Abstract
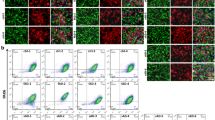
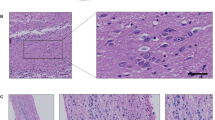
Histopathological hallmarks of dementia have been described postmortem in the brain of patients with Alzheimer’s disease (AD). Tau, a microtubule associated protein, is abnormally arranged in neurofibrillary tangles. In living AD patients, total tau (t-tau) and hyperphosphorylated tau (p-tau) levels are increased in the cerebrospinal fluid obtained by lumbar puncture. Herein, we studied the t-tau and p-tau levels as well as the subcellular distribution of t-tau in olfactory neuronal precursors obtained by exfoliation of the nasal cavity of AD patients and control participants. Data showed that t-tau and p-tau levels were increased in cell homogenates from AD patients. Also, t-tau immunoreactivity was arranged in a punctate pattern in olfactory neuronal precursors derived from an AD participant with 5 years of evolution and in the oldest participants, either control subjects or those with Alzheimer’s disease. Results support that exfoliated neuronal precursors have tau alterations demonstrated in postmortem brain and in the cerebrospinal fluid. This evidence and because the obtainment of olfactory neuronal precursors is a noninvasive procedure, detection of tau alterations shown here might be useful for an early diagnosis of AD-type dementia.








Similar content being viewed by others
References
Arnold SE, Smutzer GS, Trojanowski JQ, Moberg PJ (1998) Cellular and molecular neuropathology of the olfactory epithelium and central olfactory pathways in Alzheimer’s disease and schizophrenia. Ann N Y Acad Sci 855:762–775
Arnold SE, Lee EB, Moberg PJ et al (2010) Olfactory epithelium amyloid-β and paired helical filament-tau pathology in Alzheimer disease. Ann Neurol 67:462–469. https://doi.org/10.1002/ana.21910
Attems J, Jellinger KA (2006) Olfactory tau pathology in Alzheimer disease and mild cognitive impairment. Clin Neuropathol 25:265–271
Attems J, Walker L, Jellinger KA (2014) Olfactory bulb involvement in neurodegenerative diseases. Acta Neuropathol 127:459–475. https://doi.org/10.1007/s00401-014-1261-7
Augustinack JC, Schneider A, Mandelkow E-M, Hyman BT (2002) Specific tau phosphorylation sites correlate with severity of neuronal cytopathology in Alzheimer’s disease. Acta Neuropathol 103:26–35
Benítez-King G, Riquelme A, Ortíz-López L et al (2011) A non-invasive method to isolate the neuronal linage from the nasal epithelium from schizophrenic and bipolar diseases. J Neurosci Methods 201:35–45. https://doi.org/10.1016/j.jneumeth.2011.07.009
Benítez-King G, Valdés-Tovar M, Trueta C et al (2016) The microtubular cytoskeleton of olfactory neurons derived from patients with schizophrenia or with bipolar disorder: implications for biomarker characterization, neuronal physiology and pharmacological screening. Mol Cell Neurosci 73:84–95. https://doi.org/10.1016/j.mcn.2016.01.013
Blennow K, Hampel H (2003) CSF markers for incipient Alzheimer’s disease. Lancet Neurol 2:605–613
Borgmann-Winter KE, Rawson NE, Wang H-Y et al (2009) Human olfactory epithelial cells generated in vitro express diverse neuronal characteristics. Neuroscience 158:642–653. https://doi.org/10.1016/j.neuroscience.2008.09.059
Braak H, Alafuzoff I, Arzberger T et al (2006) Staging of Alzheimer disease-associated neurofibrillary pathology using paraffin sections and immunocytochemistry. Acta Neuropathol 112:389–404. https://doi.org/10.1007/s00401-006-0127-z
Brion JP, Anderton BH, Authelet M et al (2001) Neurofibrillary tangles and tau phosphorylation. Biochem Soc Symp 67:81–88
Buée L, Bussière T, Buée-Scherrer V et al (2000) Tau protein isoforms, phosphorylation and role in neurodegenerative disorders. Brain Res Brain Res Rev 33:95–130
Butler M, Shelanski ML (1986) Microheterogeneity of microtubule-associated tau proteins is due to differences in phosphorylation. J Neurochem 47:1517–1522
Cascella NG, Takaki M, Lin S, Sawa A (2007) Neurodevelopmental involvement in schizophrenia: the olfactory epithelium as an alternative model for research. J Neurochem 102:587–594. https://doi.org/10.1111/j.1471-4159.2007.4628.x
Christen-Zaech S, Kraftsik R, Pillevuit O et al (2003) Early olfactory involvement in Alzheimer’s disease. Can J Neurol Sci 30:20–25
Davis KL, Davidson M, Yang RK et al (1988) CSF somatostatin in Alzheimer’s disease, depressed patients, and control subjects. Biol Psychiatry 24:710–712
Delacourte A (1999) Biochemical and molecular characterization of neurofibrillary degeneration in frontotemporal dementias. Dement Geriatr Cogn Disord 10:75–79. https://doi.org/10.1159/000051218
Duan AR, Jonasson EM, Alberico EO et al (2017) Interactions between tau and different conformations of tubulin: implications for tau function and mechanism. J Mol Biol 429:1424–1438. https://doi.org/10.1016/j.jmb.2017.03.018
Folstein MF, Folstein SE, McHugh PR (1975) “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12:189–198
Fortea J, Carmona-Iragui M, Benejam B et al (2018) Plasma and CSF biomarkers for the diagnosis of Alzheimer’s disease in adults with Down syndrome: a cross-sectional study. Lancet Neurol 17:860–869. https://doi.org/10.1016/S1474-4422(18)30285-0
Goedert M, Spillantini MG, Cairns NJ, Crowther RA (1992) Tau proteins of Alzheimer paired helical filaments: abnormal phosphorylation of all six brain isoforms. Neuron 8:159–168
Greenberg SG, Davies P (1990) A preparation of Alzheimer paired helical filaments that displays distinct tau proteins by polyacrylamide gel electrophoresis. Proc Natl Acad Sci USA 87:5827–5831
Guillozet AL, Weintraub S, Mash DC, Mesulam MM (2003) Neurofibrillary tangles, amyloid, and memory in aging and mild cognitive impairment. Arch Neurol 60:729. https://doi.org/10.1001/archneur.60.5.729
Herculano-Houzel S (2014) The glia/neuron ratio: how it varies uniformly across brain structures and species and what that means for brain physiology and evolution. Glia 62:1377–1391. https://doi.org/10.1002/glia.22683
Hock C, Golombowski S, Müller-Spahn F et al (1998) Histological markers in nasal mucosa of patients with Alzheimer’s disease. Eur Neurol 40:31–36. https://doi.org/10.1159/000007953
Hu WT, Watts K, Grossman M et al (2013) Reduced CSF p-Tau181 to Tau ratio is a biomarker for FTLD-TDP. Neurology 81:1945–1952. https://doi.org/10.1212/01.wnl.0000436625.63650.27
Iqbal K, Grundke-Iqbal I, Zaidi T et al (1986) Defective brain microtubule assembly in Alzheimer’s disease. Lancet (London, England) 2:421–426
Iqbal K, Alonso AD, Chen S et al (2005) Tau pathology in Alzheimer disease and other tauopathies. Biochim Biophys Acta 1739:198–210. https://doi.org/10.1016/j.bbadis.2004.09.008
Ishiguro K, Omori A, Sato K et al (1991) A serine/threonine proline kinase activity is included in the tau protein kinase fraction forming a paired helical filament epitope. Neurosci Lett 128:195–198
Khatoon S, Grundke-Iqbal I, Iqbal K (1992) Brain levels of microtubule-associated protein tau are elevated in Alzheimer’s disease: a radioimmuno-slot-blot assay for nanograms of the protein. J Neurochem 59:750–753
Kishikawa M, Iseki M, Sakae M et al (1994) Early diagnosis of Alzheimer’s? Nature 369:365–366. https://doi.org/10.1038/369365a0
Kovacs T (2004) Mechanisms of olfactory dysfunction in aging and neurodegenerative disorders. Ageing Res Rev 3:215–232. https://doi.org/10.1016/j.arr.2003.10.003
Kovács T, Cairns NJ, Lantos PL (1999) beta-amyloid deposition and neurofibrillary tangle formation in the olfactory bulb in ageing and Alzheimer’s disease. Neuropathol Appl Neurobiol 25:481–491
Laemmli UK (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680–685
Lavoie J, Sawa A, Ishizuka K (2017) Application of olfactory tissue and its neural progenitors to schizophrenia and psychiatric research. Curr Opin Psychiatry 30:176–183. https://doi.org/10.1097/YCO.0000000000000327
Lee JH, Goedert M, Hill WD et al (1993) Tau proteins are abnormally expressed in olfactory epithelium of Alzheimer patients and developmentally regulated in human fetal spinal cord. Exp Neurol 121:93–105. https://doi.org/10.1006/exnr.1993.1074
Leszek J, Małyszczak K, Janicka B et al (2003) Total tau in cerebrospinal fluid differentiates Alzheimer’s disease from vascular dementia. Med Sci Monit 9:CR484–CR488
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Matigian N, Abrahamsen G, Sutharsan R et al (2010) Disease-specific, neurosphere-derived cells as models for brain disorders. Dis Model Mech 3:785–798. https://doi.org/10.1242/dmm.005447
Muñoz-Estrada J, Benítez-King G, Berlanga C, Meza I (2015) Altered subcellular distribution of the 75-kDa DISC1 isoform, cAMP accumulation, and decreased neuronal migration in schizophrenia and bipolar disorder: implications for neurodevelopment. CNS Neurosci Ther 21:446–453. https://doi.org/10.1111/cns.12377
Muñoz-Estrada J, Lora-Castellanos A, Meza I et al (2018) Primary cilia formation is diminished in schizophrenia and bipolar disorder: a possible marker for these psychiatric diseases. Schizophr Res 195:412–420. https://doi.org/10.1016/j.schres.2017.08.055
Murphy C, Solomon ES, Haase L et al (2009) Olfaction in aging and Alzheimer’s disease: event-related potentials to a cross-modal odor-recognition memory task discriminate ApoE epsilon4 + and ApoE epsilon 4- individuals. Ann N Y Acad Sci 1170:647–657. https://doi.org/10.1111/j.1749-6632.2009.04486.x
O’Farrell PH (1975) High resolution two-dimensional electrophoresis of proteins. J Biol Chem 250:4007–4021
Papasozomenos SC (1989) Tau protein immunoreactivity in dementia of the Alzheimer type. I. Morphology, evolution, distribution, and pathogenetic implications. Lab Invest 60:123–137
Polanco JC, Li C, Bodea L-G et al (2018) Amyloid-β and tau complexity—towards improved biomarkers and targeted therapies. Nat Rev Neurol 14:22–39. https://doi.org/10.1038/nrneurol.2017.162
Prince MJ, Wu F, Guo Y et al (2015) The burden of disease in older people and implications for health policy and practice. Lancet 385:549–62. https://doi.org/10.1016/S0140-6736(14)61347-7
Qian J, Hyman BT, Betensky RA (2017) Neurofibrillary tangle stage and the rate of progression of Alzheimer symptoms: modeling using an autopsy cohort and application to clinical trial design. JAMA Neurol 74:540–548. https://doi.org/10.1001/jamaneurol.2016.5953
Reyes PF, Deems DA, Suarez MG (1993) Olfactory-related changes in Alzheimer’s disease: a quantitative neuropathologic study. Brain Res Bull 32:1–5
Sawa A, Cascella NG (2009) Peripheral olfactory system for clinical and basic psychiatry: a promising entry point to the mystery of brain mechanism and biomarker identification in schizophrenia. Am J Psychiatry 166:137–139. https://doi.org/10.1176/appi.ajp.2008.08111702
Sergeant N, Delacourte A, Buée L (2005) Tau protein as a differential biomarker of tauopathies. Biochim Biophys Acta 1739:179–197. https://doi.org/10.1016/j.bbadis.2004.06.020
Shah H, Albanese E, Duggan C et al (2016) Research priorities to reduce the global burden of dementia by 2025. Lancet Neurol 15:1285–1294. https://doi.org/10.1016/S1474-4422(16)30235-6
Skoog I, Vanmechelen E, Andreasson LA et al (1995) A population-based study of tau protein and ubiquitin in cerebrospinal fluid in 85-year-olds: relation to severity of dementia and cerebral atrophy, but not to the apolipoprotein E4 allele. Neurodegeneration 4:433–442
Talamo BR, Rudel R, Kosik KS et al (1989) Pathological changes in olfactory neurons in patients with Alzheimer’s disease. Nature 337:736–739. https://doi.org/10.1038/337736a0
Tarawneh R, Holtzman DM (2012) The clinical problem of symptomatic Alzheimer disease and mild cognitive impairment. Cold Spring Harb Perspect Med 2:a006148–a006148. https://doi.org/10.1101/cshperspect.a006148
Tatsumi S, Uchihara T, Aiba I et al (2014) Ultrastructural differences in pretangles between Alzheimer disease and corticobasal degeneration revealed by comparative light and electron microscopy. Acta Neuropathol Commun 2:161. https://doi.org/10.1186/s40478-014-0161-3
Towbin H, Staehelin T, Gordon J (1992) Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. 1979. Biotechnology 24:145–149
Vandermeeren M, Mercken M, Vanmechelen E et al (1993) Detection of tau proteins in normal and Alzheimer’s disease cerebrospinal fluid with a sensitive sandwich enzyme-linked immunosorbent assay. J Neurochem 61:1828–1834
Wilson RS, Schneider JA, Arnold SE et al (2007) Olfactory identification and incidence of mild cognitive impairment in older age. Arch Gen Psychiatry 64:802. https://doi.org/10.1001/archpsyc.64.7.802
Zhang X, Klueber KM, Guo Z et al (2004) Adult human olfactory neural progenitors cultured in defined medium. Exp Neurol 186:112–123. https://doi.org/10.1016/j.expneurol.2003.10.022
Funding
This study was supported by the Consejo Nacional de Ciencia y Tecnología (CONACyT), México, Grant Number 178075 to Gloria Benítez-King.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they do not have conflicts of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional research committee (Comité de Ética en Investigación del Instituto Nacional de Psiquiatría Ramón de la Fuente Muñiz, IC122037.0) and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Riquelme, A., Valdés-Tovar, M., Ugalde, O. et al. Potential Use of Exfoliated and Cultured Olfactory Neuronal Precursors for In Vivo Alzheimer’s Disease Diagnosis: A Pilot Study. Cell Mol Neurobiol 40, 87–98 (2020). https://doi.org/10.1007/s10571-019-00718-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-019-00718-z