Abstract
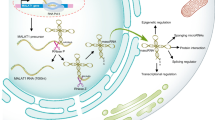
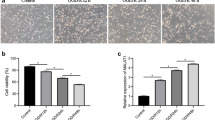
Metastasis-associated lung adenocarcinoma transcript 1 (MALAT1) is a long non-coding RNA contributing to protect the blood–brain barrier (BBB) after stroke. We searched for small molecules that may up-regulate MALAT1 and focused on polydatin (PD), a natural product, as a possible candidate. PD enhanced MALAT1 gene expression in rat brain microvascular endothelial cells, reducing cell toxicity and apoptosis after oxygen and glucose deprivation (OGD). These effects correlated with reduction of inflammatory factors and enhancement of expression of BBB markers. We found opposite changes after MALAT1 silencing. We determined that C/EBPβ is a key transcription factor for PD-mediated MALAT1 expression. PPARγ activity is involved in MALAT1 protective effects through its coactivator PGC-1α and the transcription factor CREB. This suggests that PD activates the MALAT1/CREB/PGC-1α/PPARγ signaling pathway to protect endothelial cells against ischemia. PD administration to rats subjected to brain ischemia by transient middle cerebral artery occlusion (tMCAO) reduced cerebral infarct volume and brain inflammation, protected cerebrovascular endothelial cells and BBB integrity. These effects correlated with increased expression of MALAT1, C/EBPβ, and PGC-1α. Our results strongly suggest that the beneficial effects of PD involve the C/EBPβ/MALAT1/CREB/PGC-1α/PPARγ pathway, which may provide a novel therapeutic strategy for brain ischemic stroke.











Similar content being viewed by others
Abbreviations
- ABCG1:
-
ATP-binding cassette sub-family G member 1
- BBB:
-
Blood–brain barrier
- CD36:
-
Cluster of differentiation 36
- C/EBPβ:
-
CCAAT/enhancer-binding proteinβ
- COX-2:
-
Cyclooxygenase-2
- CREB:
-
cAMP response element binding
- DMEM:
-
Dulbecco’s modified eagle’s medium
- DMSO:
-
Dimethyl sulfoxide
- FBS:
-
Fetal bovine serum
- HUVEC:
-
Human umbilical vein endothelial cells
- IL-6:
-
Interleukin-6
- LDH:
-
Lactate dehydrogenase
- LncRNA:
-
Long non-coding RNA
- MALAT1:
-
Metastasis-associated lung adenocarcinoma transcript 1
- MTT:
-
3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide
- OGD:
-
Oxygen and glucose deprivation
- PBS:
-
Phosphate-buffered saline
- PGC-1α:
-
Peroxisome proliferator-activated receptor gamma co-activator 1α
- PPARγ:
-
Peroxisome proliferative activated receptorγ
- rBMVEC:
-
Rat brain microvascular endothelial cell
- SD rats:
-
Sprague–Dawley rats
- tMCAO:
-
Transient middle cerebral artery occlusion
- TNF-α:
-
Tumor necrosis factor-α
- TTC:
-
2,3,5-Triphenyltetrazolium chloride
- ZO-1:
-
Zonula occludens-1
References
Akinrinmade O, Omoruyi S, Dietrich D, Ekpo O (2017) Long-term consumption of fermented rooibos herbal tea offers neuroprotection against ischemic brain injury in rats. Acta Neurobiol Exp 77(1):94–105
Belayev L, Hong SH, Menghani H, Marcell SJ, Obenaus A, Freitas RS, Khoutorova L, Balaszczuk V, Jun B, Oriá RB, Bazan NG (2018) Docosanoids promote neurogenesis and angiogenesis, blood-brain barrier integrity, penumbra protection, and neurobehavioral recovery after experimental ischemic stroke. Mol Neurobiol 55(8):7090-7106
Chang S, Ruan WC, Xu YZ, Wang YJ, Pang J, Zhang LY et al (2017) The natural product 4,10-aromadendranediol induces neuritogenesis in neuronal cells in vitro through activation of the ERK pathway. Acta Pharmacol Sin 38(1):29–40
Cheng Y, Zhang HT, Sun L, Guo S, Ouyang S, Zhang Y et al (2006) Involvement of cell adhesion molecules in polydatin protection of brain tissues from ischemia-reperfusion injury. Brain Res 1110(1):193–200
Chuang YF, Yang HY, Ko TL, Hsu YF, Sheu JR, Ou G et al (2014) Valproic acid suppresses lipopolysaccharide-induced cyclooxygenase-2 expression via MKP-1 in murine brain microvascular endothelial cells. Biochem Pharmacol 88(3):372–383
del Zoppo GJ (2006) Stroke and neurovascular protection. N Engl J Med 354(6):553–555
del Zoppo GJ (2009) Inflammation and the neurovascular unit in the setting of focal cerebral ischemia. Neuroscience 158(3):972–982
Doyle KP, Simon RP, Stenzelpoore MP (2008) Mechanisms of ischemic brain damage. Neuropharmacology 55(3):310–318
Fisher M (2008) Injuries to the vascular endothelium: vascular wall and endothelial dysfunction. Rev Neurol Dis 5(Suppl 1):S4–S11
Gao Y, Chen T, Lei X, Li Y, Dai X, Cao Y et al (2016) Neuroprotective effects of polydatin against mitochondrial-dependent apoptosis in the rat cerebral cortex following ischemia/reperfusion injury. Mol Med Rep 14(6):5481–5488
Guo W, Liu W, Chen Z, Gu Y, Peng S, Shen L et al (2017) Tyrosine phosphatase SHP2 negatively regulates NLRP3 inflammasome activation via ANT1-dependent mitochondrial homeostasis. Nat Commun 8(1):2168
Hawkins BT, Davis TP (2005) The blood–brain barrier/neurovascular unit in health and disease. Pharmacol Rev 57(2):173–185
Heward JA, Lindsay MA (2014) Long non-coding RNAs in the regulation of the immune response. Trends Immunol 35(9):408–419
Huang J, Upadhyay UM, Tamargo RJ (2006) Inflammation in stroke and focal cerebral ischemia. Surg Neurol 66(3):232–245
Huang K, Chen C, Hao J, Huang J, Wang S, Liu P et al (2015) Polydatin promotes Nrf2-ARE anti-oxidative pathway through activating Sirt1 to resist AGEs-induced upregulation of fibronetin and transforming growth factor-β1 in rat glomerular messangial cells. Mol Cell Endocrinol 399:178–189
Jinmo K, Miyeon K, Kanghyun L, Sangkwan M, Jamakattel-Pandit N, Hoyoung C et al (2010) Key compound groups for the neuroprotective effect of roots of Polygonum cuspidatum on transient middle cerebral artery occlusion in Sprague-Dawley rats. Nat Prod Res 24(13):1214–1226
Kenneth J, Thomas D (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2–∆∆CT method. Methods A 25(4):402–408
Kernan WN, Viscoli CM, Furie KL, Young LH, Inzucchi SE, Gorman M et al (2016) Pioglitazone after ischemic stroke or transient ischemic attack. N Engl J Med 64(1):260
Lee G, Elwood F, Mcnally J, Weiszmann J, Lindstrom M, Amaral K et al (2002) T0070907, a selective ligand for peroxisome proliferator-activated receptor γ, functions as an antagonist of biochemical and cellular activities. J Biol Chem 277(22):19649–19657
Lisa ML, Derek JP, Randy KB, Jeff EC, Jon LC, Thomas GC et al (2002) Functional consequences of cysteine modification in the ligand binding sites of peroxisome proliferator activated receptors by GW9662. Biochemistry 41(21):6640–6650
Liu J, Wang LN (2017) Peroxisome proliferator-activated receptor gamma agonists for preventing recurrent stroke and other vascular events in people with stroke or transient ischaemic attack. Cochrane Database Syst Rev 12:CD010693
Liu JY, Yao J, Li XM, Song YC, Wang XQ, Li YJ et al (2014) Pathogenic role of lncRNA-MALAT1 in endothelial cell dysfunction in diabetes mellitus. Cell Death Dis 5:e1506
Longa EZ, Weinstein PR, Carlson S, Cummins R (1989) Reversible middle cerebral artery occlusion without craniectomy in rats. Stroke 20(1):84–91
Lou T, Jiang W, Xu D, Chen T, Fu Y (2015) Inhibitory effects of polydatin on lipopolysaccharide-stimulated RAW 264.7 cells. Inflammation 38(3):1213–1220
Manea SA, Todirita A, Manea A (2013) High glucose-induced increased expression of endothelin-1 in human endothelial cells is mediated by activated CCAAT/enhancer-binding proteins. PLoS ONE 8(12):e84170
Medhi B, Aggarwal R, Chakrabarti A (2010) Neuroprotective effect of pioglitazone on acute phase changes induced by partial global cerebral ischemia in mice. Indian J Exp Biol 48(8):793–799
Michalik KM, You X, Manavski Y, Doddaballapur A, Zörnig M, Braun T et al (2014) Long noncoding RNA MALAT1 regulates endothelial cell function and vessel growth. Circ Res 114(9):1389–1397
Puthanveetil P, Chen S, Feng B, Gautam A, Chakrabarti S (2015) Long non-coding RNA malat1 regulates hyperglycaemia induced inflammatory process in the endothelial cells. J Cell Mol Med 19(6):1418–1425
Qureshi IA, Mehler MF (2012) Emerging roles of non-coding RNAs in brain evolution, development, plasticity and disease. Nat Rev Neurosci 13(8):528–541
Ruan W, Zhao F, Zhao S, Zhang L, Shi L, Pang T (2018) Knockdown of long noncoding RNA MEG3 impairs VEGF-stimulated endothelial sprouting angiogenesis via, modulating VEGFR2 expression in human umbilical vein endothelial cells. Gene 649:32–39
Ruetzler CA, Furuya K, Takeda H, Hallenbeck JM (2001) Brain vessels normally undergo cyclic activation and inactivation: evidence from tumor necrosis factor-alpha, heme oxygenase-1, and manganese superoxide dismutase immunostaining of vessels and perivascular brain cells. J Cereb Blood Flow Metab 21(3):244–252
Sandoval KE, Witt KA (2008) Blood–brain barrier tight junction permeability and ischemic stroke. Neurobiol Dis 32(2):200–219
Schellinger PD, Kaste M, Hacke W (2004) An update on thrombolytic therapy for acute stroke. Curr Opin Neurol 17(1):69–77
Sharabi K, Hua L, Tavares CDJ, Dominy JE, Camporez JP, Perry RJ et al (2017) Selective chemical inhibition of PGC-1α gluconeogenic activity ameliorates type 2 diabetes. Cell 169(1):148–160
Shi Y, Jiang X, Zhang L, Pu H, Hu X, Zhang W et al (2017) Endothelium-targeted overexpression of heat shock protein 27 ameliorates blood–brain barrier disruption after ischemic brain injury. Proc Natl Acad Sci USA 114(7):E1243–E1252
Singh S, Simpson RL, Bennett RG (2015) Relaxin activates peroxisome proliferator-activated receptor γ (PPARγ) through a pathway involving PPARγ coactivator 1α (PGC1α). J Biol Chem 290(2):950–959
Su WC, Kang BY, Kim SH, Pak YK, Cho D, Trinchieri G et al (2000) Oxidized low density lipoprotein inhibits interleukin-12 production in lipopolysaccharide-activated mouse macrophages via direct interactions between peroxisome proliferator-activated receptor-γ and nuclear factor-κB. J Biol Chem 275(42):32681–32687
Valluri S, Rupam G, Srividya S, George C (2017) Long non-coding rnas regulating immunity in insects. Noncoding RNA 3(1):14
Wang Q, Tang XN, Yenari MA (2007) The inflammatory response in stroke. J Neuroimmunol 184(1–2):53–68
Wang J, Li C, Chen T, Fang Y, Shi X, Pang T et al (2016) Nafamostat mesilate protects against acute cerebral ischemia via blood–brain barrier protection. Neuropharmacology 105:398–410
Wang W, Jiang B, Sun H, Ru X, Sun D, Wang L et al (2017) Prevalence, incidence and mortality of stroke in China: results from a nationwide population-based survey of 480,687 adults. Circulation 135(8):759–771
Wang Y, Ruan W, Mi J, Xu J, Wang H, Cao Z et al (2018a) Balasubramide derivative 3C modulates microglia activation via CaMKKβ-dependent AMPK/PGC-1α pathway in neuroinflammatory conditions. Brain Behav Immun 67:101–117
Wang Y, Huang Y, Xu Y, Ruan W, Wang H, Zhang Y et al (2018b) A Dual AMPK/Nrf2 activator reduces brain inflammation after stroke by enhancing microglia M2 polarization. Antioxid Redox Signal 28(2):141–163
Wu JS, Kao MH, Tsai HD, Cheung WM, Chen JJ, Ong WY et al (2018) Clinacanthus nutans mitigates neuronal apoptosis and ischemic brain damage through augmenting the C/EBPβ-driven PPAR-γ transcription. Mol Neurobiol 55(7):5425–5438
Xie F, Li BX, Kassenbrock A, Xue C, Wang X, Qian DZ et al (2015) Identification of a potent inhibitor of CREB-mediated gene transcription with efficacious in vivo anticancer activity. J Med Chem 58(12):5075–5087
Xu Y, Xu Y, Wang Y, Wang Y, He L, Jiang Z et al (2015) Telmisartan prevention of LPS-induced microglia activation involves M2 microglia polarization via CaMKKβ-dependent AMPK activation. Brain Behav Immun 50:298–313
Xu M, Yang X, Zeng Q, He H, Lu P, Huang G (2017) Birc5 is a novel target of peroxisome proliferator-activated receptor γ in brain microvascular endothelium cells during cerebral ischemia. Mol Med Rep 16(6):8882–8890
Xue H, Qiao Y, Ni P, Wang J, Chen C, Huang G (2011) A CRE that binds CREB and contributes to PKA-dependent regulation of the proximal promoter of human RAB25 gene. Int J Biochem Cell Biol 43(3):348–357
Yao J, Wang XQ, Li YJ, Shan K, Yang H, Wang YN et al (2016) Long non-coding RNA MALAT1 regulates retinal neurodegeneration through CREB signaling. EMBO Mol Med 8(4):346–362
Ye J, Piao H, Jiang J, Jin G, Zheng M, Yang J et al (2017) Polydatin inhibits mast cell-mediated allergic inflammation by targeting PI3K/Akt, MAPK, NF-κB and Nrf2/HO-1 pathways. Sci Rep 7(1):11895
Yin KJ, Hamblin M, Chen YE (2014) Non-coding RNAs in cerebral endothelial pathophysiology: emerging roles in stroke. Neurochem Int 77:9–16
Zhang JH, Badaut J, Tang J, Obenaus A, Hartman R, Pearce WJ (2012) The vascular neural network-a new paradigm in stroke pathophysiology. Nat Rev Neurol 8(12):711–716
Zhang J, Yuan L, Zhang X, Hamblin MH, Zhu T, Meng F et al (2016) Altered long non-coding RNA transcriptomic profiles in brain microvascular endothelium after cerebral ischemia. Exp Neurol 277:162–170
Zhang X, Tang X, Liu K, Hamblin MH, Yin KJ (2017) Long non-coding RNA Malat1 regulates cerebrovascular pathologies in ischemic stroke. J Neurosci 37(7):1797–1806
Zhao G, Jiang K, Wu H, Qiu C, Deng G, Peng X (2017) Polydatin reduces Staphylococcus aureus lipoteichoic acid-induced injury by attenuating reactive oxygen species generation and TLR2-NFkB signalling. J Cell Mol Med 21(11):2796–2808
Zilleßen P, Celner J, Kretschmann A, Pfeifer A, Racké K, Mayer P et al (2016) Metabolic role of dipeptidyl peptidase 4 (DPP4) in primary human (pre)adipocytes. Sci Rep 6:23074
Acknowledgements
This study was supported by the National Natural Science Foundation of China (81570236, 81870360, 81402385, 81571134, 81773995, 81320108029), the Natural Science Foundation of Jiangsu Province (BK20160032), “Double First-Class” University Project (CPU2018GY06, CPU2018GY20), the Six Talent Peaks Project of Jiangsu Province (T.P.), Shanghai Key Laboratory of Psychotic Disorders (13dz2260500), the Postgraduate Research & Practice Innovation Program of Jiangsu Province. We would like to acknowledge Dr. Xiujun Li of the Affiliated Drum Tower Hospital of Nanjing University Medical School, for providing pEGFP-C1-MALAT1 and control plasmids; Dr. Shanshan Guo of Ningxia Medical University for providing the C/EBPβ overexpression plasmid pcDNA3.1-C/EBPβ; Prof. Qin Jiang of Nanjing Medical University for generously providing us with the rat MALAT1 primer sequence. We also gratefully acknowledge the excellent technical assistance of Haojie Wang, Chenglong Gao, Tailin He and Xin Guan in the animal experiments.
Author information
Authors and Affiliations
Contributions
All authors listed contributed immensely to this study. WR and JL performed the experiments and wrote the paper. YX, YW, FZ, and XY performed the animal experiments and analyzed the data. HJ, LZ, JMS, LS, TP, as experts in molecular pharmacology provided technical supports and designed the research.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ruan, W., Li, J., Xu, Y. et al. MALAT1 Up-Regulator Polydatin Protects Brain Microvascular Integrity and Ameliorates Stroke Through C/EBPβ/MALAT1/CREB/PGC-1α/PPARγ Pathway. Cell Mol Neurobiol 39, 265–286 (2019). https://doi.org/10.1007/s10571-018-00646-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-018-00646-4