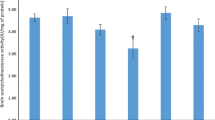
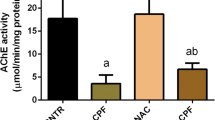
Abstract
Organotellurium compounds have been synthesized since 1840, but pharmacological and toxicological studies about them are still incipient. Therefore, the objective of this study was to verify the effect of acute administration of the organochalcogen 3-butyl-1-phenyl-2-(phenyltelluro)oct-en-1-one on some parameters of oxidative stress in the brain of 30-day-old rats. Animals were treated intraperitoneally with a single dose of the organotellurium (125, 250, or 500 μg/kg body weight) and sacrificed 60 min after the injection. The cerebral cortex, the hippocampus, and the cerebellum were dissected and homogenized in KCl. Afterward, thiobarbituric acid reactive substances (TBARS), carbonyl, sulfhydryl, catalase (CAT), superoxide dismutase (SOD), nitric oxide (NO) formation, and hydroxyl radical production were measured in the brain. The organotellurium enhanced TBARS in the cerebral cortex and the hippocampus, and increased protein damage (carbonyl) in the cerebral cortex and the cerebellum. In contrast, the compound provoked a reduced loss of thiol groups measured by the sulfhydryl assay in all the tissues studied. Furthermore, the activity of the antioxidant enzyme CAT was reduced by the organochalcogen in the cerebral cortex and the cerebellum, and the activity of SOD was inhibited in all the brain tissues. Moreover, NO production was increased in the cerebral cortex and the cerebellum by this organochalcogen, and hydroxyl radical formation was also enhanced in the cerebral cortex. Our findings indicate that this organotellurium compound induces oxidative stress in the brain of rats, corroborating that this tissue is a potential target for organochalcogen action.




Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Meth Enzymol 105:121–126
Aksenov MY, Markesbery WR (2001) Change in thiol content and expression of glutathione redox system gene in the hippocampus and cerebellum in Alzheimer’s disease. Neurosci Lett 302:141–145
Alho H, Leinonen JS, Erhola M, Lonnrot K, Acjmelacus R (1998) Assay of antioxidant capacity of human plasma and CSF in aging and disease. Restor Neurol Neurosci 12:159–165
Ávila DS, Beque MC, Folemer V, Braga AL, Zeni G, Nogueira CW, Soares FAA, Rocha JBT (2006) Diethyl 2-phenyl-2 tellurophenyl vinylphosphonate: an organotellurium compound with low toxicity. Toxicology 224:100–107
Ávila DS, Gubert P, Corte CLD, Alves D, Nogueira CW, Rocha JBT, Soares FAA (2007) A biochemical and toxicological study with 2-phenyl-2 tellurophenyl vinylphosphonate in sub- intraperitonial treatment in mice. Life Sci 80:1865–1872
Behl C, Moosmann B (2002) Oxidative nerve cell death in Alzheimer’s disease and stroke: antioxidants as neuroprotective compounds. Biol Chem 383:521–536
Berg D, Youdim MB (2006) Role of iron in neurodegenerative disorders. Top Magn Reson Imaging 17:5–17
Bogdanov MB, Andreassen OA, Dedeoglu A, Ferrante RJ, Beal MF (2001) Increased oxidative damage to DNA in a transgenic mouse of Huntington’s disease. J Neurochem 79:1246–1249
Borges VC, Rocha JBT, Nogueira CW (2005) Effect of diphenyl diselenide, diphenyl ditelluride and ebselen on cerebral Na + , K + ATPase activity in rats. Toxicology 215:191–197
Buege JA, Aust SD (1978) Microsomal Lipid Peroxidation. Meth Enzymol 52:302–309
Carvalho CAS, Gemelli T, Guerra RB, Oliboni L, Salvador M, Dani C, Araújo AS, Mascarenhas M, Funchal C (2009) Effect of in vitro exposure of human serum to 3-butyl-1-phenyl-2-(phenyltelluro)oct-en-1-one on oxidative stress. Mol Cell Biochem 332:127–134
Chen F, Vallyathan V, Castranova V, Shi X (2001) Cell apoptosis induced carcinogenic metals. Mol Cel Biochem 221:183–188
Comasseto JV, Ling LW, Petragnani N, Stefani HA (1997) Vinylic selenides and tellurides-preparation, reactivity and synthetic application. Synthesis 4:373–376
Engman L, Kanda T, Gallegos A, Williams R, Powis G (2000) Water soluble organotellurium compounds inhibit thioredoxin reductase and the growth of human cancer cells. Anti-Cancer Drug Des 15:323–330
Evelson P, Travacio M, Repetto M, Escobar J, Llesuy S, Lissi E (2001) Evaluation of total reactive antioxidant potential (TRAP) of tissue homogenates and their cytosols. Arch Biochem Biophys 388:261–266
Funchal C, Moretto MB, Vivian L, Zeni G, Rocha JBT, Pessoa-Pureur R (2006a) Diphenyl ditelluride-and methylmercury-induced hyperphosphorylation of the high molecular weight neurofilament subunit is prevented by organoslenium compounds in cerebral cortex of young rats. Toxicology 222:143–153
Funchal C, Latini A, Jacques-Silva MC, Santos AQ, Buzin L, Gottfried C, Wajner M, Pessoa-Pureur R (2006b) Morphological alterations and induction of oxidative stress in glial cells caused by the branched-chain -keto acids accumulating in maple syrup urine disease. Neurochem Int 49:640–650
Halliwell B (2001) Role of free radicals in the neurodegenerative diseases: therapeutic implications for antioxidant treatment. Drugs Aging 18:685–716
Halliwell B (2006) Oxidative stress and neurodegeneration: where are we now? J Neurochem 97:1634–1658
Halliwell B, Gutteridge JM (1981) Formation of thiobarbituric-acid-reactive substance from deoxyribose in the presence of iron salts: the role of superoxide and hydroxyl radicals. FEBS Lett 128:347–352
Halliwell B, Gutteridge JMC (2007) Measurement of reactive species. In: Halliwell B, Gutteridge JMC (eds) Free Radicals in Biology and Medicine. Oxford University Press, Oxford, pp 268–340
Heffner JA, Repine JE (1989) State of the art: pulmonary strategies of antioxidant defense. Am Rev Respir Dis 140:531–554
Hevel JM, Marletta MA (1994) Nitric oxide synthase assays. Methods Enzymol 233:250–258
Ischiropoulos H, Zhu L, Chen J, Tsai M, Martin JC, Smith CD, Beckman JS (1992) Peroxynitrite-mediated tyrosine nitration catalyzed by superoxide dismutase. Arch Biochem Biophys 298:431–437
Iwase K, Tatsuishi T, Nishimura Y, Yamaguch J, Oyama Y, Miyoshi N, Wada M (2004) Cytometric analysis of adverse action of diphenyl ditelluride on rat thymocytes: cell shrinkage as a cytotoxic parameter. Environ Toxicol 19:614–661
Jaffe EK (1995) Porphobilinogen synthase, the first source of heme’s asymetry. J Bioenerg Biomembr 27:169–179
Kann O, Kovács R (2007) Mitochondria and neuronal activity. Am J Physiol Cell Physiol 292:C641–C657
Kaur P, Yousuf S, Ansari MA, Siddiqui A, Ahmad AS, Islam F (2003) Tellurium-induced dose-dependent impairment of antioxidant status: differential effects in cerebrum, cerebellum, and brainstem of mice. Biol Trace Elem Res 94:247–258
Laden BP, Porter TD (2001) Inhibition oh human squalene monooxigenase by tellurium compounds: evidence of interaction with vicinal sulfhydryls. J Lipid Res 42:235–240
Lehtinen MK, Bonni A (2006) Modeling oxidative stress in the central nervous system. Curr Mol Med 6:871–881
Lissi E, Salim-Hanna M, Pascual C, Del Castillo MD (1995) Evaluation of total antioxidant potential (TRAP) and total antioxidant reactivity from luminol-enhanced chemiluminescence measurements. Free Radic Biol Med 18:153–158
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–267
Maciel EN, Bolzan RC, Braga AL, Rocha JBT (2000) Diphenyl diselenide and diphenyl ditelluride differentially affect d-aminolevulinate dehydratase from liver, kidney and brain of mice. J Biochem Mol Toxicol 14:310–319
Marklund, S. (1985). Handbook of methods for oxygen radical research. Boca Rat. CRC. Press., pp. 243-247
Meotti FC, Borges VC, Zeni JBT, Nogueira CW (2003) Potential renal and hepatic toxicity of diphenyl diselenide, diphenyl ditelluride and ebselen for rats and mice. Toxicol Lett 143:9–16
Moretto MB, Funchal C, Zeni G, Rocha JBT, Pessoa-Pureur R (2005) Organoselenium compounds prevent hyperphosphorylation of cytoskeletal proteins induced by the neurotoxic agent diphenyl ditelluride in cerebral cortex of young rats. Toxicology 210:213–222
Nogueira CW, Rotta LN, Perry ML, Souza DO, Rocha JBT (2001) Diphenyl diselenide and diphenyl ditelluride affect the rat glutamatergic system ¨ın vitro and in vivo. Brain Res 906:157–163
Nogueira CW, Rotta LN, Zeni G, Souza DO, Rocha JBT (2002) Exposure to ebselen changes glutamate uptake and release by rat brain synaptossomes. Neurochem Res 27:283–288
Nogueira CW, Borges VC, Zeni G, Rocha JBT (2003) Organochalcogens effects on δ-aminolevilinate dehydratase activity from human erythrocytic cells in vitro. Toxicology 191:169–178
Nogueira CW, Zeni G, Rocha JBT (2004) Organoselenium and organotellurium compounds: pharmacology and toxicology. Chem Rev 104:6255–6286
Ogra Y, Kobayashi R, Ishiwata K, Suzuki KT (2008) Comparison of distribution and metabolism between tellurium and selenium in rats. J Inorg Biochem 102:1507–1513
Penz J, Gemelli T, Carvalho CAS, Guerra RB, Oliboni L, Salvador M, Dani C, Araújo AS, Funchal C (2009) Effect of 3-butyl-1-phenyl-2-(phenyltelluro)oct-en-1-one on oxidative stress in cerebral cortex of rats. Food Chem Toxicol 47:745–751
Petragnani N (1994) Tellurium in organic synthesis. Academic Press, New York
Reznick AZ, Packer L (1994) Carbonyl assay for determination of oxidatively modified proteins. Meth Enzymol 233:357–363
Rodriguez-Martin E, Casajeros MJ, Canals S, de Bernardo S, Mena MA (2002) Thiolic antioxidants protect from nitric oxide-induced toxicity in fetal midbrain cultures. Neuropharmacology 43:877–888
Rooseboom M, Vermeulen NPE, Durgut F, Commandeur JNM (2002) Comparative study on the bioactivation mechanisms and cytotoxicity of Te-Phenyl-L-tellurocysteine, Se-Phenyl-L-selenocysteine and S-Phenyl-L-cysteine. Chem Res Toxicol 15:1610–1618
Sagara JI, Miura K, Bannai S (1993) Maintenance of neuronal glutathione by glial cells. J Neurochem 61:1672–1676
Sailer BL, Liles N, Dickerson S, Chasteen TG (2003) Cytometric determination of novel organotellurium compound toxicity in a promyelocitic (HL-60) cell line. Arch Toxicol 77:30–36
Sailer BL, Liles N, Dickerson S, Sumners S, Chasteen TG (2004) Organotellurium compound toxicity in a promyelocytic cell line compared to non-tellurium-containing organic analog. Toxicol In Vitro 18:475–482
Savegnago L, Borges VC, Alves D, Jesse C, Rocha JBT, Nogueira CW (2006) Evaluation of antioxidant activity and potential toxicity of 1-buthyltelurenyl-2-methylthioheptene. Life Sci 79:1546–1552
Seifried DE, Anderson EI, Fisher JA, Milner A (2007) Review of the interaction among dietary antioxidants and reactive oxygen species. J Nutr Biochem 18:567–579
Sies H (1991) Oxidative stress: from basic research to clinical application. Am J Med 91:31S–38S
Silveira CC, Braga AL, Guerra RB (2002) Stereoselective synthesis of alpha-phenylchalcogeno-alpha, beta -unsaturated esters. Tetrahedron Lett 43:3395–3397
Stamler JS, Toone EJ (2002) The decomposition of thionitrites. Curr Opin Chem Biol 6:779–785
Stangherlin EC, Favero AM, Zeni G, Rocha JBT, Nogueira CW (2005) Teratogenic vulnerability of Wistar rats to diphenyl ditelluride. Toxicology 2:231–239
Stangherlin EC, Ardais AP, Rocha JBT, Nogueira CW (2009) Exposure to diphenyl ditelluride, via maternal milk, causes oxidative stress in cerebral cortex, hippocampus and striatum of young rats. Arch Toxicol 83(5):485–491
Stone R, Stewart VC, Hurst RD, Clark JB, Heales SJ (1999) Astrocyte nitric oxide causes neuronal mitochondrial damage, but antioxidant release limits neuronal cell death. Ann NY Acad Sci 893:400–403
Strayo D, Adhikari S, Tilak-Jain J, Menon VP, Devasagayam TPA (2008) Antioxidant activity of an aminothiazole compound: possible mechanisms. Chem Biol Interact 173:215–223
Tremaroli V, Fedi S, Zannoni D (2007) Evidence for a tellurite-dependent generation of reactive oxygen species and absence of a tellurite-mediated adaptive response to oxidative stress in cells of Pseudomonas pseudoalcaligenes KF707. Arch Microbiol 187:127–135
Valko M, Leibfritz D, Moncol J, Cronin MTD, Mazur M, Telser J (2007) Free radicals and antioxidants in normal physiological functions and human disease. Int J Biochem Cell Biol 39:44–84
Widy-Tysziewicz E, Piechal A, Gajkowska B, Smialek M (2002) Tellurium-induced cognitive deficits in rats are related to neuropathological changes in the central nervous system. Toxicol Lett 131:203–214
Yamada N, Kojima R, Uno M, Akiyama T, Kitaura H, Narumi K, Nishiuchi K (2002) Phase-change material for use in rewritable dual-layer optical disk. SPIE 4342:55–63
Yamakura F, Matsumoto T, Ikeda K, Taka H, Fujimura T, Murayama K, Watanabe E, Tamaki M, Imai T, Takamori K (2005) Nitrated and oxidized products of a single tryptophan residue in human Cu, Zn-superoxide dismutase treated with either peroxynitrite-carbon dioxide or myeloperoxidase-hydrogen peroxide-nitrite. J Biochem 138:57–69
Yeo WS, Lee SJ, Lee JR, Kim KP (2008) Nitrosative protein tyrosine modifications: biochemistry and functional significance. BMB Rep 31:194–203
Zeni G, Braga AL, Stefani HA (2003) Palladium-catalyzed coupling of sp2- hybrized tellurides. Acc Chem Res 36:718–731
Zeni G, Ludtke D, Panatieri RB, Braga AL (2006) Vinylic tellurides: from preparation to their applicability in organic synthesis. Chem Rev 106:1032–1076
Zugno AI, Stefanello FM, Scherer EBS, Mattos C, Pederzolli CD, Andrade VM, Wannmacher CMD, Wajner M, Dutra-Filho CS, Wyse ATS (2008) Guanidinoacetate decreases antioxidant defenses and total protein sulfhydryl content in striatum of rats. Neurochem Res 33:1804–1810
Acknowledgments
This study was supported by Centro Universitário Metodista IPA and Universidade de Caxias do Sul.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Funchal, C., Carvalho, C.A.S., Gemelli, T. et al. Effect of Acute Administration of 3-Butyl-1-Phenyl-2-(Phenyltelluro)Oct-En-1-One on Oxidative Stress in Cerebral Cortex, Hippocampus, and Cerebellum of Rats. Cell Mol Neurobiol 30, 1135–1142 (2010). https://doi.org/10.1007/s10571-010-9547-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-010-9547-3