Abstract
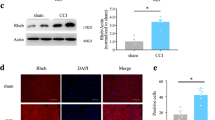
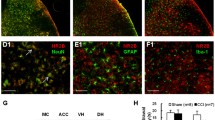
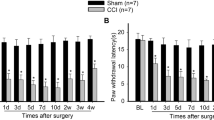
Phosphatidylinositol-3-kinase (PI3K) has been identified in the expression of central sensitization after noxious inflammatory stimuli. However, its contribution in neuropathic pain remains to be determined. Here we address the role of PI3K signaling in central sensitization in a model of neuropathic pain, and propose a novel potential drug target for neuropathic pain. Chronic constriction injury (CCI) rat model was used in the study as the model for neuropathic pain. Western blotting, whole-cell patch clamp, and von Frey assay were performed to study biochemical, electrical, and behavioral changes in CCI rats, respectively. A steroid metabolite of the fungi (wortmannin) was used to block PI3K signaling and its effects on CCI rats were tested. PI3K/Akt signaling increased in the spinal cord L4–L6 sections in the CCI rats. CCI also facilitated miniature excitatory postsynaptic potential of dorsal horn substantia gelatinosa neurons, increased phosphorylation of glutamate receptor subunit GluA1 and synapsin at the synapse, and induced mechanic allodynia. Wortmannin reversed biochemical, electrical, and behavioral changes in CCI rats. This study is the first to show PI3K/Akt signaling is required for spinal central sensitization in the CCI neuropathic pain model.





Similar content being viewed by others
References
Anggono V, Huganir RL (2012) Regulation of AMPA receptor trafficking and synaptic plasticity. Curr Opin Neurobiol 22:461–469. doi:10.1016/j.conb.2011.12.006
Baron R (2006) Mechanisms of disease: neuropathic pain–a clinical perspective. Nat Clin Pract Neurol 2:95–106. doi:10.1038/ncpneuro0113
Baron R, Binder A, Wasner G (2010) Neuropathic pain: diagnosis, pathophysiological mechanisms, and treatment. Lancet Neurol 9:807–819. doi:10.1016/S1474-4422(10)70143-5
Basbaum AI, Bautista DM, Scherrer G, Julius D (2009) Cellular and molecular mechanisms of pain. Cell 139:267–284. doi:10.1016/j.cell.2009.09.028
Bennett GJ, Xie YK (1988) A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain 33:87–107
Bouhassira D, Lanteri-Minet M, Attal N, Laurent B, Touboul C (2008) Prevalence of chronic pain with neuropathic characteristics in the general population. Pain 136:380–387. doi:10.1016/j.pain.2007.08.013
Bredt DS, Nicoll RA (2003) AMPA receptor trafficking at excitatory synapses. Neuron 40:361–379
Campbell JN, Meyer RA (2006) Mechanisms of neuropathic pain. Neuron 52:77–92. doi:10.1016/j.neuron.2006.09.021
Cantley LC (2002) The phosphoinositide 3-kinase pathway. Science 296:1655–1657. doi:10.1126/science.296.5573.1655
Chen SP et al (2017) PI3K/Akt pathway: a potential therapeutic target for chronic pain. Curr Pharm Des. doi:10.2174/1381612823666170210150147
Choi SR et al (2017) Spinal d-serine increases PKC-dependent GluN1 phosphorylation contributing to the Sigma-1 receptor-induced development of mechanical allodynia in a mouse model of neuropathic pain. J Pain 18:415–427. doi:10.1016/j.jpain.2016.12.002
Dworkin RH et al (2010) Recommendations for the pharmacological management of neuropathic pain: an overview and literature update. Mayo Clin Proc 85:S3–14. doi:10.4065/mcp.2009.0649
Ferby IM, Waga I, Hoshino M, Kume K, Shimizu T (1996) Wortmannin inhibits mitogen-activated protein kinase activation by platelet-activating factor through a mechanism independent of p85/p110-type phosphatidylinositol 3-kinase. J Biol Chem 271:11684–11688
Furman RR et al (2014) Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N Engl J Med 370:997–1007. doi:10.1056/NEJMoa1315226
Jensen TS, Finnerup NB (2014) Allodynia and hyperalgesia in neuropathic pain: clinical manifestations and mechanisms. Lancet Neurol 13:924–935. doi:10.1016/S1474-4422(14)70102-4
Ji RR, Strichartz G (2004) Cell signaling and the genesis of neuropathic pain. Sci STKE 2004:reE14. doi:10.1126/stke.2522004re14
Ji RR, Baba H, Brenner GJ, Woolf CJ (1999) Nociceptive-specific activation of ERK in spinal neurons contributes to pain hypersensitivity. Nat Neurosci 2:1114–1119. doi:10.1038/16040
Ji RR, Kohno T, Moore KA, Woolf CJ (2003) Central sensitization and LTP: do pain and memory share similar mechanisms? Trends Neurosci 26:696–705. doi:10.1016/j.tins.2003.09.017
Jiang SP, Zhang ZD, Kang LM, Wang QH, Zhang L, Chen HP (2016) Celecoxib reverts oxaliplatin-induced neuropathic pain through inhibiting PI3K/Akt2 pathway in the mouse dorsal root ganglion. Exp Neurol 275(Pt 1):11–16. doi:10.1016/j.expneurol.2015.11.001
Latremoliere A, Woolf CJ (2009) Central sensitization: a generator of pain hypersensitivity by central neural plasticity. J Pain 10:895–926. doi:10.1016/j.jpain.2009.06.012
Leinders M et al (2014) Differential distribution of PI3K isoforms in spinal cord and dorsal root ganglia: potential roles in acute inflammatory pain. Pain 155:1150–1160. doi:10.1016/j.pain.2014.03.003
Man HY et al (2003) Activation of PI3-kinase is required for AMPA receptor insertion during LTP of mEPSCs in cultured hippocampal neurons. Neuron 38:611–624
Miletic G, Hermes JL, Bosscher GL, Meier BM, Miletic V (2015) Protein kinase C gamma-mediated phosphorylation of GluA1 in the postsynaptic density of spinal dorsal horn neurons accompanies neuropathic pain, and dephosphorylation by calcineurin is associated with prolonged analgesia. Pain 156:2514–2520. doi:10.1097/j.pain.0000000000000323
Nakanishi S et al (1992) Wortmannin, a microbial product inhibitor of myosin light chain kinase. J Biol Chem 267:2157–2163
Nakanishi S, Catt KJ, Balla T (1995) A wortmannin-sensitive phosphatidylinositol 4-kinase that regulates hormone-sensitive pools of inositolphospholipids. Proc Natl Acad Sci USA 92:5317–5321
Niederberger E, Geisslinger G (2008) Proteomics in neuropathic pain research. Anesthesiology 108:314–323. doi:10.1097/01.anes.0000299838.13368.6e
Pezet S et al (2008) Phosphatidylinositol 3-kinase is a key mediator of central sensitization in painful inflammatory conditions. J Neurosci 28:4261–4270. doi:10.1523/JNEUROSCI.5392-07.2008
Qiu S et al (2014) GluA1 phosphorylation contributes to postsynaptic amplification of neuropathic pain in the insular cortex. J Neurosci 34:13505–13515. doi:10.1523/JNEUROSCI.1431-14.2014
Sandkuhler J (2009) Models and mechanisms of hyperalgesia and allodynia. Physiol Rev 89:707–758. doi:10.1152/physrev.00025.2008
Song SH, Augustine GJ (2015) Synapsin isoforms and synaptic vesicle trafficking. Mol Cells 38:936–940. doi:10.14348/molcells.2015.0233
Torrance N, Smith BH, Bennett MI, Lee AJ (2006) The epidemiology of chronic pain of predominantly neuropathic origin. Results from a general population survey. J Pain 7:281–289. doi:10.1016/j.jpain.2005.11.008
Valtorta F, Benfenati F, Greengard P (1992) Structure and function of the synapsins. J Biol Chem 267:7195–7198
Walker SM, Meredith-Middleton J, Lickiss T, Moss A, Fitzgerald M (2007) Primary and secondary hyperalgesia can be differentiated by postnatal age and ERK activation in the spinal dorsal horn of the rat pup. Pain 128:157–168. doi:10.1016/j.pain.2006.09.015
Woolf CJ, Salter MW (2000) Neuronal plasticity: increasing the gain in pain. Science 288:1765–1769
Xu JT, Tu HY, Xin WJ, Liu XG, Zhang GH, Zhai CH (2007) Activation of phosphatidylinositol 3-kinase and protein kinase B/Akt in dorsal root ganglia and spinal cord contributes to the neuropathic pain induced by spinal nerve ligation in rats. Exp Neurol 206:269–279. doi:10.1016/j.expneurol.2007.05.029
Yashpal K, Fisher K, Chabot JG, Coderre TJ (2001) Differential effects of NMDA and group I mGluR antagonists on both nociception and spinal cord protein kinase C translocation in the formalin test and a model of neuropathic pain in rats. Pain 94:17–29
Yawn BP, Wollan PC, Weingarten TN, Watson JC, Hooten WM, Melton LJ 3rd (2009) The prevalence of neuropathic pain: clinical evaluation compared with screening tools in a community population. Pain Med 10:586–593. doi:10.1111/j.1526-4637.2009.00588.x
Yu LN et al (2012) PI3K contributed to modulation of spinal nociceptive information related to ephrinBs/EphBs. PLoS ONE 7:e40930. doi:10.1371/journal.pone.0040930
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that they have no conflict of interest.
Research Involving Human and/or Animals Participants
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed.
Rights and permissions
About this article
Cite this article
Liu, W., Lv, Y. & Ren, F. PI3K/Akt Pathway is Required for Spinal Central Sensitization in Neuropathic Pain. Cell Mol Neurobiol 38, 747–755 (2018). https://doi.org/10.1007/s10571-017-0541-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-017-0541-x