert J et al (2009) Effect of glatiramer acetate on conversion to clinically definite multiple sclerosis in patients with clinically isolated syndrome (PreCISe study): a randomised, double-blind, placebo-controlled trial. Lancet (london, England) 374(9700):1503–1511
Comi G, Jeffery D, Kappos L, Montalban X, Boyko A, Rocca MA, Filippi M (2012) Placebo-controlled trial of oral laquinimod for multiple sclerosis. N Engl J Med 366(11):1000–1009
De Giglio L, De Luca F, Gurreri F, Ferrante I, Prosperini L, Borriello G, Quartuccio E, Gasperini C, Pozzilli C (2019) Effect of dalfampridine on information processing speed impairment in multiple sclerosis. Neurology 93(8):e733–e746
Dendrou CA, Fugger L, Friese MA (2015) Immunopathology of multiple sclerosis. Nat Rev Immunol 15(9):545–558
Doi Y, Oki S, Ozawa T, Hohjoh H, Miyake S, Yamamura T (2008) Orphan nuclear receptor NR4A2 expressed in T cells from multiple sclerosis mediates production of inflammatory cytokines. Proc Natl Acad Sci USA 105(24):8381–8386
Dowaidar M: Strategies for treating multiple sclerosis with gene therapy. 2021.
Eftekharian MM, Ghafouri-Fard S, Soudyab M, Omrani MD, Rahimi M, Sayad A, Komaki A, Mazdeh M, Taheri M (2017) Expression analysis of long non-coding RNAs in the blood of multiple sclerosis patients. J Mol Neurosci 63(3):333–341
Evers MM, Toonen LJ, van Roon-Mom WM (2015) Antisense oligonucleotides in therapy for neurodegenerative disorders. Adv Drug Deliv Rev 87:90–103
Fang Y, Fullwood MJ (2016) Roles, functions, and mechanisms of long non-coding RNAs in cancer. Genomics Proteomics Bioinformatics 14(1):42–54
Fang S, Zhang L, Guo J, Niu Y, Wu Y, Li H, Zhao L, Li X, Teng X, Sun X (2018) NONCODEV5: a comprehensive annotation database for long non-coding RNAs. Nucleic Acids Res 46(D1):D308–D314
Fereidan-Esfahani M, Yue WY, Wilbanks B, Johnson AJ, Warrington AE, Howe CL, Rodriguez M, Maher LJ (2020) Remyelination-promoting DNA Aptamer conjugate myaptavin-3064 binds to adult oligodendrocytes in vitro. Pharmaceuticals (Basel, Switzerland) 13(11):403
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC (1998) Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 391(6669):806–811
Fischer JS, Priore RL, Jacobs LD, Cookfair DL, Rudick RA, Herndon RM, Richert JR, Salazar AM, Goodkin DE, Granger CV (2000) Neuropsychological effects of interferon β-1a in relapsing multiple sclerosis. Ann Neurol 48(6):885–892
Fissolo N, Costa C, Nurtdinov RN, Bustamante MF, Llombart V, Mansilla MJ, Espejo C, Montalban X, Comabella M (2012) Treatment with MOG-DNA vaccines induces CD4+CD25+FoxP3+ regulatory T cells and up-regulates genes with neuroprotective functions in experimental autoimmune encephalomyelitis. J Neuroinflammation 9:139
Fokina AA, Stetsenko DA, François JC (2015) DNA enzymes as potential therapeutics: towards clinical application of 10–23 DNAzymes. Expert Opin Biol Ther 15(5):689–711
Fragoso YD, Alves-Leon SV, Barreira AA, Callegaro D, Ferreira MLB, Finkelsztejn A, Gomes S, Goncalves MVM, Machado MIM, Marques VD (2015) Fingolimod prescribed for the treatment of multiple sclerosis in patients younger than age 18 years. Pediatr Neurol 53(2):166–168
Galetta SL (2001) The controlled high risk Avonex® multiple sclerosis trial (Champs study). J Neuroophthalmol 21(4):292–295
Garren H, Robinson WH, Krasulova E, Havrdova E, Nadj C, Selmaj K, Losy J, Nadj I, Radue EW, Kidd BA et al (2008a) Phase 2 trial of a DNA vaccine encoding myelin basic protein for multiple sclerosis. Ann Neurol 63(5):611–620
Garren H, Robinson WH, Krasulová E, Havrdová E, Nadj C, Selmaj K, Losy J, Nadj I, Radue EW, Kidd BA (2008b) Phase 2 trial of a DNA vaccine encoding myelin basic protein for multiple sclerosis. Ann Neurol 63(5):611–620
Gaultier A, Wu X, Le Moan N, Takimoto S, Mukandala G, Akassoglou K, Campana WM, Gonias SL (2009) Low-density lipoprotein receptor-related protein 1 is an essential receptor for myelin phagocytosis. J Cell Sci 122(Pt 8):1155–1162
Geng X, Jia Y, Zhang Y, Shi L, Li Q, Zang A, Wang H (2020) Circular RNA: Biogenesis, degradation, functions and potential roles in mediating resistance to anticarcinogens. Epigenomics 12(3):267–283
Geurts JJ, Barkhof F (2008) Grey matter pathology in multiple sclerosis. Lancet Neurol 7(9):841–851
Ghaderian S, Shomali N, Behravesh S, Danbaran GR, Hemmatzadeh M, Aslani S, Jadidi-Niaragh F, Hosseinzadeh R, Torkamandi S, Mohammadi H (2020) The emerging role of lncRNAs in multiple sclerosis. J Neuroimmunol 347:577347
Gharzi V, Gangi M, Sayad A, Mazdeh M, Arsang-Jang S, Taheri M (2018) Expression analysis of BDNF gene and BDNF-AS long noncoding RNA in whole blood samples of multiple sclerosis patients: not always a negative correlation between them. Iran J Allergy Asthma Immunol. https://doi.org/10.18502/ijaai.v17i6.619
Gholamzad M, Ebtekar M, Ardestani MS, Azimi M, Mahmodi Z, Mousavi MJ, Aslani S (2018) A comprehensive review on the treatment approaches of multiple sclerosis: currently and in the future. Inflamm Res. https://doi.org/10.1007/s00011-018-1185-0
Glazar P, Papavasileiou P, Rajewsky N (2014) circBase: a database for circular RNAs. RNA (new York, NY) 20(11):1666–1670
Golden T, Dean NM, Honkanen RE (2002) Use of antisense oligonucleotides: advantages, controls, and cardiovascular tissue. Microcirculation 9(1):51–64
Goodman AD, Anadani N, Gerwitz L (2019) Siponimod in the treatment of multiple sclerosis. Expert Opin Investig Drugs 28(12):1051–1057
Greulich T, Hohlfeld JM, Neuser P, Lueer K, Klemmer A, Schade-Brittinger C, Harnisch S, Garn H, Renz H, Homburg U et al (2018) A GATA3-specific DNAzyme attenuates sputum eosinophilia in eosinophilic COPD patients: a feasibility randomized clinical trial. Respir Res 19(1):55
Guerrier-Takada C, Gardiner K, Marsh T, Pace N, Altman S (1983) The RNA moiety of ribonuclease P is the catalytic subunit of the enzyme. Cell 35(3 Pt 2):849–857
Hammond SM, Boettcher S, Caudy AA, Kobayashi R, Hannon GJ (2001) Argonaute2, a link between genetic and biochemical analyses of RNAi. Science (new York, NY) 293(5532):1146–1150
Hansel TT, Kropshofer H, Singer T, Mitchell JA, George AJ (2010) The safety and side effects of monoclonal antibodies. Nat Rev Drug Discovery 9(4):325–338
Harris VK, Sadiq SA (2014) Biomarkers of therapeutic response in multiple sclerosis: current status. Mol Diagn Ther 18(6):605–617
Hauser SL, Bar-Or A, Cohen JA, Comi G, Correale J, Coyle PK, Cross AH, de Seze J, Leppert D, Montalban X (2020) Ofatumumab versus teriflunomide in multiple sclerosis. N Engl J Med 383(6):546–557
Healy JM, Lewis SD, Kurz M, Boomer RM, Thompson KM, Wilson C, McCauley TG (2004) Pharmacokinetics and biodistribution of novel aptamer compositions. Pharm Res 21(12):2234–2246
Heider RM, Smestad JA, Lemus HN, Wilbanks B, Warrington AE, Peters JP, Rodriguez M, Maher LJ 3rd (2018) An assay that predicts in vivo efficacy for DNA Aptamers that stimulate Remyelination in a mouse model of multiple sclerosis. Mol Ther 9:270–277
Hobernik D, Bros M (2018) DNA Vaccines-How Far From Clinical Use? Int J Mol Sci 19(11):3605
Holliger P, Hudson PJ (2005) Engineered antibody fragments and the rise of single domains. Nat Biotechnol 23(9):1126–1136
Homburg U, Renz H, Timmer W, Hohlfeld JM, Seitz F, Luer K, Mayer A, Wacker A, Schmidt O, Kuhlmann J et al (2015) Safety and tolerability of a novel inhaled GATA3 mRNA targeting DNAzyme in patients with TH2-driven asthma. J Allergy Clin Immunol 136(3):797–800
Hoy SM (2015) Peginterferon beta-1a: a review of its use in patients with relapsing-remitting multiple sclerosis. CNS Drugs 29(2):171–179
Huang W, Thomas B, Flynn RA, Gavzy SJ, Wu L, Kim SV, Hall JA, Miraldi ER, Ng CP, Rigo F (2015) DDX5 and its associated lncRNA Rmrp modulate TH 17 cell effector functions. Nature 528(7583):517–522
Ishiguro A, Akiyama T, Adachi H, Inoue J, Nakamura Y (2011) Therapeutic potential of anti-interleukin-17A aptamer: suppression of interleukin-17A signaling and attenuation of autoimmunity in two mouse models. Arthritis Rheum 63(2):455–466
Jeck WR, Sharpless NE (2014) Detecting and characterizing circular RNAs. Nat Biotechnol 32(5):453–461
Jeck WR, Sorrentino JA, Wang K, Slevin MK, Burd CE, Liu J, Marzluff WF, Sharpless NE (2013) Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA (new York, NY) 19(2):141–157
American Journal of Clinical Hypnosis and Multiple Sclerosis : A Brief Case Report. 2011(March 2015).
Kalantari T, Karimi MH, Ciric B, Yan Y, Rostami A, Kamali-Sarvestani E (2014) Tolerogenic dendritic cells produced by lentiviral-mediated CD40- and interleukin-23p19-specific shRNA can ameliorate experimental autoimmune encephalomyelitis by suppressing T helper type 17 cells. Clin Exp Immunol 176(2):180–189
Kalliolias GD, Ivashkiv LB (2016) TNF biology, pathogenic mechanisms and emerging therapeutic strategies. Nat Rev Rheumatol 12(1):49–62
Khan KH (2013) DNA Vaccines: Roles against Diseases. Germs 3(1):26
KhorshidAhmad T, Acosta C, Cortes C, Lakowski TM, Gangadaran S, Namaka M (2016) Transcriptional regulation of brain-derived neurotrophic factor (BDNF) by methyl CpG binding protein 2 (MeCP2): a novel mechanism for re-myelination and/or myelin repair involved in the treatment of multiple sclerosis (MS). Mol Neurobiol 53(2):1092–1107
Kim SH, Dass CR (2012) Induction of caspase-2 activation by a DNA enzyme evokes tumor cell apoptosis. DNA Cell Biol 31(1):1–7
Krug N, Hohlfeld JM, Kirsten AM, Kornmann O, Beeh KM, Kappeler D, Korn S, Ignatenko S, Timmer W, Rogon C et al (2015) Allergen-induced asthmatic responses modified by a GATA3-specific DNAzyme. N Engl J Med 372(21):1987–1995
Kryza D, Debordeaux F, Azema L, Hassan A, Paurelle O, Schulz J, Savona-Baron C, Charignon E, Bonazza P, Taleb J et al (2016) Ex vivo and in vivo imaging and biodistribution of aptamers targeting the human matrix metalloprotease-9 in melanomas. PloS One 11(2):e0149387
Kulcheski FR, Christoff AP, Margis R (2016) Circular RNAs are miRNA sponges and can be used as a new class of biomarker. J Biotechnol 238:42–51
Lasda E, Parker R (2014) Circular RNAs: diversity of form and function. RNA (new York, NY) 20(12):1829–1842
Lassmann H, van Horssen J, Mahad D (2012) Progressive multiple sclerosis: pathology and pathogenesis. Nat Rev Neurol 8(11):647–656
Li X, Yan M, Hu L, Sun L, Zhang F, Ji H, Jiang J, Wang P, Liu H, Gao Y et al (2010) Involvement of Src-suppressed C kinase substrate in experimental autoimmune encephalomyelitis: a link between release of astrocyte proinflammatory factor and oligodendrocyte apoptosis. J Neurosci Res 88(9):1858–1871
Li Z, Chao T-C, Chang K-Y, Lin N, Patil VS, Shimizu C, Head SR, Burns JC, Rana TM (2014) The long noncoding RNA THRIL regulates TNFα expression through its interaction with hnRNPL. Proc Natl Acad Sci 111(3):1002–1007
Li H, Lian G, Wang G, Yin Q, Su Z (2021) A review of possible therapies for multiple sclerosis. Mol Cell Biochem. https://doi.org/10.1007/s11010-021-04119-z
Liao WH, Yang LF, Liu XY, Zhou GF, Jiang WZ, Hou BL, Sun LQ, Cao Y, Wang XY (2014) DCE-MRI assessment of the effect of Epstein-Barr virus-encoded latent membrane protein-1 targeted DNAzyme on tumor vasculature in patients with nasopharyngeal carcinomas. BMC Cancer 14:835
Limmroth V, Barkhof F, Desem N, Diamond MP, Tachas G (2014) CD49d antisense drug ATL1102 reduces disease activity in patients with relapsing-remitting MS. Neurology 83(20):1780–1788
Lobell A, Weissert R, Storch MK, Svanholm C, de Graaf KL, Lassmann H, Andersson R, Olsson T, Wigzell H (1998) Vaccination with DNA encoding an immunodominant myelin basic protein peptide targeted to Fc of immunoglobulin G suppresses experimental autoimmune encephalomyelitis. J Exp Med 187(9):1543–1548
Lobell A, Weissert R, Eltayeb S, de Graaf KL, Wefer J, Storch MK, Lassmann H, Wigzell H, Olsson T (2003) Suppressive DNA vaccination in myelin oligodendrocyte glycoprotein peptide-induced experimental autoimmune encephalomyelitis involves a T1-biased immune response. J Immunol (Baltimore, Md: 1950) 170(4):1806–1813
Lovett-Racke AE, Rocchini AE, Choy J, Northrop SC, Hussain RZ, Ratts RB, Sikder D, Racke MK (2004) Silencing T-bet defines a critical role in the differentiation of autoreactive T lymphocytes. Immunity 21(5):719–731
Ma L, Liu J (2020) Catalytic nucleic acids: biochemistry, chemical biology, biosensors, and nanotechnology. iScience 23(1):100815
Mansourabadi AH, Khosroshahi LM, Noorbakhsh F, Amirzargar A (2021) Cell therapy in transplantation: A comprehensive review of the current applications of cell therapy in transplant patients with the focus on Tregs, CAR Tregs, and Mesenchymal stem cells. Int Immunopharmacol 97:107669
Masoumi F, Ghorbani S, Talebi F, Branton WG, Rajaei S, Power C, Noorbakhsh F (2019) Malat1 long noncoding RNA regulates inflammation and leukocyte differentiation in experimental autoimmune encephalomyelitis. J Neuroimmunol 328:50–59
Melzer N, Meuth SG (2014) Disease-modifying therapy in multiple sclerosis and chronic inflammatory demyelinating polyradiculoneuropathy: common and divergent current and future strategies. Clin Exp Immunol 175(3):359–372
Memczak S, Jens M, Elefsinioti A, Torti F, Krueger J, Rybak A, Maier L, Mackowiak SD, Gregersen LH, Munschauer M et al (2013) Circular RNAs are a large class of animal RNAs with regulatory potency. Nature 495(7441):333–338
Metaxakis A, Petratou D, Tavernarakis N (2020) Molecular interventions towards multiple sclerosis treatment. Brain Sci 10(5):299
Mulero P, Midaglia L, Montalban X (2018) Ocrelizumab: a new milestone in multiple sclerosis therapy. Ther Adv Neurol Disord 11:1756286418773025
Nakhaei-Nejad M, Barilla D, Lee C-H, Blevins G, Giuliani F (2017) Characterization of lymphopenia in patients with MS treated with dimethyl fumarate and fingolimod. Neurol Neuroimmunol Neuroinflamm 5(2):e432–e432
Nastasijevic B, Wright BR, Smestad J, Warrington AE, Rodriguez M, Maher LJ (2012) Remyelination induced by a DNA aptamer in a mouse model of multiple sclerosis. PloS One 7(6):e39595
Nastasijevic B, Wright BR, Smestad J, Warrington AE, Rodriguez M, Maher LJ (2012) Remyelination induced by a DNA aptamer in a mouse model of multiple sclerosis. PloS one 7(6):e39595
Neuhaus O, Farina C, Wekerle H, Hohlfeld R (2001) Mechanisms of action of glatiramer acetate in multiple sclerosis. Neurology 56(6):702–708
Papadopoulou A, von Felten S, Traud S, Rahman A, Quan J, King R, Garren H, Steinman L, Cutter G, Kappos L et al (2012) Evolution of MS lesions to black holes under DNA vaccine treatment. J Neurol 259(7):1375–1382
Perschbacher K, Smestad JA, Peters JP, Standiford MM, Denic A, Wootla B, Warrington AE, Rodriguez M, Maher LJ III (2015) Quantitative PCR analysis of DNA aptamer pharmacokinetics in mice. Nucleic Acid Ther 25(1):11–19
Polman CH, O’Connor PW, Havrdova E, Hutchinson M, Kappos L, Miller DH, Phillips JT, Lublin FD, Giovannoni G, Wajgt A et al (2006) A randomized, placebo-controlled trial of natalizumab for relapsing multiple sclerosis. N Engl J Med 354(9):899–910
Popescu BF, Lucchinetti CF (2012) Pathology of demyelinating diseases. Annu Rev Pathol 7:185–217
Puranik N, Yadav D, Chauhan PS, Kwak M, Jin J-O (2021) Exploring the role of gene therapy for neurological disorders. Curr Gene Ther 21(1):11–22
Ransohoff RM (2007) Natalizumab for multiple sclerosis. N Engl J Med 356(25):2622–2629
Rasche L, Paul F (2018) Ozanimod for the treatment of relapsing remitting multiple sclerosis. Expert Opin Pharmacother 19(18):2073–2086
Roberts TC, Langer R, Wood MJ (2020) Advances in oligonucleotide drug delivery. Nat Rev Drug Discovery 19(10):673–694
Rozenblum GT, Kaufman T, Vitullo AD (2014) Myelin basic protein and a multiple sclerosis-related MBP-peptide bind to oligonucleotides. Mol Ther 3:e192
Rozenblum GT, Kaufman T, Vitullo AD (2014) Myelin basic protein and a multiple sclerosis-related MBP-peptide bind to oligonucleotides. Mol Ther 3:e192
Ruck T, Bittner S, Wiendl H, Meuth SG (2015) Alemtuzumab in multiple sclerosis: mechanism of action and beyond. Int J Mol Sci 16(7):16414–16439
Rudick RA (1994) Betaseron for multiple sclerosis: implications for therapeutics. Arch Neurol 51(2):125–128
Sahraian MA, Radue EW, Haller S, Kappos L (2010) Black holes in multiple sclerosis: definition, evolution, and clinical correlations. Acta Neurol Scand 122(1):1–8
Salehi Z, Doosti R, Beheshti M, Janzamin E, Sahraian MA, Izad M (2016) Differential frequency of CD8+ T Cell subsets in multiple sclerosis patients with various clinical patterns. PloS one 11(7):e0159565
Salzer J, Svenningsson R, Alping P, Novakova L, Björck A, Fink K, Islam-Jakobsson P, Malmeström C, Axelsson M, Vågberg M (2016) Rituximab in multiple sclerosis: a retrospective observational study on safety and efficacy. Neurology 87(20):2074–2081
Salzman J, Chen RE, Olsen MN, Wang PL, Brown PO (2013) Cell-type specific features of circular RNA expression. PLoS Genet 9(9):e1003777
Sandyk R (1995) Weak electromagnetic fields restore dream recall in patients with multiple sclerosis. Int J Neurosci 82(1–2):113–125
Sandyk R (1996) Treatment with electromagnetic field alters the clinical course of chronic progressive multiple sclerosis–a case report. Int J Neurosci 88(1–2):75–82
Sanghvi YS (2011) A status update of modified oligonucleotides for chemotherapeutics applications. Curr Protoc Nucleic Acid Chem. https://doi.org/10.1002/0471142700.nc0401s46
Santoro M, Nociti V, Lucchini M, De Fino C, Losavio FA, Mirabella M (2016) Expression profile of long non-coding RNAs in serum of patients with multiple sclerosis. J Mol Neurosci 59(1):18–23
Selmaj K, Kowal C, Walczak A, Nowicka J, Raine CS (2000) Naked DNA vaccination differentially modulates autoimmune responses in experimental autoimmune encephalomyelitis. J Neuroimmunol 111(1–2):34–44
Sibbald B (2001) Death but one unintended consequence of gene-therapy trial. Can Med Assoc J 164(11):1612
Silverman SK (2016) Catalytic DNA: scope, applications, and biochemistry of deoxyribozymes. Trends Biochem Sci 41(7):595–609
Sioud M, Leirdal M (2000) Design of nuclease resistant protein kinase calpha DNA enzymes with potential therapeutic application. J Mol Biol 296(3):937–947
Sipe JC (2005) Cladribine for multiple sclerosis: review and current status. Expert Rev Neurother 5(6):721–727
Smestad J, Maher LJ 3rd (2013) Ion-dependent conformational switching by a DNA aptamer that induces remyelination in a mouse model of multiple sclerosis. Nucleic Acids Res 41(2):1329–1342
Song K-M, Lee S, Ban C (2012) Aptamers and their biological applications. Sensors 12(1):612–631
Sonobe Y, Li H, Jin S, Kishida S, Kadomatsu K, Takeuchi H, Mizuno T, Suzumura A (2012) Midkine inhibits inducible regulatory T cell differentiation by suppressing the development of tolerogenic dendritic cells. J Immunol 188(6):2602–2611
Sridharan K, Gogtay NJ (2016) Therapeutic nucleic acids: current clinical status. Br J Clin Pharmacol 82(3):659–672
Statello L, Guo C-J, Chen L-L, Huarte M (2021) Gene regulation by long non-coding RNAs and its biological functions. Nat Rev Mol Cell Biol 22(2):96–118
Stephenson ML, Zamecnik PC (1978) Inhibition of Rous sarcoma viral RNA translation by a specific oligodeoxyribonucleotide. Proc Natl Acad Sci USA 75(1):285–288
Suzuki H, Zuo Y, Wang J, Zhang MQ, Malhotra A, Mayeda A (2006) Characterization of RNase R-digested cellular RNA source that consists of lariat and circular RNAs from pre-mRNA splicing. Nucleic Acids Res 34(8):e63
Taghizadeh E, Taheri F, Samadian MM, Soudyab M, Abi A (2020) Role of long non-coding RNAs (LncRNAs) in multiple sclerosis: a brief review. Neurol Sci. https://doi.org/10.1007/s10072-020-04425-2
Tanasescu R, Ionete C, Chou IJ, Constantinescu CS (2014) Advances in the treatment of relapsing-remitting multiple sclerosis. Biomed J 37(2):41–49
Tuerk C, Gold L (1990) Systematic evolution of ligands by exponential enrichment: RNA ligands to bacteriophage T4 DNA polymerase. Science (new York, NY) 249(4968):505–510
Uszczynska-Ratajczak B, Lagarde J, Frankish A, Guigó R, Johnson R (2018) Towards a complete map of the human long non-coding RNA transcriptome. Nat Rev Genet 19(9):535–548
Veauthier C, Radbruch H, Gaede G, Pfueller CF, Dorr J, Bellmann-Strobl J, Wernecke KD, Zipp F, Paul F, Sieb JP (2011) Fatigue in multiple sclerosis is closely related to sleep disorders: a polysomnographic cross-sectional study. Mult Scler (houndmills, Basingstoke, England) 17(5):613–622
Voge NV, Alvarez E (2019) Monoclonal antibodies in multiple sclerosis: present and future. Biomedicines 7(1):20
Vorobjeva MA, Krasitskaya VV, Fokina AA, Timoshenko VV, Nevinsky GA, Venyaminova AG, Frank LA (2014) RNA aptamer against autoantibodies associated with multiple sclerosis and bioluminescent detection probe on its basis. Anal Chem 86(5):2590–2594
Wang J, Takeuchi H, Sonobe Y, Jin S, Mizuno T, Miyakawa S, Fujiwara M, Nakamura Y, Kato T, Muramatsu H et al (2008) Inhibition of midkine alleviates experimental autoimmune encephalomyelitis through the expansion of regulatory T cell population. Proc Natl Acad Sci USA 105(10):3915–3920
Wang CJ, Qu CQ, Zhang J, Fu PC, Guo SG, Tang RH (2014) Lingo-1 inhibited by RNA interference promotes functional recovery of experimental autoimmune encephalomyelitis. Anat Rec (Hoboken, NJ: 2007) 297(12):2356–2363
Warrington AE, Asakura K, Bieber AJ, Ciric B, Van Keulen V, Kaveri SV, Kyle RA, Pease LR, Rodriguez M (2000) Human monoclonal antibodies reactive to oligodendrocytes promote remyelination in a model of multiple sclerosis. Proc Natl Acad Sci USA 97(12):6820–6825
Watts JK, Corey DR (2012) Silencing disease genes in the laboratory and the clinic. J Pathol 226(2):365–379
Welton JL, Loveless S, Stone T, von Ruhland C, Robertson NP, Clayton A (2017) Cerebrospinal fluid extracellular vesicle enrichment for protein biomarker discovery in neurological disease; multiple sclerosis. J Extracell Vesicles 6(1):1369805
Weng Y, Huang Q, Li C, Yang Y, Wang X, Yu J, Huang Y, Liang X-J (2020) Improved nucleic acid therapy with advanced nanoscale biotechnology. Mol Ther Nucleic Acids 19:581–601
Wilbanks B, Smestad J, Heider RM, Warrington AE, Rodriguez M, Maher LJ (2019) Optimization of a 40-mer Antimyelin DNA Aptamer Identifies a 20-mer with Enhanced Properties for Potential Multiple Sclerosis Therapy. Nucleic Acid Ther. https://doi.org/10.1089/nat.2018.0776
Wingerchuk DM, Carter JL (2014) Multiple sclerosis: current and emerging disease-modifying therapies and treatment strategies. Mayo Clin Proc 89(2):225–240
Wu G-C, Pan H-F, Leng R-X, Wang D-G, Li X-P, Li X-M, Ye D-Q (2015) Emerging role of long noncoding RNAs in autoimmune diseases. Autoimmun Rev 14(9):798–805
Yan Y, Ding X, Li K, Ciric B, Wu S, Xu H, Gran B, Rostami A, Zhang GX (2012) CNS-specific therapy for ongoing EAE by silencing IL-17 pathway in astrocytes. Mol Ther 20(7):1338–1348
Yan Y, Li Z, Zhang GX, Williams MS, Carey GB, Zhang J, Rostami A, Xu H (2013) Anti-MS4a4B treatment abrogates MS4a4B-mediated protection in T cells and ameliorates experimental autoimmune encephalomyelitis. Apoptosis 18(9):1106–1119
Yang Y, Liu Y, Wei P, Peng H, Winger R, Hussain RZ, Ben LH, Cravens PD, Gocke AR, Puttaparthi K et al (2010) Silencing Nogo-A promotes functional recovery in demyelinating disease. Ann Neurol 67(4):498–507
Yang C, He D, Yin C, Tan J (2015) Inhibition of interferon regulatory factor 4 suppresses Th1 and Th17 cell differentiation and ameliorates experimental autoimmune encephalomyelitis. Scand J Immunol 82(4):345–351
Yoshimura S, Ochi H, Isobe N, Matsushita T, Motomura K, Matsuoka T, Minohara M, Kira J-I (2010) Altered production of brain-derived neurotrophic factor by peripheral blood immune cells in multiple sclerosis. Mult Scler J 16(10):1178–1188
Yuan J-H, Yang F, Wang F, Ma J-Z, Guo Y-J, Tao Q-F, Liu F, Pan W, Wang T-T, Zhou C-C (2014) A long noncoding RNA activated by TGF-β promotes the invasion-metastasis cascade in hepatocellular carcinoma. Cancer Cell 25(5):666–681
Zhang F, Liu G, Bu Y, Ma X, Hao J (2016a) Expression profile of long noncoding RNAs and mRNAs in peripheral blood mononuclear cells from myasthenia gravis patients. J Neuroimmunol 299:124–129
Zhang Y, Yan L, Cao Y, Kong G, Lin C (2016b) Long noncoding RNA BDNF-AS protects local anesthetic induced neurotoxicity in dorsal root ganglion neurons. Biomed Pharmacother 80:207–212
Zhang H, Bi J, Yi H, Fan T, Ruan Q, Cai L, Chen YH, Wan X (2017) Silencing c-Rel in macrophages dampens Th1 and Th17 immune responses and alleviates experimental autoimmune encephalomyelitis in mice. Immunol Cell Biol 95(7):593–600
Zhou YY, Hou GQ, He SW, Xiao Z, Xu HJ, Qiu YT, Jiang S, Zheng H, Li ZY (2015) Psora-4, a Kv1.3 blocker, enhances differentiation and maturation in neural progenitor cells. CNS Neurosci Ther 21(7):558–567
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Baharlooi, H., Mansourabadi, A.H., Minbashi Moeini, M. et al. Nucleic Acids as Novel Therapeutic Modalities to Address Multiple Sclerosis Onset and Progression. Cell Mol Neurobiol 42, 2611–2627 (2022). https://doi.org/10.1007/s10571-021-01158-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-021-01158-4