Abstract
L-type Ca2+ channels (LTCCs) are key elements in electromechanical coupling in striated muscles and formation of neuromuscular junctions (NMJs). However, the significance of LTCCs in regulation of neurotransmitter release is still far from understanding. Here, we found that LTCCs can increase evoked neurotransmitter release (especially asynchronous component) and spontaneous exocytosis in two functionally different compartment of the frog NMJ, namely distal and proximal parts. The effects of LTCC blockage on evoked and spontaneous release as well as timing of exocytotic events were prevented by inhibition of either protein kinase C (PKC) or P2Y receptors (P2Y-Rs). Hence, endogenous signaling via P2Y-R/PKC axis can sustain LTCC activity. Application of ATP, a co-neurotransmitter able to activate P2Y-Rs, suppressed both evoked and spontaneous exocytosis in distal and proximal parts. Surprisingly, inhibition of LTCCs (but not PKC) decreased the negative action of exogenous ATP on evoked (only in distal part) and spontaneous exocytosis. Lipid raft disruption suppressed (1) action of LTCC antagonist on neurotransmitter release selectively in distal region and (2) contribution of LTCCs in depressant effect of ATP on evoked and spontaneous release. Thus, LTCCs can enhance and desynchronize neurotransmitter release at basal conditions (without ATP addition), but contribute to ATP-mediated decrease in the exocytosis. The former action of LTCCs relies on P2Y-R/PKC axis, whereas the latter is triggered by exogenous ATP and PKC-independent. Furthermore, relevance of lipid rafts for LTCC function as well as LTCCs for ATP effects is different in distal and proximal part of the NMJ.
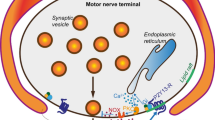
Graphic Abstract







Similar content being viewed by others
Data Availability
All mentioned data are represented in the main manuscript figures and supplementary figures. Other additional data will be made available on reasonable request.
Abbreviations
- ACh:
-
Acetylcholine
- AP:
-
Action potential
- CgTx:
-
Omega-conotoxin GVIA
- CTxB:
-
Cholera toxin B subunit
- EPP:
-
End-plate potential
- MEPP:
-
Miniature EPP
- LTCC:
-
L-type Ca2+ channel
- NMJ:
-
Neuromuscular junction
- NTCC:
-
N-type Ca2+ channel
- PKC:
-
Protein kinase C
- P2Y-R:
-
P2Y-receptor
- SV:
-
Synaptic vesicle
- VGCC:
-
Voltage-gated Ca2+ channel
References
Ando K, Kudo Y, Takahashi M (2005) Negative regulation of neurotransmitter release by calpain: a possible involvement of specific SNAP-25 cleavage. J Neurochem 94(3):651–658. https://doi.org/10.1111/j.1471-4159.2005.03160.x
Arenson MS, Evans SC (2001) Activation of protein kinase C increases acetylcholine release from frog motor nerves by a direct action on L-type Ca(2+) channels and apparently not by depolarisation of the terminal. Neuroscience 104(4):1157–1164. https://doi.org/10.1016/s0306-4522(01)00114-2
Atchison WD, O’Leary SM (1987) BAY K 8644 increases release of acetylcholine at the murine neuromuscular junction. Brain Res 419(1–2):315–319. https://doi.org/10.1016/0006-8993(87)90599-3
Balezina OP, Bogacheva PO, Orlova TY (2007) Effect of L-type calcium channel blockers on activity of newly formed synapses in mice. Bull Exp Biol Med 143(2):171–174. https://doi.org/10.1007/s10517-007-0041-y
Branisteanu DD, Branisteanu DD, Covic A, Brailoiu E, Serban DN, Haulica ID (1989) Adenosine effects upon the spontaneous quantal transmitter release at the frog neuromuscular junction in the presence of protein kinase C-blocking and -activating agents. Neurosci Lett 98(1):96–100. https://doi.org/10.1016/0304-3940(89)90380-7
Bukcharaeva EA, Kim KC, Moravec J, Nikolsky EE, Vyskocil F (1999) Noradrenaline synchronizes evoked quantal release at frog neuromuscular junctions. J Physiol 517(Pt 3):879–888. https://doi.org/10.1111/j.1469-7793.1999.0879s.x
Calin-Jageman I, Lee A (2008) Ca(v)1 L-type Ca2+ channel signaling complexes in neurons. J Neurochem 105(3):573–583. https://doi.org/10.1111/j.1471-4159.2008.05286.x
Chipman PH, Schachner M, Rafuse VF (2014) Presynaptic NCAM is required for motor neurons to functionally expand their peripheral field of innervation in partially denervated muscles. J Neurosci 34(32):10497–10510. https://doi.org/10.1523/JNEUROSCI.0697-14.2014
Cinalli AR, Guarracino JF, Fernandez V, Roquel LI, Losavio AS (2013) Inosine induces presynaptic inhibition of acetylcholine release by activation of A3 adenosine receptors at the mouse neuromuscular junction. Br J Pharmacol 169(8):1810–1823. https://doi.org/10.1111/bph.12262
De Lorenzo S, Veggetti M, Muchnik S, Losavio A (2006) Presynaptic inhibition of spontaneous acetylcholine release mediated by P2Y receptors at the mouse neuromuscular junction. Neuroscience 142(1):71–85. https://doi.org/10.1016/j.neuroscience.2006.05.062
Del Castillo J, Katz B (1954) The effect of magnesium on the activity of motor nerve endings. J Physiol 124(3):553–559. https://doi.org/10.1113/jphysiol.1954.sp005128
Dittrich M, Homan AE, Meriney SD (2018) Presynaptic mechanisms controlling calcium-triggered transmitter release at the neuromuscular junction. Curr Opin Physiol 4:15–24. https://doi.org/10.1016/j.cophys.2018.03.004
Dolphin AC, Lee A (2020) Presynaptic calcium channels: specialized control of synaptic neurotransmitter release. Nat Rev Neurosci 21(4):213–229. https://doi.org/10.1038/s41583-020-0278-2
Flink MT, Atchison WD (2003) Iberiotoxin-induced block of Ca2+-activated K+ channels induces dihydropyridine sensitivity of ACh release from mammalian motor nerve terminals. J Pharmacol Exp Ther 305(2):646–652. https://doi.org/10.1124/jpet.102.046102
Fu WM, Huang FL (1994) L-type Ca2+ channel is involved in the regulation of spontaneous transmitter release at developing neuromuscular synapses. Neuroscience 58(1):131–140. https://doi.org/10.1016/0306-4522(94)90160-0
Galkin AV, Giniatullin RA, Mukhtarov MR, Svandova I, Grishin SN, Vyskocil F (2001) ATP but not adenosine inhibits nonquantal acetylcholine release at the mouse neuromuscular junction. Eur J Neurosci 13(11):2047–2053. https://doi.org/10.1046/j.0953-816x.2001.01582.x
Gaydukov AE, Melnikova SN, Balezina OP (2009) Facilitation of acetylcholine secretion in mouse motor synapses caused by calcium release from depots upon activation of L-type calcium channels. Bull Exp Biol Med 148(2):163–166. https://doi.org/10.1007/s10517-009-0678-9
Gaydukov AE, Tarasova EO, Balezina OP (2013) Calcium-dependent phosphatase calcineurin downregulates evoked neurotransmitter release in neuromuscular junctions of mice. Neurochem J 7(1):29–33. https://doi.org/10.1134/s1819712413010030
Ginebaugh SP, Cyphers ED, Lanka V, Ortiz G, Miller EW, Laghaei R, Meriney SD (2020) The frog motor nerve terminal has very brief action potentials and three electrical regions predicted to differentially control transmitter release. J Neurosci. https://doi.org/10.1523/JNEUROSCI.2415-19.2020
Giniatullin AR, Grishin SN, Sharifullina ER, Petrov AM, Zefirov AL, Giniatullin RA (2005) Reactive oxygen species contribute to the presynaptic action of extracellular ATP at the frog neuromuscular junction. J Physiol 565(Pt 1):229–242. https://doi.org/10.1113/jphysiol.2005.084186
Giniatullin A, Petrov A, Giniatullin R (2015) The involvement of P2Y12 receptors, NADPH oxidase, and lipid rafts in the action of extracellular ATP on synaptic transmission at the frog neuromuscular junction. Neuroscience 285:324–332. https://doi.org/10.1016/j.neuroscience.2014.11.039
Guarracino JF, Cinalli AR, Fernandez V, Roquel LI, Losavio AS (2016) P2Y13 receptors mediate presynaptic inhibition of acetylcholine release induced by adenine nucleotides at the mouse neuromuscular junction. Neuroscience 326:31–44. https://doi.org/10.1016/j.neuroscience.2016.03.066
Kaeser PS, Regehr WG (2014) Molecular mechanisms for synchronous, asynchronous, and spontaneous neurotransmitter release. Annu Rev Physiol 76:333–363. https://doi.org/10.1146/annurev-physiol-021113-170338
Kaplan MM, Sultana N, Benedetti A, Obermair GJ, Linde NF, Papadopoulos S, Dayal A, Grabner M, Flucher BE (2018) Calcium influx and release cooperatively regulate AChR patterning and motor axon outgrowth during neuromuscular junction formation. Cell Rep 23(13):3891–3904. https://doi.org/10.1016/j.celrep.2018.05.085
Kasimov MR, Giniatullin AR, Zefirov AL, Petrov AM (2015) Effects of 5alpha-cholestan-3-one on the synaptic vesicle cycle at the mouse neuromuscular junction. Biochim Biophys Acta 1851(5):674–685. https://doi.org/10.1016/j.bbalip.2015.02.012
Kasimov MR, Zakyrjanova GF, Giniatullin AR, Zefirov AL, Petrov AM (2016) Similar oxysterols may lead to opposite effects on synaptic transmission: olesoxime versus 5alpha-cholestan-3-one at the frog neuromuscular junction. Biochim Biophys Acta 1861(7):606–616. https://doi.org/10.1016/j.bbalip.2016.04.010
Katz B, Miledi R (1965) The measurement of synaptic delay, and the time course of acetylcholine release at the neuromuscular junction. Proc R Soc Lond B Biol Sci 161:483–495. https://doi.org/10.1098/rspb.1965.0016
Katz E, Ferro PA, Weisz G, Uchitel OD (1996) Calcium channels involved in synaptic transmission at the mature and regenerating mouse neuromuscular junction. J Physiol 497(Pt 3):687–697. https://doi.org/10.1113/jphysiol.1996.sp021800
Ko CP, Robitaille R (2015) Perisynaptic schwann cells at the neuromuscular synapse: adaptable, multitasking glial cells. Cold Spring Harb Perspect Biol 7(10):a020503. https://doi.org/10.1101/cshperspect.a020503
Krivoi II, Petrov AM (2019) Cholesterol and the safety factor for neuromuscular transmission. Int J Mol Sci. https://doi.org/10.3390/ijms20051046
Liu Q, Chen B, Ge Q, Wang ZW (2007) Presynaptic Ca2+/calmodulin-dependent protein kinase II modulates neurotransmitter release by activating BK channels at Caenorhabditis elegans neuromuscular junction. J Neurosci 27(39):10404–10413. https://doi.org/10.1523/JNEUROSCI.5634-06.2007
Liu Y, Sugiura Y, Sudhof TC, Lin W (2019) Ablation of all synaptobrevin vSNAREs blocks evoked but not spontaneous neurotransmitter release at neuromuscular synapses. J Neurosci 39(31):6049–6066. https://doi.org/10.1523/JNEUROSCI.0403-19.2019
Mallart A (1984) Presynaptic currents in frog motor endings. Pflugers Arch 400(1):8–13. https://doi.org/10.1007/bf00670529
Marcantoni A, Baldelli P, Hernandez-Guijo JM, Comunanza V, Carabelli V, Carbone E (2007) L-type calcium channels in adrenal chromaffin cells: role in pace-making and secretion. Cell Calcium 42(4–5):397–408. https://doi.org/10.1016/j.ceca.2007.04.015
Margheri G, D’Agostino R, Trigari S, Sottini S, Del Rosso M (2014) The beta-subunit of cholera toxin has a high affinity for ganglioside GM1 embedded into solid supported lipid membranes with a lipid raft-like composition. Lipids 49(2):203–206. https://doi.org/10.1007/s11745-013-3845-8
Mercer AJ, Chen M, Thoreson WB (2011) Lateral mobility of presynaptic L-type calcium channels at photoreceptor ribbon synapses. J Neurosci 31(12):4397–4406. https://doi.org/10.1523/JNEUROSCI.5921-10.2011
Mukhutdinova KA, Kasimov MR, Giniatullin AR, Zakyrjanova GF, Petrov AM (2018) 24S-hydroxycholesterol suppresses neuromuscular transmission in SOD1(G93A) mice: a possible role of NO and lipid rafts. Mol Cell Neurosci 88:308–318. https://doi.org/10.1016/j.mcn.2018.03.006
Oliveira L, Correia-de-Sa P (2005) Protein kinase A and Ca(v)1 (L-Type) channels are common targets to facilitatory adenosine A2A and muscarinic M1 receptors on rat motoneurons. Neurosignals 14(5):262–272. https://doi.org/10.1159/000088642
Pagani MR, Reisin RC, Uchitel OD (2006) Calcium signaling pathways mediating synaptic potentiation triggered by amyotrophic lateral sclerosis IgG in motor nerve terminals. J Neurosci 26(10):2661–2672. https://doi.org/10.1523/JNEUROSCI.4394-05.2006
Palma AG, Muchnik S, Losavio AS (2011) Excitatory effect of the A2A adenosine receptor agonist CGS-21680 on spontaneous and K+-evoked acetylcholine release at the mouse neuromuscular junction. Neuroscience 172:164–176. https://doi.org/10.1016/j.neuroscience.2010.10.015
Park Y, Kim KT (2009) Dominant role of lipid rafts L-type calcium channel in activity-dependent potentiation of large dense-core vesicle exocytosis. J Neurochem 110(2):520–529. https://doi.org/10.1111/j.1471-4159.2009.06148.x
Perissinotti PP, Giugovaz Tropper B, Uchitel OD (2008) L-type calcium channels are involved in fast endocytosis at the mouse neuromuscular junction. Eur J Neurosci 27(6):1333–1344. https://doi.org/10.1111/j.1460-9568.2008.06113.x
Petrov AM, Zefirov AL (2013) Cholesterol and lipid rafts in the biological membranes. Role in the release, reception and ion channel functions. Usp Fiziol Nauk 44(1):17–38
Petrov AM, Kudryashova KE, Odnoshivkina YG, Zefirov AL (2011a) Cholesterol and lipid rafts in the plasma membrane of nerve terminal and membrane of synaptic vesicles. Neurochem J 5(1):13–19. https://doi.org/10.1134/s1819712411010089
Petrov AM, Naumenko NV, Uzinskaya KV, Giniatullin AR, Urazaev AK, Zefirov AL (2011b) Increased non-quantal release of acetylcholine after inhibition of endocytosis by methyl-beta-cyclodextrin: the role of vesicular acetylcholine transporter. Neuroscience 186:1–12. https://doi.org/10.1016/j.neuroscience.2011.04.051
Petrov AM, Yakovleva AA, Zefirov AL (2014) Role of membrane cholesterol in spontaneous exocytosis at frog neuromuscular synapses: reactive oxygen species-calcium interplay. J Physiol 592(22):4995–5009. https://doi.org/10.1113/jphysiol.2014.279695
Petrov AM, Zakyrjanova GF, Yakovleva AA, Zefirov AL (2015) Inhibition of protein kinase C affects on mode of synaptic vesicle exocytosis due to cholesterol depletion. Biochem Biophys Res Commun 456(1):145–150. https://doi.org/10.1016/j.bbrc.2014.11.049
Robitaille R, Tremblay JP (1991) Non-uniform responses to Ca2+ along the frog neuromuscular junction: effects on the probability of spontaneous and evoked transmitter release. Neuroscience 40(2):571–585. https://doi.org/10.1016/0306-4522(91)90142-b
Robitaille R, Bourque MJ, Vandaele S (1996) Localization of L-type Ca2+ channels at perisynaptic glial cells of the frog neuromuscular junction. J Neurosci 16(1):148–158
Rodrigues HA, Lima RF, Fonseca Mde C, Amaral EA, Martinelli PM, Naves LA, Gomez MV, Kushmerick C, Prado MA, Guatimosim C (2013) Membrane cholesterol regulates different modes of synaptic vesicle release and retrieval at the frog neuromuscular junction. Eur J Neurosci 38(7):2978–2987. https://doi.org/10.1111/ejn.12300
Rosello-Busquets C, de la Oliva N, Martinez-Marmol R, Hernaiz-Llorens M, Pascual M, Muhaisen A, Navarro X, Del Valle J, Soriano E (2019) Cholesterol depletion regulates axonal growth and enhances central and peripheral nerve regeneration. Front Cell Neurosci 13:40. https://doi.org/10.3389/fncel.2019.00040
Ruiz R, Casanas JJ, Torres-Benito L, Cano R, Tabares L (2010) Altered intracellular Ca2+ homeostasis in nerve terminals of severe spinal muscular atrophy mice. J Neurosci 30(3):849–857. https://doi.org/10.1523/JNEUROSCI.4496-09.2010
Santafe MM, Lanuza MA, Garcia N, Tomas J (2006) Muscarinic autoreceptors modulate transmitter release through protein kinase C and protein kinase A in the rat motor nerve terminal. Eur J Neurosci 23(8):2048–2056. https://doi.org/10.1111/j.1460-9568.2006.04753.x
Sokolova E, Grishin S, Shakirzyanova A, Talantova M, Giniatullin R (2003) Distinct receptors and different transduction mechanisms for ATP and adenosine at the frog motor nerve endings. Eur J Neurosci 18(5):1254–1264. https://doi.org/10.1046/j.1460-9568.2003.02835.x
Sosa LJ, Malter JS, Hu J, Bustos Plonka F, Oksdath M, Nieto Guil AF, Quiroga S, Pfenninger KH (2016) Protein interacting with NIMA (never in mitosis A)-1 regulates axonal growth cone adhesion and spreading through myristoylated alanine-rich C kinase substrate isomerization. J Neurochem 137(5):744–755. https://doi.org/10.1111/jnc.13612
Subramanian J, Dye L, Morozov A (2013) Rap1 signaling prevents L-type calcium channel-dependent neurotransmitter release. J Neurosci 33(17):7245–7252. https://doi.org/10.1523/JNEUROSCI.5963-11.2013
Sugiura Y, Ko CP (1997) Novel modulatory effect of L-type calcium channels at newly formed neuromuscular junctions. J Neurosci 17(3):1101–1111
Sun X, Gu XQ, Haddad GG (2003) Calcium influx via L- and N-type calcium channels activates a transient large-conductance Ca2+-activated K+ current in mouse neocortical pyramidal neurons. J Neurosci 23(9):3639–3648
Tsentsevitsky AN, Petrov AM (2021) Synaptic mechanisms of cadmium neurotoxicity. Neural Regen Res 16(9):1762–1763. https://doi.org/10.4103/1673-5374.306067
Tsentsevitsky A, Nikolsky E, Giniatullin R, Bukharaeva E (2011) Opposite modulation of time course of quantal release in two parts of the same synapse by reactive oxygen species. Neuroscience 189:93–99. https://doi.org/10.1016/j.neuroscience.2011.05.033
Tsentsevitsky AN, Zakyrjanova GF, Petrov AM (2020a) Cadmium desynchronizes neurotransmitter release in the neuromuscular junction: key role of ROS. Free Radic Biol Med 155:19–28. https://doi.org/10.1016/j.freeradbiomed.2020.05.017
Tsentsevitsky AN, Zakyrjanova GF, Petrov AM, Kovyazina IV (2020b) Breakdown of phospholipids and the elevated nitric oxide are involved in M3 muscarinic regulation of acetylcholine secretion in the frog motor synapse. Biochem Biophys Res Commun 524(3):589–594. https://doi.org/10.1016/j.bbrc.2020.01.112
Urbano FJ, Depetris RS, Uchitel OD (2001) Coupling of L-type calcium channels to neurotransmitter release at mouse motor nerve terminals. Pflugers Arch 441(6):824–831. https://doi.org/10.1007/s004240000489
Wang X, Burke SRA, Talmadge RJ, Voss AA, Rich MM (2020) Depressed neuromuscular transmission causes weakness in mice lacking BK potassium channels. J Gen Physiol. https://doi.org/10.1085/jgp.201912526
Xu JW, Slaughter MM (2005) Large-conductance calcium-activated potassium channels facilitate transmitter release in salamander rod synapse. J Neurosci 25(33):7660–7668. https://doi.org/10.1523/JNEUROSCI.1572-05.2005
Yang L, Wang B, Long C, Wu G, Zheng H (2007) Increased asynchronous release and aberrant calcium channel activation in amyloid precursor protein deficient neuromuscular synapses. Neuroscience 149(4):768–778. https://doi.org/10.1016/j.neuroscience.2007.08.025
Zakyrjanova GF, Tsentsevitsky AN, Kuznetsova EA, Petrov AM (2021) Immune-related oxysterol modulates neuromuscular transmission via non-genomic liver X receptor-dependent mechanism. Free Radic Biol Med 174:121–134. https://doi.org/10.1016/j.freeradbiomed.2021.08.013
Zefirov AL, Grigor’ev PN, Petrov AM, Minlebaev MG, Sitdikova GF (2003) Analysis of living motor nerve ending of a frog by endocytotic fluorescent marker FM 1–43. Tsitologiia 45(12):1163–1171
Funding
This study was supported in part by the Russian Science Foundation Grant #21-14-00044, https://rscf.ru/project/21-14-00044/ (3.2–3.5), and partially the government assignment for FRC Kazan Scientific Center of RAS (3.1).
Author information
Authors and Affiliations
Contributions
ANT produced and analyzed data. A.N.T. and A.M.P. interpreted results of experiments, designed, and supervised the research. A.M.P. wrote the manuscript. All the authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. All authors consent to publish the article.
Ethical Approval
The present study was conducted in compliance with the NIH Guide for the Care and Use of Laboratory Animals and the requirements of the EU Directive 2010/63/EU. The protocol of experiments was approved by the Ethical Committee of Kazan Medical University.
Informed Consent
This is not applicable. This manuscript does not contain any studies with human participants.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tsentsevitsky, A.N., Petrov, A.M. L-type Ca2+ Channels at Low External Calcium Differentially Regulate Neurotransmitter Release in Proximal–Distal Compartments of the Frog Neuromuscular Junction. Cell Mol Neurobiol 42, 2833–2847 (2022). https://doi.org/10.1007/s10571-021-01152-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-021-01152-w