Abstract
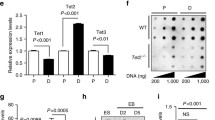
Adult neurogenesis originates from neural stem cells (NSCs) in specific regions of the adult brain. The molecular mechanisms that control the self-renewal and multipotency of NSCs have not been fully elucidated. In recent years, emerging evidence has revealed that ten-eleven translocation (TET) family DNA dioxygenases TET1 and TET2 play important roles in the central nervous system. Here, I present evidence that Tet1 and Tet2 are expressed in cultured NSCs derived from adult mouse brain and play an important role in the proliferative self-renewal of NSCs in an undifferentiated state. The investigation of intracellular molecular networks involving both Tet1 and Tet2 by gene knockdown and comprehensive genetic analyses showed that overlapping molecular mechanisms involving TET1 and TET2 regulate the expression of at least 16 genes required for DNA replication and cell cycle control. Interestingly, transcriptional regulation of the selected gene through TET1 and TET2 did not correlate with direct CpG demethylation of the gene promoter. These findings suggest that TET1 and TET2 play an important role in the proliferation of NSCs in the adult mouse brain by specifically regulating common genes for DNA replication and the cell cycle.






Similar content being viewed by others
References
Abbosh C, Lawkowski A, Zaben M, Gray W (2011) GalR2/3 mediates proliferative and trophic effects of galanin on postnatal hippocampal precursors. J Neurochem 117(3):425–436. doi:10.1111/j.1471-4159.2011.07204.x
Alvarez-Buylla A, Lim DA (2004) For the long run: maintaining germinal niches in the adult brain. Neuron 41(5):683–686
Chattopadhyay S, Bielinsky AK (2007) Human Mcm10 regulates the catalytic subunit of DNA polymerase-alpha and prevents DNA damage during replication. Mol Biol Cell 18(10):4085–4095. doi:10.1091/mbc.E06-12-1148
Cimmino L, Abdel-Wahab O, Levine RL, Aifantis I (2011) TET family proteins and their role in stem cell differentiation and transformation. Cell Stem Cell 9(3):193–204. doi:10.1016/j.stem.2011.08.007
Clelland CD, Choi M, Romberg C, Clemenson GD Jr, Fragniere A, Tyers P, Jessberger S, Saksida LM, Barker RA, Gage FH, Bussey TJ (2009) A functional role for adult hippocampal neurogenesis in spatial pattern separation. Science 325(5937):210–213. doi:10.1126/science.1173215
Coskun V, Tsoa R, Sun YE (2012) Epigenetic regulation of stem cells differentiating along the neural lineage. Curr Opin Neurobiol 22(5):762–767. doi:10.1016/j.conb.2012.07.001
Dawlaty MM, Breiling A, Le T, Raddatz G, Barrasa MI, Cheng AW, Gao Q, Powell BE, Li Z, Xu M, Faull KF, Lyko F, Jaenisch R (2013) Combined deficiency of Tet1 and Tet2 causes epigenetic abnormalities but is compatible with postnatal development. Dev Cell 24(3):310–323. doi:10.1016/j.devcel.2012.12.015
Delhommeau F, Dupont S, Della Valle V, James C, Trannoy S, Masse A, Kosmider O, Le Couedic JP, Robert F, Alberdi A, Lecluse Y, Plo I, Dreyfus FJ, Marzac C, Casadevall N, Lacombe C, Romana SP, Dessen P, Soulier J, Viguie F, Fontenay M, Vainchenker W, Bernard OA (2009) Mutation in TET2 in myeloid cancers. N Engl J Med 360(22):2289–2301. doi:10.1056/NEJMoa0810069
Doetsch F, Garcia-Verdugo JM, Alvarez-Buylla A (1997) Cellular composition and three-dimensional organization of the subventricular germinal zone in the adult mammalian brain. J Neurosci 17(13):5046–5061
Doetsch F, Caille I, Lim DA, Garcia-Verdugo JM, Alvarez-Buylla A (1999) Subventricular zone astrocytes are neural stem cells in the adult mammalian brain. Cell 97(6):703–716
Edwards S, Li CM, Levy DL, Brown J, Snow PM, Campbell JL (2003) Saccharomyces cerevisiae DNA polymerase epsilon and polymerase sigma interact physically and functionally, suggesting a role for polymerase epsilon in sister chromatid cohesion. Mol Cell Biol 23(8):2733–2748
Eki T, Okumura K, Amin A, Ishiai M, Abe M, Nogami M, Taguchi H, Hurwitz J, Murakami Y, Hanaoka F (1996) Mapping of the human homologue (ORC1L) of the yeast origin recognition complex subunit 1 gene to chromosome band 1p32. Genomics 36(3):559–561. doi:10.1006/geno.1996.0511
Fabel K, Tam B, Kaufer D, Baiker A, Simmons N, Kuo CJ, Palmer TD (2003) VEGF is necessary for exercise-induced adult hippocampal neurogenesis. Eur J Neurosci 18(10):2803–2812
Forsburg SL (2004) Eukaryotic MCM proteins: beyond replication initiation. Microbiol Mol Biol Rev 68(1):109–131
Freudenberg JM, Ghosh S, Lackford BL, Yellaboina S, Zheng X, Li R, Cuddapah S, Wade PA, Hu G, Jothi R (2012) Acute depletion of Tet1-dependent 5-hydroxymethylcytosine levels impairs LIF/Stat3 signaling and results in loss of embryonic stem cell identity. Nucleic Acids Res 40(8):3364–3377. doi:10.1093/nar/gkr1253
Gage FH (2000) Mammalian neural stem cells. Science 287(5457):1433–1438
Hitoshi S, Ishino Y, Kumar A, Jasmine S, Tanaka KF, Kondo T, Kato S, Hosoya T, Hotta Y, Ikenaka K (2011) Mammalian Gcm genes induce Hes5 expression by active DNA demethylation and induce neural stem cells. Nat Neurosci 14(8):957–964. doi:10.1038/nn.2875
Huang Y, Wang G, Liang Z, Yang Y, Cui L, Liu CY (2016) Loss of nuclear localization of TET2 in colorectal cancer. Clin Epigenetics 8:9. doi:10.1186/s13148-016-0176-7176
Ito S, D’Alessio AC, Taranova OV, Hong K, Sowers LC, Zhang Y (2010) Role of Tet proteins in 5mC to 5hmC conversion, ES-cell self-renewal and inner cell mass specification. Nature 466(7310):1129–1133. doi:10.1038/nature09303
Izumi M, Yanagi K, Mizuno T, Yokoi M, Kawasaki Y, Moon KY, Hurwitz J, Yatagai F, Hanaoka F (2000) The human homolog of Saccharomyces cerevisiae Mcm10 interacts with replication factors and dissociates from nuclease-resistant nuclear structures in G(2) phase. Nucleic Acids Res 28(23):4769–4777
Johnson RE, Klassen R, Prakash L, Prakash S (2015) A major role of DNA polymerase delta in replication of both the leading and lagging DNA strands. Mol Cell 59(2):163–175. doi:10.1016/j.molcel.2015.05.038
Johnston ST, Shtrahman M, Parylak S, Goncalves JT, Gage FH (2016) Paradox of pattern separation and adult neurogenesis: a dual role for new neurons balancing memory resolution and robustness. Neurobiol Learn Mem 129:60–68. doi:10.1016/j.nlm.2015.10.013
Kaas GA, Zhong C, Eason DE, Ross DL, Vachhani RV, Ming GL, King JR, Song H, Sweatt JD (2013) TET1 controls CNS 5-methylcytosine hydroxylation, active DNA demethylation, gene transcription, and memory formation. Neuron 79(6):1086–1093. doi:10.1016/j.neuron.2013.08.032
Kanke M, Kodama Y, Takahashi TS, Nakagawa T, Masukata H (2012) Mcm10 plays an essential role in origin DNA unwinding after loading of the CMG components. EMBO J 31(9):2182–2194. doi:10.1038/emboj.2012.68
Kempermann G, Jessberger S, Steiner B, Kronenberg G (2004) Milestones of neuronal development in the adult hippocampus. Trends Neurosci 27(8):447–452. doi:10.1016/j.tins.2004.05.013S0166223604001675
Kim H, Jang WY, Kang MC, Jeong J, Choi M, Sung Y, Park S, Kwon W, Jang S, Kim MO, Kim SH, Ryoo ZY (2016) TET1 contributes to neurogenesis onset time during fetal brain development in mice. Biochem Biophys Res Commun 471(4):437–443. doi:10.1016/j.bbrc.2016.02.060
Kirby ED, Kuwahara AA, Messer RL, Wyss-Coray T (2015) Adult hippocampal neural stem and progenitor cells regulate the neurogenic niche by secreting VEGF. Proc Natl Acad Sci USA 112(13):4128–4133. doi:10.1073/pnas.1422448112
Kneissl M, Putter V, Szalay AA, Grummt F (2003) Interaction and assembly of murine pre-replicative complex proteins in yeast and mouse cells. J Mol Biol 327(1):111–128
Kondo T, Raff M (2004) Chromatin remodeling and histone modification in the conversion of oligodendrocyte precursors to neural stem cells. Genes Dev 18(23):2963–2972. doi:10.1101/gad.309404
Kriaucionis S, Heintz N (2009) The nuclear DNA base 5-hydroxymethylcytosine is present in Purkinje neurons and the brain. Science 324(5929):929–930. doi:10.1126/science.1169786
Kucharova K, Stallcup WB (2010) The NG2 proteoglycan promotes oligodendrocyte progenitor proliferation and developmental myelination. Neuroscience 166(1):185–194. doi:10.1016/j.neuroscience.2009.12.014
Kumar D, Aggarwal M, Kaas GA, Lewis J, Wang J, Ross DL, Zhong C, Kennedy A, Song H, Sweatt JD (2015) Tet1 oxidase regulates neuronal gene transcription, active DNA hydroxy-methylation, object location memory, and threat recognition memory. Neuroepigenetics 4:12–27. doi:10.1016/j.nepig.2015.10.002
Kunimoto H, Fukuchi Y, Sakurai M, Sadahira K, Ikeda Y, Okamoto S, Nakajima H (2012) Tet2 disruption leads to enhanced self-renewal and altered differentiation of fetal liver hematopoietic stem cells. Sci Rep 2:273. doi:10.1038/srep00273
Labib K, Tercero JA, Diffley JF (2000) Uninterrupted MCM2-7 function required for DNA replication fork progression. Science 288(5471):1643–1647
Langemeijer SM, Kuiper RP, Berends M, Knops R, Aslanyan MG, Massop M, Stevens-Linders E, van Hoogen P, van Kessel AG, Raymakers RA, Kamping EJ, Verhoef GE, Verburgh E, Hagemeijer A, Vandenberghe P, de Witte T, van der Reijden BA, Jansen JH (2009) Acquired mutations in TET2 are common in myelodysplastic syndromes. Nat Genet 41(7):838–842. doi:10.1038/ng.391
Lei M, Tye BK (2001) Initiating DNA synthesis: from recruiting to activating the MCM complex. J Cell Sci 114(Pt 8):1447–1454
Li Y, Benezra R (1996) Identification of a human mitotic checkpoint gene: hsMAD2. Science 274(5285):246–248
Louis SA, Rietze RL, Deleyrolle L, Wagey RE, Thomas TE, Eaves AC, Reynolds BA (2008) Enumeration of neural stem and progenitor cells in the neural colony-forming cell assay. Stem Cells 26(4):988–996. doi:10.1634/stemcells.2007-0867
Lugert S, Basak O, Knuckles P, Haussler U, Fabel K, Gotz M, Haas CA, Kempermann G, Taylor V, Giachino C (2010) Quiescent and active hippocampal neural stem cells with distinct morphologies respond selectively to physiological and pathological stimuli and aging. Cell Stem Cell 6(5):445–456. doi:10.1016/j.stem.2010.03.017
Lygerou Z, Nurse P (2000) Cell cycle. License withheld–geminin blocks DNA replication. Science 290(5500):2271–2273
Mi Y, Gao X, Dai J, Ma Y, Xu L, Jin W (2015) A novel function of TET2 in CNS: sustaining neuronal survival. Int J Mol Sci 16(9):21846–21857. doi:10.3390/ijms160921846
Montalban-Loro R, Domingo-Muelas A, Bizy A, Ferron SR (2015) Epigenetic regulation of stemness maintenance in the neurogenic niches. World J Stem Cells 7(4):700–710. doi:10.4252/wjsc.v7.i4.700
Moran-Crusio K, Reavie L, Shih A, Abdel-Wahab O, Ndiaye-Lobry D, Lobry C, Figueroa ME, Vasanthakumar A, Patel J, Zhao X, Perna F, Pandey S, Madzo J, Song C, Dai Q, He C, Ibrahim S, Beran M, Zavadil J, Nimer SD, Melnick A, Godley LA, Aifantis I, Levine RL (2011) Tet2 loss leads to increased hematopoietic stem cell self-renewal and myeloid transformation. Cancer Cell 20(1):11–24. doi:10.1016/j.ccr.2011.06.001
Nakashiba T, Cushman JD, Pelkey KA, Renaudineau S, Buhl DL, McHugh TJ, Rodriguez Barrera V, Chittajallu R, Iwamoto KS, McBain CJ, Fanselow MS, Tonegawa S (2012) Young dentate granule cells mediate pattern separation, whereas old granule cells facilitate pattern completion. Cell 149(1):188–201. doi:10.1016/j.cell.2012.01.046
Namihira M, Kohyama J, Semi K, Sanosaka T, Deneen B, Taga T, Nakashima K (2009) Committed neuronal precursors confer astrocytic potential on residual neural precursor cells. Dev Cell 16(2):245–255. doi:10.1016/j.devcel.2008.12.014
Neri F, Dettori D, Incarnato D, Krepelova A, Rapelli S, Maldotti M, Parlato C, Paliogiannis P, Oliviero S (2015a) TET1 is a tumour suppressor that inhibits colon cancer growth by derepressing inhibitors of the WNT pathway. Oncogene 34(32):4168–4176. doi:10.1038/onc.2014.356
Neri F, Incarnato D, Krepelova A, Dettori D, Rapelli S, Maldotti M, Parlato C, Anselmi F, Galvagni F, Oliviero S (2015b) TET1 is controlled by pluripotency-associated factors in ESCs and downmodulated by PRC2 in differentiated cells and tissues. Nucleic Acids Res 43(14):6814–6826. doi:10.1093/nar/gkv392
Okumura K, Nogami M, Taguchi H, Dean FB, Chen M, Pan ZQ, Hurwitz J, Shiratori A, Murakami Y, Ozawa K et al (1995) Assignment of the 36.5-kDa (RFC5), 37-kDa (RFC4), 38-kDa (RFC3), and 40-kDa (RFC2) subunit genes of human replication factor C to chromosome bands 12q24.2-q24.3, 3q27, 13q12.3-q13, and 7q11.23. Genomics 25(1):274–278
Olynik BM, Rastegar M (2012) The genetic and epigenetic journey of embryonic stem cells into mature neural cells. Front Genet 3:81. doi:10.3389/fgene.2012.00081
Ray J, Gage FH (2006) Differential properties of adult rat and mouse brain-derived neural stem/progenitor cells. Mol Cell Neurosci 31(3):560–573. doi:10.1016/j.mcn.2005.11.010
Rudenko A, Dawlaty MM, Seo J, Cheng AW, Meng J, Le T, Faull KF, Jaenisch R, Tsai LH (2013) Tet1 is critical for neuronal activity-regulated gene expression and memory extinction. Neuron 79(6):1109–1122. doi:10.1016/j.neuron.2013.08.003
Sahay A, Scobie KN, Hill AS, O’Carroll CM, Kheirbek MA, Burghardt NS, Fenton AA, Dranovsky A, Hen R (2011) Increasing adult hippocampal neurogenesis is sufficient to improve pattern separation. Nature 472(7344):466–470. doi:10.1038/nature09817
Santarelli L, Saxe M, Gross C, Surget A, Battaglia F, Dulawa S, Weisstaub N, Lee J, Duman R, Arancio O, Belzung C, Hen R (2003) Requirement of hippocampal neurogenesis for the behavioral effects of antidepressants. Science 301(5634):805–809. doi:10.1126/science.1083328301/5634/805
Shimozaki K, Clemenson GD Jr, Gage FH (2013) Paired related homeobox protein 1 is a regulator of stemness in adult neural stem/progenitor cells. J Neurosci 33(9):4066–4075. doi:10.1523/JNEUROSCI.4586-12.2013
Shinohara A, Ogawa H, Ogawa T (1992) Rad51 protein involved in repair and recombination in S. cerevisiae is a RecA-like protein. Cell 69(3):457–470
Shinohara A, Ogawa H, Matsuda Y, Ushio N, Ikeo K, Ogawa T (1993) Cloning of human, mouse and fission yeast recombination genes homologous to RAD51 and recA. Nat Genet 4(3):239–243. doi:10.1038/ng0793-239
Snyder JS, Soumier A, Brewer M, Pickel J, Cameron HA (2011) Adult hippocampal neurogenesis buffers stress responses and depressive behaviour. Nature 476(7361):458–461. doi:10.1038/nature10287
Spella M, Britz O, Kotantaki P, Lygerou Z, Nishitani H, Ramsay RG, Flordellis C, Guillemot F, Mantamadiotis T, Taraviras S (2007) Licensing regulators Geminin and Cdt1 identify progenitor cells of the mouse CNS in a specific phase of the cell cycle. Neuroscience 147(2):373–387. doi:10.1016/j.neuroscience.2007.03.050
Steiner B, Klempin F, Wang L, Kott M, Kettenmann H, Kempermann G (2006) Type-2 cells as link between glial and neuronal lineage in adult hippocampal neurogenesis. Glia 54(8):805–814. doi:10.1002/glia.20407
Sun Y, Jin K, Mao XO, Zhu Y, Greenberg DA (2001) Neuroglobin is up-regulated by and protects neurons from hypoxic-ischemic injury. Proc Natl Acad Sci USA 98(26):15306–15311. doi:10.1073/pnas.251466698251466698
Szulwach KE, Li X, Li Y, Song CX, Wu H, Dai Q, Irier H, Upadhyay AK, Gearing M, Levey AI, Vasanthakumar A, Godley LA, Chang Q, Cheng X, He C, Jin P (2011) 5-hmC-mediated epigenetic dynamics during postnatal neurodevelopment and aging. Nat Neurosci 14(12):1607–1616. doi:10.1038/nn.2959
Tahiliani M, Koh KP, Shen Y, Pastor WA, Bandukwala H, Brudno Y, Agarwal S, Iyer LM, Liu DR, Aravind L, Rao A (2009) Conversion of 5-methylcytosine to 5-hydroxymethylcytosine in mammalian DNA by MLL partner TET1. Science 324(5929):930–935. doi:10.1126/science.1170116
Takizawa T, Nakashima K, Namihira M, Ochiai W, Uemura A, Yanagisawa M, Fujita N, Nakao M, Taga T (2001) DNA methylation is a critical cell-intrinsic determinant of astrocyte differentiation in the fetal brain. Dev Cell 1(6):749–758
Tye BK, Sawyer S (2000) The hexameric eukaryotic MCM helicase: building symmetry from nonidentical parts. J Biol Chem 275(45):34833–34836. doi:10.1074/jbc.R000018200R000018200
Watase G, Takisawa H, Kanemaki MT (2012) Mcm10 plays a role in functioning of the eukaryotic replicative DNA helicase, Cdc45-Mcm-GINS. Curr Biol 22(4):343–349. doi:10.1016/j.cub.2012.01.023
Wiehle L, Raddatz G, Musch T, Dawlaty MM, Jaenisch R, Lyko F, Breiling A (2015) Tet1 and Tet2 Protect DNA Methylation Canyons against Hypermethylation. Mol Cell Biol 36(3):452–461. doi:10.1128/MCB.00587-15
Williams K, Christensen J, Pedersen MT, Johansen JV, Cloos PA, Rappsilber J, Helin K (2011) TET1 and hydroxymethylcytosine in transcription and DNA methylation fidelity. Nature 473(7347):343–348. doi:10.1038/nature10066
Wu BK, Brenner C (2014) Suppression of TET1-dependent DNA demethylation is essential for KRAS-mediated transformation. Cell Rep 9(5):1827–1840. doi:10.1016/j.celrep.2014.10.063
Wu H, D’Alessio AC, Ito S, Wang Z, Cui K, Zhao K, Sun YE, Zhang Y (2011) Genome-wide analysis of 5-hydroxymethylcytosine distribution reveals its dual function in transcriptional regulation in mouse embryonic stem cells. Genes Dev 25(7):679–684. doi:10.1101/gad.2036011
Xu Y, Wu F, Tan L, Kong L, Xiong L, Deng J, Barbera AJ, Zheng L, Zhang H, Huang S, Min J, Nicholson T, Chen T, Xu G, Shi Y, Zhang K, Shi YG (2011) Genome-wide regulation of 5hmC, 5mC, and gene expression by Tet1 hydroxylase in mouse embryonic stem cells. Mol Cell 42(4):451–464. doi:10.1016/j.molcel.2011.04.005
Yao J, Mu Y, Gage FH (2012) Neural stem cells: mechanisms and modeling. Protein Cell 3(4):251–261. doi:10.1007/s13238-012-2033-6
Zhang CL, Zou Y, He W, Gage FH, Evans RM (2008) A role for adult TLX-positive neural stem cells in learning and behaviour. Nature 451(7181):1004–1007. doi:10.1038/nature06562
Zhang RR, Cui QY, Murai K, Lim YC, Smith ZD, Jin S, Ye P, Rosa L, Lee YK, Wu HP, Liu W, Xu ZM, Yang L, Ding YQ, Tang F, Meissner A, Ding C, Shi Y, Xu GL (2013) Tet1 regulates adult hippocampal neurogenesis and cognition. Cell Stem Cell 13(2):237–245. doi:10.1016/j.stem.2013.05.006
Zhao C, Deng W, Gage FH (2008) Mechanisms and functional implications of adult neurogenesis. Cell 132(4):645–660. doi:10.1016/j.cell.2008.01.033
Zhao Z, Chen L, Dawlaty MM, Pan F, Weeks O, Zhou Y, Cao Z, Shi H, Wang J, Lin L, Chen S, Yuan W, Qin Z, Ni H, Nimer SD, Yang FC, Jaenisch R, Jin P, Xu M (2015) Combined loss of Tet1 and Tet2 promotes B cell, but not myeloid malignancies. Mice. Cell Rep 13(8):1692–1704. doi:10.1016/j.celrep.2015.10.037
Zhu Y, Jin K, Mao XO, Greenberg DA (2003) Vascular endothelial growth factor promotes proliferation of cortical neuron precursors by regulating E2F expression. FASEB J 17(2):186–193. doi:10.1096/fj.02-0515com17/2/186
Acknowledgments
I thank K. Tsurunaga and the members of the Life Science Support Center and Atomic Bomb Disease Institute at Nagasaki University for technical assistance and helpful discussions. Crimson Interactive Pvt. Ltd. (Ulatus) and Edanz Group Japan are acknowledged for their assistance with preparation of the manuscript. This work was supported by a Grant-in-Aid for Scientific Research (C) Grant Number 15K06711 from the Japan Society for the Promotion of Science (JSPS) and a Grant-in-Aid of the Alumni Association of Nagasaki University School of Medicine.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
10571_2016_432_MOESM1_ESM.tiff
Gene expression of TET family members in cultured adult NSCs analyzed by conventional RT-PCR. Total RNA from cultured adult NSCs was reverse transcribed with [RT (+)] or without [RT (−)] reverse transcriptase and then amplified by the hot start-based PCR method with specific primer sets for Tet1, Tet2, and Tet3 genes. Supplementary material 1 (TIFF 2884 kb)
Rights and permissions
About this article
Cite this article
Shimozaki, K. Ten-Eleven Translocation 1 and 2 Confer Overlapping Transcriptional Programs for the Proliferation of Cultured Adult Neural Stem Cells. Cell Mol Neurobiol 37, 995–1008 (2017). https://doi.org/10.1007/s10571-016-0432-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-016-0432-6