Abstract
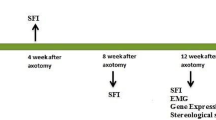
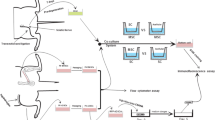
This study aimed to evaluate whether combination therapy of bone marrow stromal cells (BMSCs) transplantation and chondroitinase ABC (ChABC) treatment further enhances axonal regeneration and functional recovery after acellular nerve allograft repair of the sciatic nerve gap in rats. Eight Sprague–Dawley rats were used as nerve donors, and 32 Wistar rats were randomly divided into four groups: Group I: acellular rat sciatic nerve (ARSN) group; Group II: ChABC treatment; Group III: BMSCs transplantation; and Group IV: ChABC treatment and BMSCs transplantation. The results showed that compared with ARSN control group, BMSC transplantation promoted axonal regeneration, the secretion of neural trophic factors NGF, BDNF and axon angiogenesis in nerve graft. ChABC treatment degraded chondroitin sulfate proteoglycans in ARSN in vitro and in vivo and improved BMSCs survival in ARSN. The combination therapy caused much better beneficial effects evidenced by increasing sciatic function index, nerve conduction velocity, restoration rate of tibialis anterior wet muscle weight, and myelinated nerve number, but did not further boost the therapeutic effects on neurotrophic factor production, axon angiogenesis, and sensory functional recovery by BMSC transplantation. Taken together, for the first time, we demonstrate the synergistic effects of BMSC transplantation and BMSCs treatment on peripheral nerve regeneration, and our findings may help establish novel strategies for cell transplantation therapy for peripheral nerve injury.






Similar content being viewed by others
References
Chau CH, Shum DK, Li H, Pei J, Lui YY, Wirthlin L, Chan YS, Xu XM (2004) Chondroitinase ABC enhances axonal regrowth through Schwann cell-seeded guidance channels after spinal cord injury. FASEB J 18(1):194–196
Chen CJ, Ou YC, Liao SL, Chen WY, Chen SY, Wu CW, Wang CC, Wang WY, Huang YS, Hsu SH (2007) Transplantation of bone marrow stromal cells for peripheral nerve repair. Exp Neurol 204(1):443–453
Cuevas P, Carceller F, Dujovny M, Garcia-Gomez I, Gonzalez Corrochano R, Diaz-Gonzalez D (2002) Peripheral nerve regeneration by bone marrow stromal cells. Neurol Res 24(7):634
Cuevas P, Carceller F, Garcia-Gomez I, Yan M, Dujovny M (2004) Bonemarrow stromal cell implantation for peripheral nerve repair. Neurol Res 26(2):230–232
Flores AJ, Lavernia CJ, Owens PW (2000) Anatomy and physiology of peripheral nerve injury repair. Am J Orthop 29(3):167–173
Fouad K, Schnell L, Bunge MB, Schwab ME, Liebscher T, Pearse DD (2005) Combining Schwann cell bridges and olfactory-ensheathing glia grafts with chondroitinase promotes locomotor recovery after complete transection of the spinal cord. J Neurosci 25(5):1169–1178
Graham JB, Neubauer D, Xue QS, Muir D (2007) Chondroitinase applied to peripheral nerve repair averts retrograde axonal regeneration. Exp Neurol 203(1):185–195
Hattori T, Matsuyama Y, Sakai Y, Ishiguro N, Hirata H, Nakamura R (2008) Chondrotinase ABC enhances axonal regeneration across nerve gaps. J Clin Neurosci 15(2):185–191
Krekoski CA, Neubauer D, Zuo J, Muir D (2001) Axonal regeneration into acellular nerve grafts is enhanced by degradation of chondroitin sulfate proteoglycan. J Neurosci 21(16):6206–6213
Kwok JC, Afshari F, García-Alías G, Fawcett JW (2008) Proteoglycans in the central nervous system: plasticity, regeneration and their stimulation with chondroitinase ABC. Restor Neurol Neurosci 26(2–3):131–145
Lin YC, Ramadan M, Hronik-Tupaj M, Kaplan DL, Philips BJ, Sivak W, Rubin JP, Marra KG (2011) Spatially controlled delivery of neurotrophic factors in silk fibroin-based nerve conduits for peripheral nerve repair. Ann Plast Surg 67(2):147–155
Liu CHJ, Sun JZH, Tong XJ, Zhang CSH, Cao DSH (2003) The preparation of scaffolds from natural nerve by the method of tissue engineering. Prog Anat Sci 9:197–200 (in Chinese)
Millesi H, Terzis JK (1984) Nomenclature in peripheral nerve surgery. Committee report of the International Society of Reconstructive Microsurgery. Clin Plast Surg 11(1):3–8
Mimura T, Dezawa M, Kanno H, Sawada H, Yamamoto I (2004) Peripheral nerve regeneration by transplantation of bone marrow stromal cell-derived Schwann cells in adult rats. J Neurosurg 101(5):806–812
Mohammadi R, Azizi S, Delirezh N, Hobbenaghi R, Amini K (2011) Comparison of beneficial effects of undifferentiated cultured bone marrow stromal cells and omental adipose-derived nucleated cell fractions on sciatic nerve regeneration. Muscle Nerve 43(2):157–163
Neubauer D, Graham JB, Muir D (2007) Chondroitinase treatment increases the effective length of acellular nerve grafts. Exp Neurol 207(1):163–170
Olmarker K, Strömberg J, Blomquist J, Zachrisson P, Nannmark U, Nordborg C, Rydevik B (1996) Chondroitinase ABC (pharmaceutical grade) for chemonucleolysis. Functional and structural evaluation after local application on intraspinal nerve structures and blood vessels. Spine 21(17):1952–1956
Pestronk A, Drachman DB (1978) A new stain for quantitative measurement of sprouting at neuromuscular junctions. Muscle Nerve 1(1):70–74
Silver J, Miller JH (2004) Regeneration beyond the glial scar. Nat Rev Neurosci 5(2):146–156
Sun XH, Che YQ, Tong XJ, Zhang LX, Feng Y, Xu AH, Tong L, Jia H, Zhang X (2009) Improving nerve regeneration of acellular nerve allografts seeded with SCs bridging the sciatic nerve defects of rat. Cell Mol Neurobiol 29(3):347–353
Tao Li J, Somasundaram C, Bian K, Xiong W, Mahmooduddin F, Nath RK, Murad F (2010) Nitric oxide signaling and neural stem cell differentiation in peripheral nerve regeneration. Eplasty 10:e42
Tohill M, Terenghi G (2004) Stem-cell plasticity and therapy for injuries of the peripheral nervous system. Biotechnol Appl Biochem 40(1):17–24
Tong XJ, Liu CHJ, Zhang CSH, Cao DSH, Yu P (2004) Experimental study of the repairing effect of acellular nerve allografts on the sciatic nerve gap of rat. Acta Anat Sin 35(3):230–233 (in Chinese)
Wood MD, MacEwan MR, French AR, Moore AM, Hunter DA, Mackinnon SE, Moran DW, Borschel GH, Sakiyama-Elbert SE (2010) Fibrin matrices with affinity-based delivery systems and neurotrophic factors promote functional nerve regeneration. Biotechnol Bioeng 106(6):970–979
Zhang C, Yao C, He XJ, Li HP (2010) Repair of subacute spinal cord crush injury by bone marrow stromal cell transplantation and chondroitinase ABC microinjection in adult rats. Nan Fang Yi Ke Da Xue Xue Bao 30(9):2030–2035 (in Chinese)
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.

10571_2011_9764_MOESM1_ESM.jpg
Sup Fig. 1 Detection of motor end plate in tibial muscles. Eight weeks after surgery, the tibial muscles in groups I–IV (A–D) were subjected to ACE staining. The quantitative analysis of IOD was shown in (E). Shown were representative images (n = 6). Bar: 50 μm. (JPEG 3632 kb)

10571_2011_9764_MOESM2_ESM.jpg
Sup Fig. 2 Detection of HRP retrograde tracing. Eight weeks after surgery, positive sensory neurons in L3–6 ganglio from groups I to IV (A–D) were labeled brown. The quantitative analysis of IOD was shown in (E). Shown were representative images (n = 6). Bar: 20 μm. (JPEG 4460 kb)

10571_2011_9764_MOESM3_ESM.jpg
Sup Fig. 3 Immunohistochemical staining of VEGF and CD34 in ARSN in different groups. Eight weeks after surgery, ARSN in groups I–IV was subjected to IHC with antibody against VEGF (A–D, groups I–IV, respectively) and CD34 (E–H, groups I–IV, respectively). The quantitative analysis of IOD was shown in (I). *P < 0.05 vs. group I (n = 6). Bar: 50 μm. (JPEG 1068 kb)

10571_2011_9764_MOESM4_ESM.jpg
Sup Fig. 4 Immunohistochemical staining of NGF and BDNF in ARSN in different groups. Eight weeks after surgery, ARSN in groups I–IV was subjected to IHC with antibody against NGF (A–D, group I–IV, respectively) and BDNF (E–H, group I–IV, respectively). The quantitative analysis of IOD was shown in (I). *P < 0.05 vs. groupI(n = 6). Bar: 50 μm. (JPEG 1044 kb)
Rights and permissions
About this article
Cite this article
Wang, Y., Jia, H., Li, WY. et al. Synergistic Effects of Bone Mesenchymal Stem Cells and Chondroitinase ABC on Nerve Regeneration After Acellular Nerve Allograft in Rats. Cell Mol Neurobiol 32, 361–371 (2012). https://doi.org/10.1007/s10571-011-9764-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-011-9764-4