Abstract
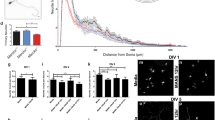
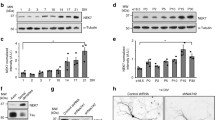
The establishment of polarity is an essential process in early neuronal development. Cdc42, a GTPase of the Rho family, is a key regulator of cytoskeletal dynamics and neuronal polarity. However, the mechanisms underlying the action of cdc42 in regulating axonogenesis have not been elucidated. Here, we expressed wild-type cdc42, a constitutively active cdc42 mutant (cdc42F28L) and a dominant negative cdc42 mutant (cdc42N17), respectively, in the primary hippocampal neurons to alter the activity of cdc42. We found that cdc42 activities were paralleled with the capacities to promote axonogenesis in the cultured neurons. Cdc42 also enhanced microtubule stability in the cultured neurons. Pharmacologically stabilizing microtubules significantly abrogated the defective axonogenesis induced by cdc42 inhibition. Moreover, cdc42 promoted the dephosphorylation of collapsing response mediator protein-2 (CRMP-2) at Thr514 by increasing GSK-3β phosphorylation at Ser9 in the cultured neurons. These findings suggest that cdc42 may facilitate axonogenesis by promoting microtubule stabilization in rat primary hippocampal neurons.







Similar content being viewed by others
Data Availability
The raw data supporting the conclusions of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
Abbreviations
- CRMP-2:
-
Collapsing response mediator protein-2
- cdc42wt:
-
Wild-type cdc42
- mAb:
-
Monoclonal antibodies
- pAb:
-
Polyclonal antibodies
- SD:
-
Sprague–Dawley
- PVDF:
-
Polyvinylidene difluoride
- ECL:
-
Enhanced chemiluminescence
- A/T:
-
Acetylated to tyrosinated α-tubulin
- RGCs:
-
Xenopus retinal ganglion cells
- GSK-3β:
-
Glycogen synthase kinase-3β
- aPKC:
-
Atypical protein kinase C
- NA:
-
No axon
- SA:
-
A single axon
- MA:
-
Multiple axons
References
Arimura N, Kaibuchi K (2007) Neuronal polarity: from extracellular signals to intracellular mechanisms. Nat Rev Neurosci 8(3):194–205. https://doi.org/10.1038/nrn2056
Barnes AP, Polleux F (2009) Establishment of axon-dendrite polarity in developing neurons. Annu Rev Neurosci 32:347–381. https://doi.org/10.1146/annurev.neuro.31.060407.125536
Cau J, Hall A (2005) Cdc42 controls the polarity of the actin and microtubule cytoskeletons through two distinct signal transduction pathways. J Cell Sci 118(Pt 12):2579–2587. https://doi.org/10.1242/jcs.02385
Conde C, Caceres A (2009) Microtubule assembly, organization and dynamics in axons and dendrites. Nat Rev Neurosci 10(5):319–332. https://doi.org/10.1038/nrn2631
Craig AM, Banker G (1994) Neuronal polarity. Annu Rev Neurosci 17:267–310. https://doi.org/10.1146/annurev.ne.17.030194.001411
Dotti CG, Sullivan CA, Banker GA (1988) The establishment of polarity by hippocampal neurons in culture. J Neurosci 8(4):1454–1468. https://doi.org/10.1523/jneurosci.08-04-01454.1988
Eickholt BJ, Walsh FS, Doherty P (2002) An inactive pool of GSK-3 at the leading edge of growth cones is implicated in Semaphorin 3A signaling. J Cell Biol 157(2):211–217. https://doi.org/10.1083/jcb.200201098
Etienne-Manneville S, Hall A (2003) Cdc42 regulates GSK-3beta and adenomatous polyposis coli to control cell polarity. Nature 421(6924):753–756. https://doi.org/10.1038/nature01423
Fukata Y, Itoh TJ, Kimura T, Menager C, Nishimura T, Shiromizu T, Watanabe H, Inagaki N, Iwamatsu A, Hotani H, Kaibuchi K (2002) CRMP-2 binds to tubulin heterodimers to promote microtubule assembly. Nat Cell Biol 4(8):583–591. https://doi.org/10.1038/ncb825
Garvalov BK, Flynn KC, Neukirchen D, Meyn L, Teusch N, Wu X, Brakebusch C, Bamburg JR, Bradke F (2007) Cdc42 regulates cofilin during the establishment of neuronal polarity. J Neurosci 27(48):13117–13129. https://doi.org/10.1523/jneurosci.3322-07.2007
Goldstein B, Macara IG (2007) The PAR proteins: fundamental players in animal cell polarization. Dev Cell 13(5):609–622. https://doi.org/10.1016/j.devcel.2007.10.007
Gomis-Rüth S, Wierenga CJ, Bradke F (2008) Plasticity of polarization: changing dendrites into axons in neurons integrated in neuronal circuits. Curr Biol 18(13):992–1000. https://doi.org/10.1016/j.cub.2008.06.026
Gonzalez-Billault C, Muñoz-Llancao P, Henriquez DR, Wojnacki J, Conde C, Caceres A (2012) The role of small GTPases in neuronal morphogenesis and polarity. Cytoskeleton (Hoboken) 69(7):464–485. https://doi.org/10.1002/cm.21034
Hall A, Lalli G (2010) Rho and Ras GTPases in axon growth, guidance, and branching. Cold Spring Harb Perspect Biol 2(2):a001818. https://doi.org/10.1101/cshperspect.a001818
Hirai S, Banba Y, Satake T, Ohno S (2011) Axon formation in neocortical neurons depends on stage-specific regulation of microtubule stability by the dual leucine zipper kinase-c-Jun N-terminal kinase pathway. J Neurosci 31(17):6468–6480. https://doi.org/10.1523/jneurosci.5038-10.2011
Hu J, Huang HZ, Wang X, Xie AJ, Wang X, Liu D, Wang JZ, Zhu LQ (2015) Activation of glycogen synthase kinase-3 mediates the olfactory deficit-induced hippocampal impairments. Mol Neurobiol 52(3):1601–1617. https://doi.org/10.1007/s12035-014-8953-9
Inagaki N, Chihara K, Arimura N, Ménager C, Kawano Y, Matsuo N, Nishimura T, Amano M, Kaibuchi K (2001) CRMP-2 induces axons in cultured hippocampal neurons. Nat Neurosci 4(8):781–782. https://doi.org/10.1038/90476
Jin Z, Strittmatter SM (1997) Rac1 mediates collapsin-1-induced growth cone collapse. J Neurosci 17(16):6256–6263
Kaufmann N, Wills ZP, Van Vactor D (1998) Drosophila Rac1 controls motor axon guidance. Development 125(3):453–461
Lei WL, Xing SG, Deng CY, Ju XC, Jiang XY, Luo ZG (2012) Laminin/β1 integrin signal triggers axon formation by promoting microtubule assembly and stabilization. Cell Res 22(6):954–972. https://doi.org/10.1038/cr.2012.40
Liu J, Pan C, Guo L, Wu M, Guo J, Peng S, Wu Q, Zuo Q (2016) A new mechanism of trastuzumab resistance in gastric cancer: MACC1 promotes the Warburg effect via activation of the PI3K/AKT signaling pathway. J Hematol Oncol 9(1):76. https://doi.org/10.1186/s13045-016-0302-1
Namekata K, Harada C, Guo X, Kimura A, Kittaka D, Watanabe H, Harada T (2012) Dock3 stimulates axonal outgrowth via GSK-3beta-mediated microtubule assembly. J Neurosci 32(1):264–274. https://doi.org/10.1523/jneurosci.4884-11.2012
Ruchhoeft ML, Ohnuma S, Mcneill L, Holt CE, Harris WA (1999) The neuronal architecture of Xenopus retinal ganglion cells is sculpted by rho-family GTPases in vivo. J Neurosci 19(19):8454–8463
Schelski M, Bradke F (2017) Neuronal polarization: from spatiotemporal signaling to cytoskeletal dynamics. Mol Cell Neurosci 84:11–28. https://doi.org/10.1016/j.mcn.2017.03.008
Schwamborn JC, Puschel AW (2004) The sequential activity of the GTPases Rap1B and Cdc42 determines neuronal polarity. Nat Neurosci 7(9):923–929. https://doi.org/10.1038/nn1295
Sengottuvel V, Leibinger M, Pfreimer M, Andreadaki A, Fischer D (2011) Taxol facilitates axon regeneration in the mature CNS. J Neurosci 31(7):2688–2699. https://doi.org/10.1523/jneurosci.4885-10.2011
Sinha S, Yang W (2008) Cellular signaling for activation of Rho GTPase Cdc42. Cell Signal 20(11):1927–1934. https://doi.org/10.1016/j.cellsig.2008.05.002
Stankiewicz TR, Linseman DA (2014) Rho family GTPases: key players in neuronal development, neuronal survival, and neurodegeneration. Front Cell Neurosci 8:314. https://doi.org/10.3389/fncel.2014.00314
Tu SS, Wu WJ, Yang W, Nolbant P, Hahn K, Cerione RA (2002) Antiapoptotic Cdc42 mutants are potent activators of cellular transformation. Biochemistry 41(41):12350–12358. https://doi.org/10.1021/bi026167h
Wang HH, Li Y, Li A, Yan F, Li ZL, Liu ZY, Zhang L, Zhang J, Dong WR, Zhang L (2018) Forskolin induces hyperphosphorylation of tau accompanied by cell cycle reactivation in primary hippocampal neurons. Mol Neurobiol 55(1):696–706. https://doi.org/10.1007/s12035-016-0348-7
Westermann S, Weber K (2003) Post-translational modifications regulate microtubule function. Nat Rev Mol Cell Biol 4(12):938–947. https://doi.org/10.1038/nrm1260
Witte H, Neukirchen D, Bradke F (2008) Microtubule stabilization specifies initial neuronal polarization. J Cell Biol 180(3):619–632. https://doi.org/10.1083/jcb.200707042
Yoshimura T, Kawano Y, Arimura N, Kawabata S, Kikuchi A, Kaibuchi K (2005) GSK-3beta regulates phosphorylation of CRMP-2 and neuronal polarity. Cell 120(1):137–149. https://doi.org/10.1016/j.cell.2004.11.012
Zhang X, Zhu J, Yang GY, Wang QJ, Qian L, Chen YM, Chen F, Tao Y, Hu HS, Wang T, Luo ZG (2007) Dishevelled promotes axon differentiation by regulating atypical protein kinase C. Nat Cell Biol 9(7):743–754. https://doi.org/10.1038/ncb1603
Zhu LQ, Zheng HY, Peng CX, Liu D, Li HL, Wang Q, Wang JZ (2010) Protein phosphatase 2A facilitates axonogenesis by dephosphorylating CRMP2. J Neurosci 30(10):3839–3848. https://doi.org/10.1523/jneurosci.5174-09.2010
Funding
This work is supported in part by grants from the National Natural Science Foundation of China (81572222 and 30900725) and Guangdong Provincial Natural Science Foundation (2014A030313333 and 8151051501000005).
Author information
Authors and Affiliations
Contributions
AL and HZ contributed equally to this work; HW designed research; AL, HZ, YC, FY, ZYL, and ZLL performed research; AL, HZ, WD, LZ, and HW analyzed data; and HW wrote the manuscript. All authors approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical Approval
The experimental protocol was established according to the Guidelines for the Care and Use of Laboratory Animals of the National Institutes of Health and approved by the Animal Care and Use Committee of Southern Medical University.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, A., Zhu, HM., Chen, Y. et al. Cdc42 Facilitates Axonogenesis by Enhancing Microtubule Stabilization in Primary Hippocampal Neurons. Cell Mol Neurobiol 41, 1599–1610 (2021). https://doi.org/10.1007/s10571-021-01051-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-021-01051-0