Abstract
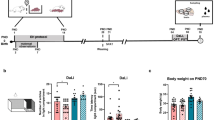
Early-life experiences have been linked to individual’s epigenetic status and social behaviour. Therefore, the present study aims to test whether the presence of mother suppress the early-life stressful social experience (SSE)-induced effect on social behaviour of adolescent and adult rats, and associated epigenetic changes. To test this, experimental groups [maternally separated pups (MSP)/pups with their mother (M+P)] were allowed to experience the presence of a stranger (ST), and then their social behaviour was compared with the maternal separated (MS) and control (Con) group. We observed that MS, MSP-ST group showed less social interaction with the unknown conspecifics than known conspecifics compared to other groups. Subsequently, we found that SSE elevated the level of DNA methyltransferases (Dnmt3a), ten-eleven translocation (Tet3), methyl-CpG-binding protein-2 (MeCP2) and Repressor Element-1 Silencing Transcription Factor (REST) in amygdala of adolescent and adult MS, MSP-ST groups compared to other groups. As expected, SSE altered the histone (H3) lysine (K14/K9) acetylation (ac) and H3K4/K9 methylation (me2/me3). SSE decreased the level of H3K14ac and H3K9ac in adolescents and then increased in adults. Interestingly, H3K4me2/me3 levels were elevated in adolescent and adults. Whereas H3K9me2/me3 shows contrasting pattern in adolescent, but H3K9me2/me3 levels were increased in adults. In addition, the expression of brain-derived neurotrophic factor (BDNF) was reduced in MS, MSP-ST groups’ adolescent and adult rats. Observed correlation between epigenetic changes and social behaviour possibly contributed by early-life SSE in the absence of mother, but mother’s presence suppresses the effect of early-life SSE.












Similar content being viewed by others
References
Anier K, Malinovskaja K, Pruus K, Aonurm-Helm A, Zharkovsky A, Kalda A (2014) Maternal separation is associated with DNA methylation and behavioural changes in adult rats. Eur Neuropsychopharmacol 24(3): 459–68. https://doi.org/10.1016/j.euroneuro.2013.07.012
Berger SL (2007) The complex language of chromatin regulation during transcription. Nature 447:407–412. https://doi.org/10.1038/nature05915
Bian Y, Yang L, Wang Z, Wang Q, Zeng L, Xu G (2015) Repeated three-hour maternal separation induces depression-like behaviour and affects the expression of hippocampal plasticity-related proteins in C57BL/6N mice. Neural Plast. https://doi.org/10.1155/2015/627837
Boersma GJ, Lee RS, Cordner ZA, Ewald ER, Purcell RH, Moghadam AA, Tamashiro KL (2014) Prenatal stress decreases Bdnf expression and increases methylation of Bdnf exon IV in rats. Epigenetics 9(3):437–447. https://doi.org/10.4161/epi.27558
Bondar NP, Lepeshko AA, Reshetnikov VV (2018) Effects of early-life stress on social and anxiety-like behaviors in adult mice: sex-specific effects. Behav Neurol. https://doi.org/10.1155/2018/1538931
Bose R, Spulber S, Kilian P, Heldring N, Lönnerberg P, Johnsson A, Conti M, Hermanson O, Ceccatelli S (2015) Tet3 mediates stable glucocorticoid-induced alterations in DNA methylation and DNMT3a/Dkk1 expression in neural progenitors. Cell Death Dis 6:e1793. https://doi.org/10.1038/cddis.2015.159
Bruce WA, Donaldson IJ, Wood IC, Yerbury SA, Sadowski MI, Chapman M, Göttgens B, Buckley NJ (2004) Genome-wide analysis of repressor element 1 silencing transcription factor/ neuron-restrictive silencing factor (REST/NRSF) target genes. Proc Natl Acad Sci U S A 2004(101):10458–10463. https://doi.org/10.1073/pnas.0401827101
Chidlow G, Wood JP, Casson RJ (2014) Expression of inducible heat shock proteins Hsp27 and Hsp70 in the visual pathway of rats subjected to various models of retinal ganglion cell injury. PLoS ONE 9(12): e114838. https://doi.org/10.1371/journal.pone.0114838
Colquitt BM, Allen WE, Barnea G, Lomvardas (2013) Alteration of genic 5-hydroxymethylcytosine patterning in olfactory neurons correlates with changes in gene expression and cell identity. Proc Natl Acad Sci U S A 110:14682–14687. https://doi.org/10.1073/pnas.1302759110
Covington HE 3rd, Maze I, LaPlant QC, Vlalou VF, Ohnishi YN, Berton O et al (2009) Antidepressant actions of histone deacetylases inhibitors. J Neurosci 29:11451–11460. https://doi.org/10.1523/JNEUROSCI.1758-09.2009
Covington HE 3rd, Maze I, Sun H, Bomze HM, DeMaio KD, Wu EY et al (2011) A role for repressive histone methylation in cocaine-induced vulnerability to stress. Neuron 71: 656–670. https://doi.org/10.1016/j.neuron.2011.06.007
DʹAmato FR, Cabib S (1987) Chronic exposure to a novel odor increases pups’ vocalizations, maternal care, and alters dopaminergic functioning in developing mice. Behav Neural Biol 48:197–205
Doherty TS, Forster A, Roth TL (2016) Global and gene-specific DNA methylation alterations in the adolescent amygdala and hippocampus in an animal model of caregiver maltreatment. Behav Brain Res 298(0 0):55–61. https://doi.org/10.1016/j.bbr.2015.05.028
Elliot E, Manashirov S, Zwang R, Gil S, Tsoory M, Shemesh Y, Chen A (2016) Dnmt3a in the medial prefrontal cortex regulated anxiety-like behaviour in adult mice. J Neurosci 36(3):730–740. https://doi.org/10.1523/JNEUROSCI.0971-15.2016
Ershov NI, Nondar NP, Lepeshko AA, Reshetnikov VV, Ryabushkina JA, Merkulova TI (2018) Consequences of early life stress on genomic landscape of H3K4me3 in prefrontal cortex of adult mice. BMC Genom 19(Suppl 3):93. https://doi.org/10.1186/s12864-018-4479-2
Feifel AJ, Shair HN, Schmauss C (2017) Lasting effects of early life stress in mice: interaction of maternal environment and infant genes. Genes Brain Behav 16(8):768–780. https://doi.org/10.1111/gbb.12395
Ferland CL, Schrader LA (2011) Regulation of histone acetylation in the hippocampus of chronically stressed rats: a potential role of sirtuins. Neuroscience 174:104–114
File SE, Hyde JR (1978) Can social interaction be used to measure anxiety? Br J Pharmacol 62:19–24
Gere MK, Villabø MA, Torgersen S, Kendall PC (2012) Overprotective parenting and child anxiety: the role of co-occurring child behaviour problems. J Anxiety Disord 26(6):642–649. https://doi.org/10.1016/j.janxdis.2012.04.003
Gupta S, Kim SY, Artis S, Molfese DL, Schumacher A, Sweatt JD, Paylor RE, Lubin FD (2011) Histone methylation regulates memory formation. J Neurosci 30(10):3589–3599. https://doi.org/10.1523/JNEUROSCI.3732-09.2010
Guy J, Cheval H, Selfridge J, Bird A (2010) The role of MeCP2 in the brain. Annu Rev Cell Dev Biol 27:631–652. https://doi.org/10.1146/annurev-cellbio-092910-154121
Hahn MA, Qui R, Wu X, Li AX, Zhang H, Wang J, Jui J, Jin SG, Jiang Y, Pfeifer GP, Lu Q (2013) Dynamics of 5-hydroxymethylcytosine and chromatin marks in mammalian neurogenesis. Cell Rep 3:291–300. https://doi.org/10.1016/j.celrep.2013.01.011
Holmes A, Le Guisquet AM, Vogel E, Millstein RA, Leman S, Belzung C (2005) Early life genetic, epigenetic and environmental factors shaping emotionality in rodents. Neurosci Biobehav Rev 29:1335–1346. https://doi.org/10.1016/j.neubiorev.2005.04.012
Hunter RG, McCarthy KJ, Milne TA, Pfaff DW, McEwen BS (2009) Regulation of hippocampal H3 histone methylation by acute and chronic stress. Proc Natl Acad Sci U S A 106(49):20912–20917. https://doi.org/10.1073/pnas.0911143106
Hunter RG, Murakami G, Dewell S, Seligsohn MA, Baker ME, Datson NA, McEwen BS, Pfaff DW (2012) Acute stress and hippocampal histone H3 lysine 9 trimethylation, a retrotransposon silencing response. Proc Natl Acad Sci U S A 109(43):17657–17662. https://doi.org/10.1073/pnas.1215810109
Kalin NH, Takahashi LK, Chen FL (1994) Restraint stress increases corticotropin-releasing hormone mRNA content in the amygdala and paraventricular nucleus. Brain Res 656(1):182–186
Kao GS, Cheng LY, Chen LH, Tzeng WY, Cherng CG, Su CC, Wang CY, Yu L (2012) Neonatal isolation decreases cued fear conditioning and frontal cortical histone 3 lysine 9 methylation in adult female rats. Eur J Pharmacol 697:65–72. https://doi.org/10.1016/j.ejphar.2012.09.040
Lachner M, Jenuwein T (2002) The many faces of histone lysine methylation. Curr Opin Cell Biol 14(3):286–298.
Leite IS, Castelhano AS, Cysneiros RM (2016) Effect of diazepam on sociability of rats submitted to neonatal seizures. Data Brief 7:686–691. https://doi.org/10.1016/j.dib.2016.03.029
Liu D, Qiu HM, Fei HZ, Hu XY, Xia HJ, Wang LJ, Qin LJ, Jiang XH, Zhou QX (2014) Histone acetylation and expression of mono-aminergic transmitters synthetases involved in CUS-induced depressive rats. Exp Biol Med (Maywood) 239(3):330–336. https://doi.org/10.1177/1535370213513987
Luchetti A, Oddi D, Lampis V, Centofante E, Felsani A, Battaglia M, D’Amato FR (2015) Early handling and repeated cross-fostering have opposite effect on mouse emotionally. Front Behav Neurosci 9:93. https://doi.org/10.3389/fnbeh.2015.00093
McGann JC, Oyer JA, Garg S, Yao H, Liu J, Feng X, Liao L, Yates JR 3rd, Mandel G (2014) Polycomb- and REST-associated histone deacetylases are independent pathways toward a mature neuronal phenotype. eLife 3:e04235. https://doi.org/10.7554/eLife.04235
McGowan PO, Suderman M, Sasaki A, Huang TC, Hallett M, Meaney MJ, Szyf M (2011) Broad epigenetic signature of maternal care in the brain of adult rats. PLoS ONE 6:e14739. https://doi.org/10.1371/journal.pone.0014739
McLeod BD, Wood JJ, Weisz JR (2007) Examining the association between parenting and childhood anxiety: a meta-analysis. Clin Psychol Rev 27(2):155–172. https://doi.org/10.1016/j.cpr.2006.09.002
Mitchelmore C, Gede L (2014) Brain derived neurotrophic factor: Epigenetic regulation in psychiatric disorders. Brain Res 1586:162–172. https://doi.org/10.1016/j.brainres.2014.06.037
Moriceau S, Sullivan RM (2006) Maternal presence serves as a switch between learning fear and attraction in infancy. Nat Neurosci 9:1004–1006. https://doi.org/10.1038/nn1733
Moriceau S, Roth TL, Okotoghaide T, Sullivan RM (2004) Corticosterone controls the developmental emergence of fear and amygdala function to predator odors in infant rat pups. Int J Dev Neurosci 22:415–422
Moriceau S, Shionoya K, Jakubs K, Sullivan RM (2009) Early-life stress disrupts attachment learning: the role of amygdala corticosterone locus ceruleus corticotropin releasing hormone and olfactory bulb norepinephrine. J Neurosci 29:15745–15755. https://doi.org/10.1523/JNEUROSCI.4106-09.2009
Morse SJ, Butler AA, Davis RL, Soller IJ, Lubin FD (2015) Environmental enrichment reverses histone methylation changes in the aged hippocampus and restores age-related memory deficits. Biology (Basel) 4(2): 298–313. https://doi.org/10.3390/biology4020298
Murgatroyd C, Spengler D (2014) Polycomb binding precedes early-life stress responsive DNA methylation at the Avp enhancer. PLoS ONE 9(3):e90277. https://doi.org/10.1371/journal.pone.0090277
Murgatroyd C, Patchev AV, Wu Y, Micale V, Bockmuhl Y, Fischer D. Holsboer F, WotjaK CT, Almeida OF, Spengler D (2009) Dynamic DNA methylation programs persistent adverse effects of early-life stress. Nat Neurosci 12:1559–1566. https://doi.org/10.1038/nn.2436
Nugent BM, Wright CL, Shetty AC, Hodes GE, Lenz KM, Mahurkar A, Russo SJ, Devine SE, McCarthy MM (2015) Brain feminization requires active repression of masculinisation via DNA methylation. Nat Neurosci 18(5):690–697. https://doi.org/10.1038/nn.3988
Perera A, Eisen D, Wagner M, Laube SK, Künzel AF, Koch S, Steinbacher J, Schulze E, Splith V, Mittermeier N, Müller M, Biel M, Carell T, Michalakis S (2015) TET3 recruited by REST for contex-specific hydroxymethylation and induction of gene expression. Cell Rep 11:283–294. https://doi.org/10.1016/j.celrep.2015.03.020
Peterson CL, Laniel MA (2004) Histones and histone modifications. Curr Biol 14:R546–R551. https://doi.org/10.1016/j.cub.2004.07.007
Pusalkar M, Suri D, kelkar A, Bjattacjarya A, Galande S, Vaidya VA (2016) Early stress evokes dysregulation of histone modifiers in the medial prefrontal cortex across the life span. Dev Psychobiol 58:198–210. https://doi.org/10.1002/dev.21365
Roth TL, Lubin FD, Funk AJ, Sweatt JD (2009) Lasting epigenetic influence of early-life adversity on the BDNF gene. Biol Psychiatry 65:760–769. https://doi.org/10.1016/j.biopsych.2008.11.028
Ruthenberg AJ, Allis CD, Wysocka J (2007) Methylation of lysine 4 on histone H3: Intricacy of writing and reading a single epigenetic mark. Mol Cell 25:15–30. https://doi.org/10.1016/j.molcel.2006.12.014
Sailaja BS, Carmon DC, Zimmerman H, Meshorer E (2012) Stress-induced epigenetic transcriptional memory of acetylcholinesterase by HDAC4. Proc Natl Acad Sci U S A https://doi.org/10.1073/pnas.1209990110
Singh-Taylor AS, Molet J, Jiang S, Korosi A, Bolton JL, Noam Y, Simeone K, Cope J, Chen Y, Mortazavi A, Baram TZ (2018) NRSF-dependent epigenetic mechanisms contribute to programming of stress-sensitive neurons by neonatal experience, promoting resilience. Mol Psychiatry 23(3):648–657
Tahiliani M, Koh KP, Shen Y, Pastor WA, Bandukwala H, Brudno Y, Agarwal S, Iyer LM, Liu DR, Aravind L, Rao A (2009) Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by MLL partner TET1. Science 324: 930–935. https://doi.org/10.1126/science.1170116
Uchida S, Hara K, Kobayashi A, Funato H, Hobala T, Otsuki K, Yamagata H, McEwen BS, Watanabe Y (2010) Early life stress enhances behavioral vulnerability to stress through the activation of REST4-mediated gene transcription in the medial prefrontal cortex of rodents. J Neurosci 30(45):15007–15018
Veenema AH, Bredewold R, Neumann ID (2007) Opposite effects of maternal separation on intermale and maternal aggression in C57BL/6 mice: link to hypothalamic vasopressin and oxytocin immunoreactivity. Psychoneuroendocrinology 32(5):437–450. https://doi.org/10.1016/j.psyneuen.2007.02.008
Wang HT, Huang FL, Hu ZL, Zhang WJ, Qiao XQ, Dai RP, Li F, Li CQ (2017) Early-life social isolation-induced depressive-live behaviour in rats results in microglial activation and neuronal histone methylation that are mitigated by minocycline. Neurotox Res 31(4):505–520. https://doi.org/10.1007/s12640-016-9696-3
Zimmermann CA, Hoffmann A, Raabe F, Spengler D (2015) Role of Mecp2 in experience-dependent epigenetic programming. Genes 6:60–86. https://doi.org/10.3390/genes6010060
Acknowledgements
CK is recipient of University Research Fellowship from BDU. KER thank Department of Science and Technology for providing financial support through major project (EMR/2016/005217 Dated: 21.03.2018). Department of Animal Science supported by DST-PURSE, UGC-SAP-DRS-II and DST-FIST.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of Interest.
Ethical Approval
All procedures performed in studies involving animals were in accordance with the ethical standards of the institution or practice at which the studies were conducted.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Karen, C., Rajan, K.E. Social Behaviour and Epigenetic Status in Adolescent and Adult Rats: The Contribution of Early-Life Stressful Social Experience. Cell Mol Neurobiol 39, 371–385 (2019). https://doi.org/10.1007/s10571-019-00655-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-019-00655-x