Abstract
Rabies virus (RABV) is a neurotropic virus exclusively infecting neurons in the central nervous system. RABV encodes five proteins. Among them, the viral glycoprotein (RVG) plays a key role in viral entry into neurons and rabies pathogenesis. It was shown that the nature of the C-terminus of the RABV G protein, which possesses a PDZ-binding motif (PBM), modulates the virulence of the RABV strain. The neuronal protein partners recruited by this PBM may alter host cell function. This study was conducted to investigate the effect of RVG on synaptic function in the hippocampal dentate gyrus (DG) of rat. Two μl (108 T.U./ml) of the lentiviral vector containing RVG gene was injected into the DG of rat hippocampus. After 2 weeks, the rat’s brain was cross-sectioned and RVG-expressing cells were detected by fluorescent microscopy. Hippocampal synaptic activity of the infected rats was then examined by recording the local field potentials from DG after stimulation of the perforant pathway. Short-term synaptic plasticity was also assessed by double pulse stimulation. Expression of RVG in DG increased long-term potentiation population spikes (LTP-PS), whereas no facilitation of LTP-PS was found in neurons expressing δRVG (deleted PBM). Furthermore, RVG and δRVG strengthened paired-pulse facilitation. Heterosynaptic long-term depression (LTD) in the DG was significantly blocked in RVG-expressing group compared to the control group. This blockade was dependent to PBM motif as rats expressing δRVG in the DG-expressed LTD comparable to the RVG group. Our data demonstrate that RVG expression facilitates both short- and long-term synaptic plasticity in the DG indicating that it may involve both pre- and postsynaptic mechanisms to alter synaptic function. Further studies are needed to elucidate the underlying mechanisms.



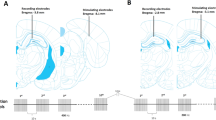
adapted from atlas of Paxinos and Watson showing the position of the stimulating electrodes in the perforant pathway and recording electrodes in dentate gyrus. b A fluorescent image shows location of recording electrodes and transduced cells in the dentate gyrus. c Input and output curves indicate no difference of normalized responses to seven stimulus intensities between groups. d Baseline responses to the 50% maximum of intensity during 30 min are more prominent in rats expressing RVG (n = 10) in dentate gyrus, compared to control (n = 11) and δRVG (n = 10) groups. *p < 0.05 and **p < 0.01 compared to the control group. #p < 0.05 and ##p < 0.01 compared to δRVG group



Similar content being viewed by others
References
Artola A, Singer W (1993) Long-term depression of excitatory synaptic transmission and its relationship to long-term potentiation. Trends Neurosci 16(11):480–487
Azimzadeh Jamalkandi S, Mozhgani S-H, Gholami Pourbadie H, Mirzaie M, Noorbakhsh F, Vaziri B, Gholami A, Ansari-Pour N, Jafari M (2016) Systems biomedicine of rabies delineates the affected signaling pathways. Front Microbiol 7:1688
Baer G, Bellini W, Fishbein D (1990) Rhabdoviruses. In: Fields BN, Knipe DM (eds) Virology, 2nd edn. Raven Press, New York, pp 883–930
Bear MF (1996) A synaptic basis for memory storage in the cerebral cortex. Proc Natl Acad Sci USA 93(24):13453–13459
Bliss TV, Collingridge GL (1993) A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361(6407):31–39
Bouzamondo E, Ladogana A, Tsiang H (1993) Alteration of potassium-evoked 5-HT release from virus-infected rat cortical synaptosomes. NeuroReport 4(5):555–558
Caillet-Saguy C, Maisonneuve P, Delhommel F, Terrien E, Babault N, Lafon M, Cordier F, Wolff N (2015) Strategies to interfere with PDZ-mediated interactions in neurons: what we can learn from the rabies virus. Prog Biophys Mol Biol 119(1):53–59
Castro-Alamancos MA, Connors BW (1997) Distinct forms of short-term plasticity at excitatory synapses of hippocampus and neocortex. Proc Natl Acad Sci USA 94(8):4161–4166
Cherian S, Singh R, Anjaneya SKP (2015) Rabies glycoprotein: a benefit to the virus, us or both? J Vet Sci 1(1):73–81
Christie BR, Abraham WC (1992) NMDA-dependent heterosynaptic long-term depression in the dentate gyrus of anaesthetized rats. Synapse 10(1):1–6
Commins S, O’Mara SM (2000) Interactions between paired-pulse facilitation, low-frequency stimulation, and behavioral stress in the pathway from hippocampal area CA1 to the subiculum: dissociation of baseline synaptic transmission from paired-pulse facilitation and depression of the same pathway. Psychobiology 28(1):1–11
Cummings JA, Mulkey RM, Nicoll RA, Malenka RC (1996) Ca2+ signaling requirements for long-term depression in the hippocampus. Neuron 16(4):825–833
DelRosso L, Hoque R (2013) A case of obstructive sleep apnea, gastroesophageal reflux disease, and chronic hiccups: will CPAP help? J Clin Sleep Med 9(1):92–95
Deng P-Y, Klyachko VA (2011) The diverse functions of short-term plasticity components in synaptic computations. Commun Integr Biol 4(5):543–548
Dhingra V, Li X, Liu Y, Fu ZF (2007) Proteomic profiling reveals that rabies virus infection results in differential expression of host proteins involved in ion homeostasis and synaptic physiology in the central nervous system. J Neurovirol 13(2):107–117
Dumrongphol H, Srikiatkhachorn A, Hemachudha T, Kotchabhakdi N, Govitrapong P (1996) Alteration of muscarinic acetylcholine receptors in rabies viral-infected dog brains. J Neurol Sci 137(1):1–6
Ermine A, Flamand A (1977) Rna syntheses in BHK21 cells infected by rabies virus. Ann Microbiol 128(4):477–488
Farzaneh M, Sayyah M, Eshraghi HR, Panahi N, Delavar HM, Pourbadie HG (2018) Transduction efficacy and retrograde movement of a lentiviral vector pseudotyped by modified rabies glycoprotein throughout the trisynaptic circuit of the rat hippocampus. J Gen Med 20(9):e3046
Fourcaudot E, Gambino F, Casassus G, Poulain B, Humeau Y, Lüthi A (2009) L-type voltage-dependent Ca 2+ channels mediate expression of presynaptic LTP in amygdala. Nat Neurosci 12(9):1093–1095
Fu ZF, Jackson AC (2005) Neuronal dysfunction and death in rabies virus infection. J Neurovirol 11(1):101–106
Gonzalez J, Morales IS, Villarreal DM, Derrick BE (2014) Low-frequency stimulation induces long-term depression and slow onset long-term potentiation at perforant path-dentate gyrus synapses in vivo. J Neurophysiol 111(6):1259–1273
Gosztonyi G (1994) Reproduction of lyssaviruses: ultrastructural composition of lyssavirus and functional aspects of pathogenesis. Curr Top Microbiol Immunol 187:43–68
Gourmelon P, Briet D, Clarençon D, Court L, Tsiang H (1991) Sleep alterations in experimental street rabies virus infection occur in the absence of major EEG abnormalities. Brain Res 554(1–2):159–165
Hayashi Y, Shi S-H, Esteban JA, Piccini A, Poncer J-C, Malinow R (2000) Driving AMPA receptors into synapses by LTP and CaMKII: requirement for GluR1 and PDZ domain interaction. Science 287(5461):2262–2267
Hemachudha T, Tirawatnpong S, Phanthumchinda K (1989) Seizures as the initial manifestation of paralytic rabies. J Neurol Neurosurg Psychiatry 52(6):808–810
Iwata M, Komori S, Unno T, Minamoto N, Ohashi H (1999) Modification of membrane currents in mouse neuroblastoma cells following infection with rabies virus. Br J Pharmacol 126(8):1691–1698
Iwata M, Unno T, Minamoto N, Ohashi H, Komori S (2000) Rabies virus infection prevents the modulation by α2-adrenoceptors, but not muscarinic receptors, of Ca2+ channels in NG108-15 cells. Eur J Pharmacol 404(1–2):79–88
Jackman SL, Regehr WG (2017) The mechanisms and functions of synaptic facilitation. Neuron 94(3):447–464
Jackson AC (2010) Why does the prognosis remain so poor in human rabies? Expert Rev Anti Infect Ther 8(6):623–625
Khan Z, Terrien E, Delhommel F, Lefebvre-Omar C, Bohl D, Vitry S, Bernard C, Ramirez J, Chaffotte A, Ricquier K (2019) Structure-based optimization of a PDZ binding motif within a viral peptide stimulates neurite outgrowth. J Biol Chem 294(37):13755–13768
Ladogana A, Bouzamondo E, Pocchiari M, Tsiang H (1994) Modification of tritiated γ-amino-n-butyric acid transport in rabies virus-infected primary cortical cultures. J Gen Virol 75(Pt 3):623–627
Lafon M (2011) Evasive strategies in rabies virus infection. Adv Virus Res 79:33–53
Lee H-J, Zheng JJ (2010) PDZ domains and their binding partners: structure, specificity, and modification. Cell Commun Signal 8(1):8
Leung LS, Fu X-W (1994) Factors affecting paired-pulse facilitation in hippocampal CA1 neurons in vitro. Brain Res 650(1):75–84
Li X-Q, Sarmento L, Fu ZF (2005) Degeneration of neuronal processes after infection with pathogenic, but not attenuated, rabies viruses. J Virol 79(15):10063–10068
Madore HP, England JM (1977) Rabies virus protein synthesis in infected BHK-21 cells. J Virol 22(1):102–112
Mankouri J, Dallas ML, Hughes ME, Griffin SD, Macdonald A, Peers C, Harris M (2009) Suppression of a pro-apoptotic K+ channel as a mechanism for hepatitis C virus persistence. Proc Natl Acad Sci USA 106(37):15903–15908
Opazo P, Choquet D (2011) A three-step model for the synaptic recruitment of AMPA receptors. Mol Cell Neurosci 46(1):1–8
Prehaud C, Lay S, Dietzschold B, Lafon M (2003) Glycoprotein of nonpathogenic rabies viruses is a key determinant of human cell apoptosis. J Virol 77(19):10537–10547
Pulmanausahakul R, Li J, Schnell MJ, Dietzschold B (2008) The glycoprotein and the matrix protein of rabies virus affect pathogenicity by regulating viral replication and facilitating cell-to-cell spread. J Virol 82(5):2330–2338
Scott CA, Rossiter JP, Andrew RD, Jackson AC (2008) Structural abnormalities in neurons are sufficient to explain the clinical disease and fatal outcome of experimental rabies in yellow fluorescent protein-expressing transgenic mice. J Virol 82(1):513–521
Shi S-H, Hayashi Y, Esteban JA, Malinow R (2001) Subunit-specific rules governing AMPA receptor trafficking to synapses in hippocampal pyramidal neurons. Cell 105(3):331–343
Song Y, Hou J, Qiao B, Li Y, Xu Y, Duan M, Guan Z, Zhang M, Sun L (2013) Street rabies virus causes dendritic injury and F-actin depolymerization in the hippocampus. J Gen Virol 94(2):276–283
Stanton PK, Sejnowski TJ (1989) Associative long-term depression in the hippocampus induced by hebbian covariance. Nature 339(6221):215–218
Stein L, Rech R, Harrison L, Brown C (2010) Immunohistochemical study of rabies virus within the central nervous system of domestic and wildlife species. Vet Pathol 47(4):630–633
Tian Z, Chen Y, Yan W (2019) Clinical features of rabies patients with abnormal sexual behaviors as the presenting manifestations: a case report and literature review. BMC Infect Dis 19(1):679
Tordo N, Kouknetzoff A (1993) The rabies virus genome: an overview. Onderstepoort J Vet Res 60(4):263–269
Tsiang H (1993) Pathophysiology of rabies virus infection of the nervous system. Adv Virus Res 42:375–412
Xie X, Berger TW, Barrionuevo G (1992) Isolated NMDA receptor-mediated synaptic responses express both LTP and LTD. J Neurophysiol 67(4):1009–1013
Yousaf MZ, Qasim M, Zia S, Ashfaq UA, Khan S (2012) Rabies molecular virology, diagnosis, prevention and treatment. Virol J 9(1):1–5
Acknowledgements
This research is supported by Grant No, 868 from Pasteur Institute and is a part of PhD thesis of Soheil Ghassemi.
Author information
Authors and Affiliations
Contributions
HGP and MS involved in study concept and design and study supervision, participated in experimental design and statistical analysis, and obtained funding: SGH, TA, HMD, and SA participated in acquisition of the data. HGP, KA, and MS performed analysis and interpretation of the data. HGP, CP, ML, AG, NN, and MS drafted the manuscript. MS involved in administrative, technical, and material support. All authors had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare.
Ethical Approval
All animal experiments were carried out in accordance with the Review Board and Ethics Committee of Pasteur Institute (Authorization code 93-0201-785, 22 April 2014) and conform to the European Communities Council Directive 2010-63-EU.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ghassemi, S., Asgari, T., Mirzapour-Delavar, H. et al. Lentiviral Expression of Rabies Virus Glycoprotein in the Rat Hippocampus Strengthens Synaptic Plasticity. Cell Mol Neurobiol 42, 1429–1440 (2022). https://doi.org/10.1007/s10571-020-01032-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-020-01032-9