Abstract
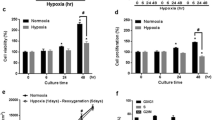
Transplantation of bone marrow stromal cells (BMSCs) is a promising therapy for ischemic stroke, but the poor oxygen environment in brain lesions limits the efficacy of cell-based therapies. Here, we tested whether hypoxic preconditioning (HP) could augment the efficacy of BMSC transplantation in a rat ischemic stroke model and investigated the underlying mechanism of the effect of HP. In vitro, BMSCs were divided into five passage (P0, P1, P2, P3, and P4) groups, and HP was applied to the groups by incubating the cells with 1% oxygen for 0, 4, 8, 12, and 24 h, respectively. We demonstrated that the expression of hypoxia-inducible factor-1α (HIF-1α) was increased in the HP-treated BMSCs, while their viability was unchanged. We also found that HP decreased the apoptosis of BMSCs during subsequent simulated ischemia–reperfusion (I/R) injury, especially in the 8-h HP group. In vivo, a rat transient focal cerebral ischemia model was established. These rats were administered normal cultured BMSCs (N-BMSCs), HP-treated BMSCs (H-BMSCs), or DMEM cell culture medium (control) at 24 h after the ischemic insult. Compared with the DMEM control group, the two BMSC-transplanted groups exhibited significantly improved functional recovery and reduced infarct volume, especially the H-BMSC group. Moreover, HP decreased neuronal apoptosis and enhanced the expression of BDNF and VEGF in the ischemic brain. Survival and differentiation of transplanted BMSCs were also increased by HP, and the quantity of engrafted BMSCs was significantly correlated with neurological function improvement. These results suggest that HP may enhance the therapeutic efficacy of BMSCs in an ischemic stroke model. The underlying mechanism likely involves the inhibition of caspase-3 activation and an increasing expression of HIF-1α, which promotes angiogenesis and neurogenesis and thereby reduces neuronal death and improves neurological function.







Similar content being viewed by others
References
Ahmad S, Elsherbiny NM, Haque R, Khan MB, Ishrat T, Shah ZA, Khan MM, Ali M, Jamal A, Katare DP, Liou GI, Bhatia K (2014) Sesamin attenuates neurotoxicity in mouse model of ischemic brain stroke. Neurotoxicology 45:100–110. doi:10.1016/j.neuro.2014.10.002
Andres RH, Choi R, Steinberg GK, Guzman R (2008) Potential of adult neural stem cells in stroke therapy. Regen Med 3(6):893–905. doi:10.2217/17460751.3.6.893
Cai XY, Xia Y, Yang SH, Liu XZ, Shao ZW, Liu YL, Yang W, Xiong LM (2015) Ropivacaine- and bupivacaine-induced death of rabbit annulus fibrosus cells in vitro: involvement of the mitochondrial apoptotic pathway. Osteoarthr Cartil. doi:10.1016/j.joca.2015.05.013
Chacko SM, Ahmed S, Selvendiran K, Kuppusamy ML, Khan M, Kuppusamy P (2010) Hypoxic preconditioning induces the expression of prosurvival and proangiogenic markers in mesenchymal stem cells. Am J Physiol Cell Physiol 299(6):C1562–C1570. doi:10.1152/ajpcell.00221.2010
Chang HY, Liu HS, Lai MD, Tsai YS, Tzai TS, Cheng HL, Chow NH (2014) Hypoxia promotes nuclear translocation and transcriptional function in the oncogenic tyrosine kinase RON. Cancer Res 74(16):4549–4562. doi:10.1158/0008-5472.CAN-13-3730
Chen J, Zhang C, Jiang H, Li Y, Zhang L, Robin A, Katakowski M, Lu M, Chopp M (2005) Atorvastatin induction of VEGF and BDNF promotes brain plasticity after stroke in mice. J Cereb Blood Flow Metab 25(2):281–290. doi:10.1038/sj.jcbfm.9600034
Cui QL, Almazan G (2007) IGF-I-induced oligodendrocyte progenitor proliferation requires PI3 K/Akt, MEK/ERK, and Src-like tyrosine kinases. J Neurochem 100(6):1480–1493. doi:10.1111/j.1471-4159.2006.04329.x
Deng W, Han Q, Liao L, Li C, Ge W, Zhao Z, You S, Deng H, Zhao RC (2004) Allogeneic bone marrow-derived flk-1 + Sca-1-mesenchymal stem cells leads to stable mixed chimerism and donor-specific tolerance. Exp Hematol 32(9):861–867. doi:10.1016/j.exphem.2004.06.009
Du S, Guan J, Mao G, Liu Y, Ma S, Bao X, Gao J, Feng M, Li G, Ma W, Yang Y, Zhao RC, Wang R (2014) Intra-arterial delivery of human bone marrow mesenchymal stem cells is a safe and effective way to treat cerebral ischemia in rats. Cell Transpl 23(Suppl 1):S73–S82. doi:10.3727/096368914X685023
Feng Y, Bhatt AJ (2015) Corticosteroid responses following hypoxic preconditioning provide neuroprotection against subsequent hypoxic-ischemic brain injury in the newborn rats. Int J Dev Neurosci 44:6–13. doi:10.1016/j.ijdevneu.2015.04.010
Francis KR, Wei L (2010) Human embryonic stem cell neural differentiation and enhanced cell survival promoted by hypoxic preconditioning. Cell Death Dis 1:e22. doi:10.1038/cddis.2009.22
Guo F, Lv S, Lou Y, Tu W, Liao W, Wang Y, Deng Z (2012) Bone marrow stromal cells enhance the angiogenesis in ischaemic cortex after stroke: involvement of notch signalling. Cell Biol Int 36(11):997–1004. doi:10.1042/CBI20110596
Haider H, Ashraf M (2008) Strategies to promote donor cell survival: combining preconditioning approach with stem cell transplantation. J Mol Cell Cardiol 45(4):554–566. doi:10.1016/j.yjmcc.2008.05.004
Horne MK, Nisbet DR, Forsythe JS, Parish CL (2010) Three-dimensional nanofibrous scaffolds incorporating immobilized BDNF promote proliferation and differentiation of cortical neural stem cells. Stem Cells Dev 19(6):843–852. doi:10.1089/scd.2009.0158
Huang W, Mo X, Qin C, Zheng J, Liang Z, Zhang C (2013) Transplantation of differentiated bone marrow stromal cells promotes motor functional recovery in rats with stroke. Neurol Res 35(3):320–328. doi:10.1179/1743132812Y.0000000151
Jung KH, Roh JK (2008) Circulating endothelial progenitor cells in cerebrovascular disease. J Clin Neurol 4(4):139–147. doi:10.3988/jcn.2008.4.4.139
Kaneko Y, Tajiri N, Shinozuka K, Glover LE, Weinbren NL, Cortes L, Borlongan CV (2012) Cell therapy for stroke: emphasis on optimizing safety and efficacy profile of endothelial progenitor cells. Curr Pharm Des 18(25):3731–3734
Keogh CL, Yu SP, Wei L (2007) The effect of recombinant human erythropoietin on neurovasculature repair after focal ischemic stroke in neonatal rats. J Pharmacol Exp Ther 322(2):521–528. doi:10.1124/jpet.107.121392
Lapi D, Vagnani S, Sapio D, Mastantuono T, Boscia F, Pignataro G, Penna C, Pagliaro P, Colantuoni A (2015) Effects of bone marrow mesenchymal stem cells (BM-MSCs) on rat pial microvascular remodeling after transient middle cerebral artery occlusion. Front Cell Neurosci 9:329. doi:10.3389/fncel.2015.00329
Liman TG, Endres M (2012) New vessels after stroke: postischemic neovascularization and regeneration. Cerebrovasc Dis 33(5):492–499. doi:10.1159/000337155
Liu L, Ning X, Han S, Zhang H, Sun L, Shi Y, Sun S, Guo C, Yin F, Qiao T, Wu K, Fan D (2008) Hypoxia induced HIF-1 accumulation and VEGF expression in gastric epithelial mucosa cell: involvement of ERK1/2 and PI3 K/Akt. Mol Biol (Mosk) 42(3):459–469
Liu J, Hao H, Xia L, Ti D, Huang H, Dong L, Tong C, Hou Q, Zhao Y, Liu H, Fu X, Han W (2015) Hypoxia pretreatment of bone marrow mesenchymal stem cells facilitates angiogenesis by improving the function of endothelial cells in diabetic rats with lower ischemia. PLoS ONE 10(5):e0126715. doi:10.1371/journal.pone.0126715
Malgieri A, Kantzari E, Patrizi MP, Gambardella S (2010) Bone marrow and umbilical cord blood human mesenchymal stem cells: state of the art. Int J Clin Exp Med 3(4):248–269
Mohyeldin A, Garzon-Muvdi T, Quinones-Hinojosa A (2010) Oxygen in stem cell biology: a critical component of the stem cell niche. Cell Stem Cell 7(2):150–161. doi:10.1016/j.stem.2010.07.007
Momin EN, Mohyeldin A, Zaidi HA, Vela G, Quinones-Hinojosa A (2010) Mesenchymal stem cells: new approaches for the treatment of neurological diseases. Curr Stem Cell Res Ther 5(4):326–344
Nishijima Y, Akamatsu Y, Weinstein PR, Liu J (2015) Collaterals: implications in cerebral ischemic diseases and therapeutic interventions. Brain Res 1623:18–29. doi:10.1016/j.brainres.2015.03.006
Oliveira MS, Barreto-Filho JB (2015) Placental-derived stem cells: culture, differentiation and challenges. World J Stem Cells 7(4):769–775. doi:10.4252/wjsc.v7.i4.769
Otero L, Zurita M, Bonilla C, Aguayo C, Rico MA, Rodriguez A, Vaquero J (2012) Allogeneic bone marrow stromal cell transplantation after cerebral hemorrhage achieves cell transdifferentiation and modulates endogenous neurogenesis. Cytotherapy 14(1):34–44. doi:10.3109/14653249.2011.608349
Parra-Cordero M, Rodrigo R, Barja P, Bosco C, Rencoret G, Sepulveda-Martinez A, Quezada S (2013) Prediction of early and late pre-eclampsia from maternal characteristics, uterine artery Doppler and markers of vasculogenesis during first trimester of pregnancy. Ultrasound Obstet Gynecol 41(5):538–544. doi:10.1002/uog.12264
Ren TJ, Qiang R, Jiang ZL, Wang GH, Sun L, Jiang R, Zhao GW, Han LY (2013) Improvement in regional CBF by l-serine contributes to its neuroprotective effect in rats after focal cerebral ischemia. PLoS ONE 8(6):e67044. doi:10.1371/journal.pone.0067044
Rowart P, Erpicum P, Detry O, Weekers L, Gregoire C, Lechanteur C, Briquet A, Beguin Y, Krzesinski JM, Jouret F (2015) Mesenchymal stromal cell therapy in ischemia/reperfusion injury. J Immunol Res 2015:602597. doi:10.1155/2015/602597
Schanzer A, Wachs FP, Wilhelm D, Acker T, Cooper-Kuhn C, Beck H, Winkler J, Aigner L, Plate KH, Kuhn HG (2004) Direct stimulation of adult neural stem cells in vitro and neurogenesis in vivo by vascular endothelial growth factor. Brain Pathol 14(3):237–248
Schmidt NO, Koeder D, Messing M, Mueller FJ, Aboody KS, Kim SU, Black PM, Carroll RS, Westphal M, Lamszus K (2009) Vascular endothelial growth factor-stimulated cerebral microvascular endothelial cells mediate the recruitment of neural stem cells to the neurovascular niche. Brain Res 1268:24–37. doi:10.1016/j.brainres.2009.02.065
Shehadah A, Chen J, Pal A, He S, Zeitlin A, Cui X, Zacharek A, Cui Y, Roberts C, Lu M, Hariri R, Chopp M (2014) Human placenta-derived adherent cell treatment of experimental stroke promotes functional recovery after stroke in young adult and older rats. PLoS ONE 9(1):e86621. doi:10.1371/journal.pone.0086621
Shen LH, Ye M, Ding XS, Han Q, Zhang C, Liu XF, Huang H, Wu EB, Huang HF, Gu XS (2012) Protective effects of MCI-186 on transplantation of bone marrow stromal cells in rat ischemic stroke model. Neuroscience 223:315–324. doi:10.1016/j.neuroscience.2012.08.001
Sicard KM, Henninger N, Fisher M, Duong TQ, Ferris CF (2006) Differential recovery of multimodal MRI and behavior after transient focal cerebral ischemia in rats. J Cereb Blood Flow Metabol 26(11):1451–1462. doi:10.1038/sj.jcbfm.9600299
Singh P, Kaur R, Kaur A (2013) Endovascular treatment of acute ischemic stroke. J Neurosci Rural Pract 4(3):298–303. doi:10.4103/0976-3147.118787
Sofroniew MV (2005) Reactive astrocytes in neural repair and protection. Neuroscientist 11(5):400–407. doi:10.1177/1073858405278321
Souidi N, Stolk M, Seifert M (2013) Ischemia-reperfusion injury: beneficial effects of mesenchymal stromal cells. Curr Opin Organ Transpl 18(1):34–43. doi:10.1097/MOT.0b013e32835c2a05
Sun J, Wei ZZ, Gu X, Zhang JY, Zhang Y, Li J, Wei L (2015) Intranasal delivery of hypoxia-preconditioned bone marrow-derived mesenchymal stem cells enhanced regenerative effects after intracerebral hemorrhagic stroke in mice. Exp Neurol 272:78–87. doi:10.1016/j.expneurol.2015.03.011
Taie S, Ono J, Iwanaga Y, Tomita S, Asaga T, Chujo K, Ueki M (2009) Hypoxia-inducible factor-1 alpha has a key role in hypoxic preconditioning. J Clin Neurosci 16(8):1056–1060. doi:10.1016/j.jocn.2008.09.024
Uchiyama T, Engelman RM, Maulik N, Das DK (2004) Role of Akt signaling in mitochondrial survival pathway triggered by hypoxic preconditioning. Circulation 109(24):3042–3049. doi:10.1161/01.CIR.0000130647.29030.90
van Velthoven CT, Kavelaars A, van Bel F, Heijnen CJ (2009) Regeneration of the ischemic brain by engineered stem cells: fuelling endogenous repair processes. Brain Res Rev 61(1):1–13. doi:10.1016/j.brainresrev.2009.03.003
Wang GH, Jiang ZL, Fan XJ, Zhang L, Li X, Ke KF (2007) Neuroprotective effect of taurine against focal cerebral ischemia in rats possibly mediated by activation of both GABAA and glycine receptors. Neuropharmacology 52(5):1199–1209. doi:10.1016/j.neuropharm.2006.10.022
Wang JA, Chen TL, Jiang J, Shi H, Gui C, Luo RH, Xie XJ, Xiang MX, Zhang X (2008a) Hypoxic preconditioning attenuates hypoxia/reoxygenation-induced apoptosis in mesenchymal stem cells. Acta Pharmacol Sin 29(1):74–82. doi:10.1111/j.1745-7254.2008.00716.x
Wang Y, Deng Y, Zhou GQ (2008b) SDF-1alpha/CXCR4-mediated migration of systemically transplanted bone marrow stromal cells towards ischemic brain lesion in a rat model. Brain Res 1195:104–112. doi:10.1016/j.brainres.2007.11.068
Wang F, Zhou H, Du Z, Chen X, Zhu F, Wang Z, Zhang Y, Lin L, Qian M, Zhang X, Li X, Hao A (2015) Cytoprotective effect of melatonin against hypoxia/serum deprivation-induced cell death of bone marrow mesenchymal stem cells in vitro. Eur J Pharmacol 748:157–165. doi:10.1016/j.ejphar.2014.09.033
Wei L, Fraser JL, Lu ZY, Hu X, Yu SP (2012) Transplantation of hypoxia preconditioned bone marrow mesenchymal stem cells enhances angiogenesis and neurogenesis after cerebral ischemia in rats. Neurobiol Dis 46(3):635–645. doi:10.1016/j.nbd.2012.03.002
Yan F, Yao Y, Chen L, Li Y, Sheng Z, Ma G (2012) Hypoxic preconditioning improves survival of cardiac progenitor cells: role of stromal cell derived factor-1alpha-CXCR4 axis. PLoS ONE 7(7):e37948. doi:10.1371/journal.pone.0037948
Yan T, Venkat P, Ye X, Chopp M, Zacharek A, Ning R, Cui Y, Roberts C, Kuzmin-Nichols N, Sanberg CD, Chen J (2014) HUCBCs increase angiopoietin 1 and induce neurorestorative effects after stroke in T1DM rats. CNS Neurosci Ther 20(10):935–944. doi:10.1111/cns.12307
Yu SP, Wei Z, Wei L (2013a) Preconditioning strategy in stem cell transplantation therapy. Transl Stroke Res 4(1):76–88. doi:10.1007/s12975-012-0251-0
Yu X, Lu C, Liu H, Rao S, Cai J, Liu S, Kriegel AJ, Greene AS, Liang M, Ding X (2013b) Hypoxic preconditioning with cobalt of bone marrow mesenchymal stem cells improves cell migration and enhances therapy for treatment of ischemic acute kidney injury. PLoS ONE 8(5):e62703. doi:10.1371/journal.pone.0062703
Yu Q, Liu L, Lin J, Wang Y, Xuan X, Guo Y, Hu S (2015) SDF-1alpha/CXCR4 Axis mediates the migration of mesenchymal stem cells to the hypoxic-ischemic brain lesion in a rat model. Cell J 16(4):440–447
Zhang F, Wang S, Luo Y, Ji X, Nemoto EM, Chen J (2009) When hypothermia meets hypotension and hyperglycemia: the diverse effects of adenosine 5′-monophosphate on cerebral ischemia in rats. J Cereb Blood Flow Metabol 29(5):1022–1034. doi:10.1038/jcbfm.2009.28
Zuliani T, Obriot H, Tual M, Lachman-Weber N, Dumas M, Formstecher P, Polakowska R, Ratinaud MH (2008) Variable Bax antigenicity is linked to keratinocyte position within epidermal strata and UV-induced apoptosis. Exp Dermatol 17(2):125–132. doi:10.1111/j.1600-0625.2007.00660.x
Funding
This work was supported by the Chinese Natural Science Foundation (Grants 81000497 and 81471257) and Natural Science Foundation of Jiangsu Province of China (Grant BK20161283), and sponsored by Qing Lan Project (to G.W). The funders had no role in the study design, the data collection and analysis, the decision to publish, or the preparation of the manuscript.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare the absence of competing interests.
Additional information
Jin Chen and Yuanyuan Yang have contributed equally to this work.
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
10571_2016_445_MOESM1_ESM.tif
Fig. S1. Confocal microscopy detected that HP promote the increase of BrdU-positive cells expressing nestin. Double immunofluorescence staining for engrafted BMSCs (BrdU-positive cells, red) and Nestin (green)-positive cells in the ischemic brain at day 7 after occlusion. Scale bar 250 μm. (TIFF 3697 kb)
Rights and permissions
About this article
Cite this article
Chen, J., Yang, Y., Shen, L. et al. Hypoxic Preconditioning Augments the Therapeutic Efficacy of Bone Marrow Stromal Cells in a Rat Ischemic Stroke Model. Cell Mol Neurobiol 37, 1115–1129 (2017). https://doi.org/10.1007/s10571-016-0445-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-016-0445-1