Abstract
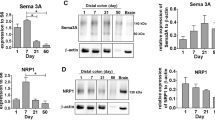
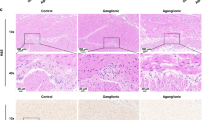
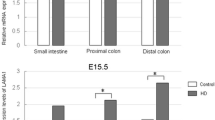
Although approximately 50% of cases have a known genetic defect, the precise pathogenesis of Hirschsprung disease (HSCR) is still unclear. We recently reported that expression of fibronectin (FN), which is involved in the migration, colonization, and differentiation of enteric neural crest cells (ENCCs), is increased in aganglionic colonic segments obtained from patients. We hypothesized that abnormally high levels of FN might play a role in the etiology of HSCR. Here, to test this hypothesis, we investigated aganglionic, transitional, and ganglionic colon segments from 63 children with HSCR and distal colon from thirty healthy Wistar rats at embryonic day 20, in addition to in vitro studies with PC12 Adh neural crest cells. We measured the protein and mRNA expression levels of FN, together with a panel of excitatory (VGLUT1, GluA1, GluN1, PSD-95, and NL-1) and inhibitory (GAD67, GABA AR-α1, NL-2, and SLC32) synaptic markers. Expression of all these synaptic markers was significantly decreased in aganglionic colon, compared to ganglionic colon, whereas expression of FN was significantly increased. In a neural crest cell line, PC12 Adh, knockdown of FN with small-interfering RNA increased the expression of synaptic markers. Co-culture of colons from embryonic day 20 rats with RGD recombinant protein, which contains the RGD motif of FN, reduced the expression of excitatory and inhibitory synaptic markers. These results are consistent with the idea that the etiology of HSCR involves aberrant overexpression of FN, which may impair synaptic function and enteric nervous system development, leading to motor dysfunction of intestinal muscles.




Similar content being viewed by others
References
Akbareian SE, Nagy N, Steiger CE et al (2013) Enteric neural crest-derived cells promote their migration by modifying their microenvironment through tenascin-C production. Dev Biol 382:446–456
Allan IJ, Newgreen DF (2010) The origin and differentiation of enteric neurons of the intestine of the fowl embryo. Dev Dyn 157:137–154
Anderson RB, Newgreen DF, Young HM (2006) Neural crest and the development of the enteric nervous system. Adv Exp Med Biol 589:181–196. https://doi.org/10.1007/978-0-387-46954-6_11
Auteri M, Zizzo MG, Serio R (2015) GABA and GABA receptors in the gastrointestinal tract: from motility to inflammation. Pharmacol Res 93:11–21. https://doi.org/10.1016/j.phrs.2014.12.001
Breau MA, Dahmani A, Broders-Bondon F et al (2009) Beta1 integrins are required for the invasion of the caecum and proximal hindgut by enteric neural crest cells. Development (Camb Engl) 136:2791–2801. https://doi.org/10.1242/dev.031419
Burns AJ, Nikhil T (2013) Neural stem cell therapies for enteric nervous system disorders. Nat Rev Gastroenterol Hepatol 11:317–328
Chih B, Engelman H, Scheiffele P (2005) Control of excitatory and inhibitory synapse formation by neuroligins. Science (New York NY) 307:1324–1328. https://doi.org/10.1126/science.1107470
Cima I, Corazza N, Dick B et al (2004) Intestinal epithelial cells synthesize glucocorticoids and regulate T cell activation. J Exp Med 200:1635–1646. https://doi.org/10.1084/jem.20031958
Deidda G, Parrini M, Naskar S et al (2015) Reversing excitatory GABAAR signaling restores synaptic plasticity and memory in a mouse model of Down syndrome. Nat Med 21:318–326. https://doi.org/10.1038/nm.3827
Druckenbrod NR, Epstein ML (2009) Age-dependent changes in the gut environment restrict the invasion of the hindgut by enteric neural progenitors. Development (Camb Engl) 136:3195–3203. https://doi.org/10.1242/dev.031302
Furness JB (2012) The enteric nervous system and neurogastroenterology. Nat Rev Gastroenterol Hepatol 9:286–294. https://doi.org/10.1038/nrgastro.2012.32
Hamidi H, Ivaska J (2017) Vascular morphogenesis: an integrin and fibronectin highway. Curr Biol 27:R158–R161. https://doi.org/10.1016/j.cub.2016.12.036
Huang G, Li F, Zhao X et al (2017) Functional and biomimetic materials for engineering of the three-dimensional cell microenvironment. Chem Rev 117:12764–12850. https://doi.org/10.1021/acs.chemrev.7b00094
Humphries MJ, Obara M, Olden K et al (1989) Role of fibronectin in adhesion, migration, and metastasis. Cancer Investig 7:373–393
Hynes RO, Naba A (2012) Overview of the matrisome—an inventory of extracellular matrix constituents and functions. Cold Spring Harb Perspect Biol 4:a004903. https://doi.org/10.1101/cshperspect.a004903
Irie M, Hata Y, Takeuchi M et al (1997) Binding of neuroligins to PSD-95. Science (New York, NY) 277:1511–1515
Kaszaki J, Erces D, Varga G et al (2012) Kynurenines and intestinal neurotransmission: the role of N-methyl-d-aspartate receptors. J Neural Transm (Vienna Austria 1996) 119:211–223. https://doi.org/10.1007/s00702-011-0658-x
Langer JC (2004) Persistent obstructive symptoms after surgery for Hirschsprung’s disease: development of a diagnostic and therapeutic algorithm. J Pediatr Surg 39:1458–1462
Lof Granstrom A, Svenningsson A, Hagel E et al (2016) Maternal risk factors and perinatal characteristics for Hirschsprung disease. Pediatrics. https://doi.org/10.1542/peds.2015-4608
Lu J, Jiao Z, Yu Y et al (2018) Programming for increased expression of hippocampal GAD67 mediated the hypersensitivity of the hypothalamic–pituitary–adrenal axis in male offspring rats with prenatal ethanol exposure. Cell Death Dis 9:659. https://doi.org/10.1038/s41419-018-0663-1
Ma QL, Lim GP, Harris-White ME et al (2006) Antibodies against beta-amyloid reduce Abeta oligomers, glycogen synthase kinase-3beta activation and tau phosphorylation in vivo and in vitro. J Neurosci Res 83:374–384. https://doi.org/10.1002/jnr.20734
Maka M, Stolt CC, Wegner M (2005) Identification of Sox8 as a modifier gene in a mouse model of Hirschsprung disease reveals underlying molecular defect. Dev Biol 277:155–169. https://doi.org/10.1016/j.ydbio.2004.09.014
Martucciello G, Ceccherini I, Lerone M et al (2000) Special basic science review ☆: pathogenesis of Hirschsprung’s disease. J Pediatr Surg 35:1017–1025
Nakamura H, Henderson D, Puri P (2017) A meta-analysis of clinical outcome of intestinal transplantation in patients with total intestinal aganglionosis. Pediatr Surg Int 33:837–841. https://doi.org/10.1007/s00383-017-4107-2
Okello EJ, McDougall GJ, Kumar S et al (2011) In vitro protective effects of colon-available extract of Camellia sinensis (tea) against hydrogen peroxide and beta-amyloid (Abeta((1-42))) induced cytotoxicity in differentiated PC12 cells. Phytomed Int J Phytother Phytopharmacol 18:691–696. https://doi.org/10.1016/j.phymed.2010.11.004
Roberts RR, Bornstein JC, Bergner AJ et al (2008) Disturbances of colonic motility in mouse models of Hirschsprung’s disease. Am J Physiol Gastrointest Liver Physiol 294:G996–G1008. https://doi.org/10.1152/ajpgi.00558.2007
Rodolphe S, Mathilde M, Bergeron KF et al (2015) A collagen VI-dependent pathogenic mechanism for Hirschsprung’s disease. J Clin Investig 125:4483–4496
Ruttenstock E, Puri P (2009) A meta-analysis of clinical outcome in patients with total intestinal aganglionosis. Pediatr Surg Int 25:833–839. https://doi.org/10.1007/s00383-009-2439-2
Seifi M, Swinny JD (2016) Immunolocalization of AMPA receptor subunits within the enteric nervous system of the mouse colon and the effect of their activation on spontaneous colonic contractions. Neurogastroenterol Motil Off J Eur Gastrointest Motil Soc 28:705–720. https://doi.org/10.1111/nmo.12768
Sharkey KA (2015) Emerging roles for enteric glia in gastrointestinal disorders. J Clin Investig 125:918–925
Siracusa R, Impellizzeri D, Cordaro M et al (2017) Anti-inflammatory and neuroprotective effects of Co-UltraPEALut in a mouse model of vascular dementia. Front Neurol 8:233. https://doi.org/10.3389/fneur.2017.00233
Stanzel RD, Lourenssen S, Blennerhassett MG (2008) Inflammation causes expression of NGF in epithelial cells of the rat colon. Exp Neurol 211:203–213. https://doi.org/10.1016/j.expneurol.2008.01.028
Stensrud KJ, Emblem R, Bjornland K (2010) Functional outcome after operation for Hirschsprung disease—transanal vs transabdominal approach. J Pediatr Surg 45:1640–1644. https://doi.org/10.1016/j.jpedsurg.2010.02.065
Tan Z, Kang T, Zhang X et al (2019) Nerve growth factor prevents arsenic-induced toxicity in PC12 cells through the AKT/GSK-3beta/NFAT pathway. 234:4726–4738. https://doi.org/10.1002/jcp.27255
Tomita S (2019) Molecular constituents and localization of the ionotropic GABA receptor complex in vivo. Curr Opin Neurobiol 57:81–86. https://doi.org/10.1016/j.conb.2019.01.017
Tonnies E, Trushina E (2017) Oxidative stress, synaptic dysfunction, and Alzheimer’s disease. J Alzheimer’s Dis 57:1105–1121. https://doi.org/10.3233/jad-161088
Traynelis SF, Wollmuth LP, McBain CJ et al (2010) Glutamate receptor ion channels: structure, regulation, and function. Pharmacol Rev 62:405–496. https://doi.org/10.1124/pr.109.002451
Uesaka T, Nagashimada M, Yonemura S et al (2008) Diminished Ret expression compromises neuronal survival in the colon and causes intestinal aganglionosis in mice. J Clin Investig 118:1890–1898. https://doi.org/10.1172/jci34425
Vladimir GI, Vladimir P (2015) The second brain in autism spectrum disorder: could connexin 43 expressed in enteric glial cells play a role? Front Cell Neurosci 9:242
Wallace AS, Burns AJ (2005) Development of the enteric nervous system, smooth muscle and interstitial cells of Cajal in the human gastrointestinal tract. Cell Tissue Res 319:367–382. https://doi.org/10.1007/s00441-004-1023-2
Young HM, McKeown SJ (2016) Motility: Hirschsprung disease—laying down a suitable path. Nat Rev Gastroenterol Hepatol 13:7–8. https://doi.org/10.1038/nrgastro.2015.213
Young HM, Newgreen D (2001) Enteric neural crest-derived cells: origin, identification, migration, and differentiation. Anat Rec 262:1–15. https://doi.org/10.1002/1097-0185(20010101)262:1%3c1:aid-ar1006%3e3.0.co;2-2
Young HM, Bergner AJ, Anderson RB et al (2004) Dynamics of neural crest-derived cell migration in the embryonic mouse gut ☆. Dev Biol 270:455–473
Zheng Y, Lv X, Wang D et al (2017) Down-regulation of fibronectin and the correlated expression of neuroligin in Hirschsprung disease. 29. https://doi.org/10.1111/nmo.13134
Zhu S, Gouaux E (2017) Structure and symmetry inform gating principles of ionotropic glutamate receptors. Neuropharmacology 112:11–15. https://doi.org/10.1016/j.neuropharm.2016.08.034
Zizzo MG, Mulè F, Serio R (2007) Functional evidence for GABA as modulator of the contractility of the longitudinal muscle in mouse duodenum: Role of GABA and GABA receptors. Neuropharmacology 52:1685–1690
Funding
This work was funded by the National Natural Science Foundation of China (Projects Nos. 81471487, 81701492 and 81270720).
Author information
Authors and Affiliations
Contributions
Ni Gao, conceived project, design, and execution of most experiments, data acquisition and analysis, writing—original draft, review, and editing; Peimin Hou, data acquisition and analysis, writing; Jian Wang and Tingting Zhou, data acquisition and analysis; Dongming Wang and Qiangye Zhang, data acquisition and analysis; Weijing Mu, writing—review and editing; Xiaona Lv and Aiwu Li, conceived project, resources, supervision, funding acquisition, writing—review and editing.
Corresponding authors
Ethics declarations
Conflict of interests
None declared.
Ethical Approval
Our study was approved by the Ethics Committee of Qilu Hospital, Shandong University. All procedures involving animals were conducted in accordance with the ethical standards of the institution.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Gao, N., Hou, P., Wang, J. et al. Increased Fibronectin Impairs the Function of Excitatory/Inhibitory Synapses in Hirschsprung Disease. Cell Mol Neurobiol 40, 617–628 (2020). https://doi.org/10.1007/s10571-019-00759-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-019-00759-4