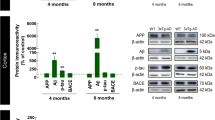
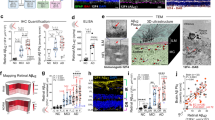
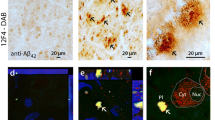
Abstract
Visual changes are some of the earliest symptoms that patients with Alzheimer’s disease (AD) experience. Pathophysiological processes such as amyloid-β plaque formation, vascular changes, neuroinflammation, and loss of retinal ganglion cells (RGCs) have been detected in the retina of AD patients and animal models. However, little is known about the molecular processes that underlie retinal neurodegeneration in AD. The cellular architecture and constant sensory activity of the retina impose high metabolic demands. We thus hypothesized that energy metabolism might be compromised in the AD retina similarly to what has been observed in the AD brain. To address this question, we explored cellular alterations and retinal metabolic activity in the 5 × FAD mouse model of AD. We used 8-month-old female 5 × FAD mice, in which the AD-related pathology has been shown to be apparent. We observed that RGC density is selectively affected in the retina of 5 × FAD mice. To map retinal metabolic activity, we incubated isolated retinal tissue with [U-13C] glucose and analyzed tissue extracts by gas chromatography-mass spectrometry. We found that the retinas of 5 × FAD mice exhibit glucose hypometabolism. Moreover, we detected decreased glutamine synthesis in 5 × FAD retinas but no changes in the expression of markers of Müller glia, the main glial cell type responsible for glutamate uptake and glutamine synthesis in the retina. These findings suggest that AD presents with metabolic alterations not only in the brain but also in the retina that may be detrimental to RGC activity and survival, potentially leading to the visual impairments that AD patients suffer.





Similar content being viewed by others
Data Availability
All data of this study are available upon request to the corresponding authors.
References
Andersen JV, Christensen SK, Aldana BI, Nissen JD, Tanila H, Waagepetersen HS (2017) Alterations in cerebral cortical glucose and glutamine metabolism precedes amyloid plaques in the APPswe/PSEN1dE9 mouse model of Alzheimer’s disease. Neurochem Res 42(6):1589–1598. https://doi.org/10.1007/s11064-016-2070-2
Andersen JV, Christensen SK, Westi EW, Diaz-delCastillo M, Tanila H, Schousboe A, Waagepetersen HS (2021) Deficient astrocyte metabolism impairs glutamine synthesis and neurotransmitter homeostasis in a mouse model of Alzheimer’s disease. Neurobiol Dis 148:105198. https://doi.org/10.1016/j.nbd.2020.105198
Antonetti DA, Barber AJ, Bronson SK, Freeman WM, Gardner TW, Jefferson LS, Simpson IA (2006) Diabetic retinopathy: seeing beyond glucose-induced microvascular disease. Diabetes 55(9):2401–2411. https://doi.org/10.2337/db05-1635
Atri A (2019) The Alzheimer’s disease clinical spectrum: diagnosis and management. Med Clin North Am 103(2):263–293. https://doi.org/10.1016/j.mcna.2018.10.009
Bartsch U, Faissner A, Trotter J, Dörries U, Bartsch S, Mohajeri H, Schachner M (1994) Tenascin demarcates the boundary between the myelinated and nonmyelinated part of retinal ganglion cell axons in the developing and adult mouse. J Neurosci 14(8):4756–4768. https://doi.org/10.1523/jneurosci.14-08-04756.1994
Berisha F, Feke GT, Trempe CL, McMeel JW, Schepens CL (2007) Retinal abnormalities in early Alzheimer’s disease. Invest Ophthalmol Vis Sci 48(5):2285–2289. https://doi.org/10.1167/iovs.06-1029
Biemann K (1963) Mass Spectrometry. Annu Rev Biochem 32:755–780. https://doi.org/10.1146/annurev.bi.32.070163.003543
Bouter C, Bouter Y (2019) (18)F-FDG-PET in mouse models of Alzheimer’s disease. Front Med 6:71. https://doi.org/10.3389/fmed.2019.00071
Bringmann A, Pannicke T, Biedermann B, Francke M, Iandiev I, Grosche J, Reichenbach A (2009) Role of retinal glial cells in neurotransmitter uptake and metabolism. Neurochem Int 54(3–4):143–160. https://doi.org/10.1016/j.neuint.2008.10.014
Bringmann A, Grosche A, Pannicke T, Reichenbach A (2013) GABA and glutamate uptake and metabolism in retinal glial (Muller) cells. Front Endocrinol 4:48. https://doi.org/10.3389/fendo.2013.00048
Bui BV, Hu RG, Acosta ML, Donaldson P, Vingrys AJ, Kalloniatis M (2009) Glutamate metabolic pathways and retinal function. J Neurochem 111(2):589–599. https://doi.org/10.1111/j.1471-4159.2009.06354.x
Butterfield DA, Halliwell B (2019) Oxidative stress, dysfunctional glucose metabolism and Alzheimer disease. Nat Rev Neurosci 20(3):148–160. https://doi.org/10.1038/s41583-019-0132-6
Chételat G, Arbizu J, Barthel H, Garibotto V, Law I, Morbelli S, Drzezga A (2020) Amyloid-PET and (18)F-FDG-PET in the diagnostic investigation of Alzheimer’s disease and other dementias. Lancet Neurol 19(11):951–962. https://doi.org/10.1016/s1474-4422(20)30314-8
Cheung CY, Ong YT, Hilal S, Ikram MK, Low S, Ong YL, Wong TY (2015) Retinal ganglion cell analysis using high-definition optical coherence tomography in patients with mild cognitive impairment and Alzheimer’s disease. J Alzheimers Dis 45(1):45–56. https://doi.org/10.3233/jad-141659
Chiquita S, Rodrigues-Neves AC, Baptista FI, Carecho R, Moreira PI, Castelo-Branco M, Ambrósio AF (2019) The retina as a window or mirror of the brain changes detected in Alzheimer’s disease: critical aspects to unravel. Mol Neurobiol 56(8):5416–5435. https://doi.org/10.1007/s12035-018-1461-6
Cunha JP, Moura-Coelho N, Proença RP, Dias-Santos A, Ferreira J, Louro C, Castanheira-Dinis A (2016) Alzheimer’s disease: a review of its visual system neuropathology. Optical coherence tomography-a potential role as a study tool in vivo. Graefes Arch Clin Exp Ophthalmol 254(11):2079–2092. https://doi.org/10.1007/s00417-016-3430-y
De Strooper B, Karran E (2016) The cellular phase of Alzheimer’s disease. Cell 164(4):603–615. https://doi.org/10.1016/j.cell.2015.12.056
Doustar J, Torbati T, Black KL, Koronyo Y, Koronyo-Hamaoui M (2017) Optical coherence tomography in Alzheimer’s disease and other neurodegenerative diseases. Front Neurol 8:701. https://doi.org/10.3389/fneur.2017.00701
Dubois B, Feldman HH, Jacova C, Hampel H, Molinuevo JL, Blennow K, Cummings JL (2014) Advancing research diagnostic criteria for Alzheimer’s disease: the IWG-2 criteria. Lancet Neurol 13(6):614–629. https://doi.org/10.1016/s1474-4422(14)70090-0
Ehinger B (1977) Glial and neuronal uptake of GABA, glutamic acid, glutamine and glutathione in the rabbit retina. Exp Eye Res 25(3):221–234. https://doi.org/10.1016/0014-4835(77)90089-6
Eimer WA, Vassar R (2013) Neuron loss in the 5XFAD mouse model of Alzheimer’s disease correlates with intraneuronal Aβ42 accumulation and caspase-3 activation. Mol Neurodegener 8:2. https://doi.org/10.1186/1750-1326-8-2
Gibson GE, Shi Q (2010) A mitocentric view of Alzheimer’s disease suggests multi-faceted treatments. J Alzheimers Dis 20(Suppl 2):S591-607. https://doi.org/10.3233/jad-2010-100336
Gordon BA, Blazey TM, Su Y, Hari-Raj A, Dincer A, Flores S, Benzinger TLS (2018) Spatial patterns of neuroimaging biomarker change in individuals from families with autosomal dominant Alzheimer’s disease: a longitudinal study. Lancet Neurol 17(3):241–250. https://doi.org/10.1016/s1474-4422(18)30028-0
Grimaldi A, Brighi C, Peruzzi G, Ragozzino D, Bonanni V, Limatola C, Di Angelantonio S (2018) Inflammation, neurodegeneration and protein aggregation in the retina as ocular biomarkers for Alzheimer’s disease in the 3xTg-AD mouse model. Cell Death Dis 9(6):685. https://doi.org/10.1038/s41419-018-0740-5
Harris JJ, Attwell D (2012) The energetics of CNS white matter. J Neurosci 32(1):356–371. https://doi.org/10.1523/jneurosci.3430-11.2012
Hart NJ, Koronyo Y, Black KL, Koronyo-Hamaoui M (2016) Ocular indicators of Alzheimer’s: exploring disease in the retina. Acta Neuropathol 132(6):767–787. https://doi.org/10.1007/s00401-016-1613-6
Haydinger CD, Kittipassorn T, Peet DJ (2020) Power to see-drivers of aerobic glycolysis in the mammalian retina: a review. Clin Exp Ophthalmol 48(8):1057–1071. https://doi.org/10.1111/ceo.13833
Heneka MT, Carson MJ, El Khoury J, Landreth GE, Brosseron F, Feinstein DL, Kummer MP (2015) Neuroinflammation in Alzheimer’s disease. Lancet Neurol 14(4):388–405. https://doi.org/10.1016/s1474-4422(15)70016-5
Holtzman DM, Morris JC, Goate AM (2011) Alzheimer’s disease: the challenge of the second century. Sci Transl Med 3(77):7sr771. https://doi.org/10.1126/scitranslmed.3002369
Hunter JJ, Merigan WH, Schallek JB (2019) Imaging retinal activity in the living eye. Annu Rev Vis Sci 5:15–45. https://doi.org/10.1146/annurev-vision-091517-034239
Hynd MR, Scott HL, Dodd PR (2004) Glutamate-mediated excitotoxicity and neurodegeneration in Alzheimer’s disease. Neurochem Int 45(5):583–595. https://doi.org/10.1016/j.neuint.2004.03.007
Jang C, Chen L, Rabinowitz JD (2018) Metabolomics and isotope tracing. Cell 173(4):822–837. https://doi.org/10.1016/j.cell.2018.03.055
Koronyo-Hamaoui M, Koronyo Y, Ljubimov AV, Miller CA, Ko MK, Black KL, Farkas DL (2011) Identification of amyloid plaques in retinas from Alzheimer’s patients and noninvasive in vivo optical imaging of retinal plaques in a mouse model. Neuroimage 54(Suppl 1):S204-217. https://doi.org/10.1016/j.neuroimage.2010.06.020
Lau A, Tymianski M (2010) Glutamate receptors, neurotoxicity and neurodegeneration. Pflug Arch 460(2):525–542. https://doi.org/10.1007/s00424-010-0809-1
Lim JKH, Li QX, He Z, Vingrys AJ, Chinnery HR, Mullen J, Nguyen CTO (2020) Retinal functional and structural changes in the 5xFAD mouse model of Alzheimer’s disease. Front Neurosci 14:862. https://doi.org/10.3389/fnins.2020.00862
London A, Benhar I, Schwartz M (2013) The retina as a window to the brain-from eye research to CNS disorders. Nat Rev Neurol 9(1):44–53. https://doi.org/10.1038/nrneurol.2012.227
Macdonald IR, DeBay DR, Reid GA, O’Leary TP, Jollymore CT, Mawko G, Darvesh S (2014) Early detection of cerebral glucose uptake changes in the 5XFAD mouse. Curr Alzheimer Res 11(5):450–460. https://doi.org/10.2174/1567205011666140505111354
Magharious MM, D’Onofrio PM, Koeberle PD (2011) Optic nerve transection: a model of adult neuron apoptosis in the central nervous system. J Vis Exp. https://doi.org/10.3791/2241
Masland RH (2012) The neuronal organization of the retina. Neuron 76(2):266–280. https://doi.org/10.1016/j.neuron.2012.10.002
Masters CL, Bateman R, Blennow K, Rowe CC, Sperling RA, Cummings JL (2015) Alzheimer’s disease. Nat Rev Dis Primers 1:15056. https://doi.org/10.1038/nrdp.2015.56
Mawhinney TP, Robinett RS, Atalay A, Madson MA (1986) Analysis of amino acids as their tert.-butyldimethylsilyl derivatives by gas-liquid chromatography and mass spectrometry. J Chromatogr 358(1):231–242. https://doi.org/10.1016/s0021-9673(01)90333-4
McNair LF, Kornfelt R, Walls AB, Andersen JV, Aldana BI, Nissen JD, Waagepetersen HS (2017) Metabolic characterization of acutely isolated hippocampal and cerebral cortical slices using [U-(13)C]glucose and [1,2-(13)C]acetate as substrates. Neurochem Res 42(3):810–826. https://doi.org/10.1007/s11064-016-2116-5
Mirzaei N, Shi H, Oviatt M, Doustar J, Rentsendorj A, Fuchs DT, Koronyo-Hamaoui M (2020) Alzheimer’s retinopathy: seeing disease in the eyes. Front Neurosci 14:921. https://doi.org/10.3389/fnins.2020.00921
Mosconi L, Pupi A, De Leon MJ (2008) Brain glucose hypometabolism and oxidative stress in preclinical Alzheimer’s disease. Ann N Y Acad Sci 1147:180–195. https://doi.org/10.1196/annals.1427.007
Oakley H, Cole SL, Logan S, Maus E, Shao P, Craft J, Vassar R (2006) Intraneuronal beta-amyloid aggregates, neurodegeneration, and neuron loss in transgenic mice with five familial Alzheimer’s disease mutations: potential factors in amyloid plaque formation. J Neurosci 26(40):10129–10140. https://doi.org/10.1523/jneurosci.1202-06.2006
Oesch NW, Kothmann WW, Diamond JS (2011) Illuminating synapses and circuitry in the retina. Curr Opin Neurobiol 21(2):238–244. https://doi.org/10.1016/j.conb.2011.01.008
Perez Ortiz JM, Swerdlow RH (2019) Mitochondrial dysfunction in Alzheimer’s disease: role in pathogenesis and novel therapeutic opportunities. Br J Pharmacol 176(18):3489–3507. https://doi.org/10.1111/bph.14585
Pow DV, Crook DK (1996) Direct immunocytochemical evidence for the transfer of glutamine from glial cells to neurons: use of specific antibodies directed against the d-stereoisomers of glutamate and glutamine. Neuroscience 70(1):295–302. https://doi.org/10.1016/0306-4522(95)00363-n
Rauen T, Rothstein JD, Wässle H (1996) Differential expression of three glutamate transporter subtypes in the rat retina. Cell Tissue Res 286(3):325–336. https://doi.org/10.1007/s004410050702
Rauen T, Taylor WR, Kuhlbrodt K, Wiessner M (1998) High-affinity glutamate transporters in the rat retina: a major role of the glial glutamate transporter GLAST-1 in transmitter clearance. Cell Tissue Res 291(1):19–31. https://doi.org/10.1007/s004410050976
Reichenbach A, Reichelt W (1986) Postnatal development of radial glial (Müller) cells of the rabbit retina. Neurosci Lett 71(2):125–130. https://doi.org/10.1016/0304-3940(86)90545-8
Riepe RE, Norenberg MD (1978) Glutamine synthetase in the developing rat retina: an immunohistochemical study. Exp Eye Res 27(4):435–444. https://doi.org/10.1016/0014-4835(78)90022-2
Riepe RE, Norenburg MD (1977) Müller cell localisation of glutamine synthetase in rat retina. Nature 268(5621):654–655. https://doi.org/10.1038/268654a0
Sadleir KR, Eimer WA, Cole SL, Vassar R (2015) Aβ reduction in BACE1 heterozygous null 5XFAD mice is associated with transgenic APP level. Mol Neurodegener 10:1. https://doi.org/10.1186/1750-1326-10-1
Sadleir KR, Popovic J, Vassar R (2018) ER stress is not elevated in the 5XFAD mouse model of Alzheimer’s disease. J Biol Chem 293(48):18434–18443. https://doi.org/10.1074/jbc.RA118.005769
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9(7):676–682. https://doi.org/10.1038/nmeth.2019
Snyder PJ, Alber J, Alt C, Bain LJ, Bouma BE, Bouwman FH, Snyder HM (2021) Retinal imaging in Alzheimer’s and neurodegenerative diseases. Alzheimers Dement 17(1):103–111. https://doi.org/10.1002/alz.12179
Steele ML, Robinson SR (2012) Reactive astrocytes give neurons less support: implications for Alzheimer’s disease. Neurobiol Aging 33(2):423.e421–413. https://doi.org/10.1016/j.neurobiolaging.2010.09.018
Toft-Kehler AK, Skytt DM, Kolko M (2018) A perspective on the Müller cell-neuron metabolic partnership in the inner retina. Mol Neurobiol 55(6):5353–5361. https://doi.org/10.1007/s12035-017-0760-7
Vardimon L, Ben-Dror I, Havazelet N, Fox LE (1993) Molecular control of glutamine synthetase expression in the developing retina tissue. Dev Dyn 196(4):276–282. https://doi.org/10.1002/aja.1001960410
Vohra R, Aldana BI, Bulli G, Skytt DM, Waagepetersen H, Bergersen LH, Kolko M (2019) Lactate-mediated protection of retinal ganglion cells. J Mol Biol 431(9):1878–1888. https://doi.org/10.1016/j.jmb.2019.03.005
Walton HS, Dodd PR (2007) Glutamate-glutamine cycling in Alzheimer’s disease. Neurochem Int 50(7–8):1052–1066. https://doi.org/10.1016/j.neuint.2006.10.007
Wang R, Reddy PH (2017) Role of glutamate and NMDA receptors in Alzheimer’s disease. J Alzheimers Dis 57(4):1041–1048. https://doi.org/10.3233/jad-160763
White RD, Neal MJ (1976) The uptake of L-glutamate by the retina. Brain Res 111(1):79–93. https://doi.org/10.1016/0006-8993(76)91050-7
Williams PA, Thirgood RA, Oliphant H, Frizzati A, Littlewood E, Votruba M, Morgan JE (2013) Retinal ganglion cell dendritic degeneration in a mouse model of Alzheimer’s disease. Neurobiol Aging 34(7):1799–1806. https://doi.org/10.1016/j.neurobiolaging.2013.01.006
Xiao NA, Zhang J, Zhou M, Wei Z, Wu XL, Dai XM, Chen XC (2015) Reduction of glucose metabolism in olfactory bulb is an earlier Alzheimer’s disease-related biomarker in 5XFAD mice. Chin Med J (Engl) 128(16):2220–2227. https://doi.org/10.4103/0366-6999.162507
Yin F, Sancheti H, Patil I, Cadenas E (2016) Energy metabolism and inflammation in brain aging and Alzheimer’s disease. Free Radic Biol Med 100:108–122. https://doi.org/10.1016/j.freeradbiomed.2016.04.200
Yu DY, Cringle SJ, Balaratnasingam C, Morgan WH, Yu PK, Su EN (2013) Retinal ganglion cells: energetics, compartmentation, axonal transport, cytoskeletons and vulnerability. Prog Retin Eye Res 36:217–246. https://doi.org/10.1016/j.preteyeres.2013.07.001
Yu Y, Herman P, Rothman DL, Agarwal D, Hyder F (2018) Evaluating the gray and white matter energy budgets of human brain function. J Cereb Blood Flow Metab 38(8):1339–1353. https://doi.org/10.1177/0271678x17708691
Acknowledgements
The authors thank Laboratory Technician Charlotte Taul Brændstrup for skillful assistance in this study.
Funding
The work was supported by the BRIDGE—Translational Excellence Programme funded by the Novo Nordisk Foundation (Grant agreement no. NNF18SA0034956), Hørslev-Fonden (203866-MAS/mbs1), and Beckett Fonden (19-2-4285).
Author information
Authors and Affiliations
Contributions
MK and BIA developed the concept and hypothesis for the study. Material preparation, data collection and analysis were equally performed by ALMT and BS-M. EWW, JVA and ZAM provided support with the maintenance of mouse colonies and sample preparation. JH supervised the immunohistochemistry analysis of retina whole-mounts and assisted with image acquisition. MK, BIA, JVA, RV, and KKF critically revised the work. The first draft of the manuscript was written by ALMT and BS-M and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Tams, A.L.M., Sanz-Morello, B., Westi, E.W. et al. Decreased Glucose Metabolism and Glutamine Synthesis in the Retina of a Transgenic Mouse Model of Alzheimer’s Disease. Cell Mol Neurobiol 42, 291–303 (2022). https://doi.org/10.1007/s10571-021-01126-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-021-01126-y