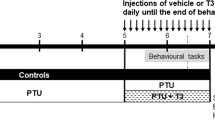
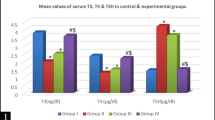
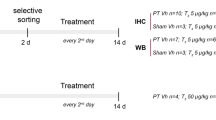
Thyroid hormones play important roles in brain function. However, few information is available about the effect of 3,5,3′-triiodo-l-thyronine (T3) or thyroxine (T4) on the in vitro phosphorylation of intermediate filament (IF) proteins from cerebral cortex of rats. In this study we investigated the involvement of GABAergic mechanisms mediating the effects of T3 and T4 on the in vitro incorporation of 32P into IF proteins from cerebral cortex of 10-day-old male rats. Tissue slices were incubated with or without T3, T4, γ-aminobutiric acid (GABA), kinase inhibitors or specific GABA antagonists and 32P-orthophosphate for 30 min. The IF-enriched cytoskeletal fraction was extracted in a high salt Triton-containing buffer and the in vitro 32P incorporation into IF proteins was measured. We first observed that 1 μM T3 and 0.1 μM T4 significantly increased the in vitro incorporation of 32P into the IF proteins studied through the PKA and PKCaMII activities. A similar effect on IF phosphorylation was achieved by incubating cortical slices with GABA. Furthermore, by using specific GABA antagonists, we verified that T3 induced a stimulatory effect on IF phosphorylation through noncompetitive mechanisms involving GABAA, beyond GABAB receptors. In contrast, T4 effects were mediated mainly by GABAB mechanisms. In conclusion, our results demonstrate a rapid nongenomic action of T3 and T4 on the phosphorylating system associated to the IF proteins in slices of cerebral cortex of 10 day-old male rats and point to GABAergic mechanisms mediating such effects.






Similar content being viewed by others
REFERENCES
Ackerley, S., Grierson, A. J., Brownlees, J., Thornhiel, P., Anderton, B. H., Leight, P. N., Shew, C. E., and Mielerc, C. J. (2000). Glutamate slow axonal transport of neurofilaments in transfected neurons. J. Cell Biol. 150:165–175.
Alberts, B., Johnson, A., Lewis, J., Raff, M., Roberts, K., and Walter, P. (2002). The cytoskeleton. In Alberts, B., Johnson, A., Lewis, J., Raff, M., Roberts, K., and Walter, P. (eds.), Molecular Biology of the Cell, Garland Science, New York, pp. 907–982.
Bernal, J. (2002). Action of thyroid hormone in brain. J. Endocrinol. Invest. 25:268–288.
Chapell, R., Martin, J., Machu, T. K., and Leidenheimer, N. J. (1998). Direct cannel-gating and modulatory effects of riiodothyronine on recombinant GABA (A) receptors. Eur. J. Pharmacol. 349:115–121.
Chen, Y., Chen, P. L., Chen, C. F., Sharp, Z. D., and Lee, W. T. (1999). Thyroid hormone, T3-dependent phosphorylation and translocation of Trip230 from the Golgi complex to the nucleus. Proc. Natl. Acad. Sci. U.S.A. 96:4443–4448.
Chijiwa, T., Mishima, A., Hagiwara, M., Sano, M., Hayashi, K., Inoue, T., Naito, K., Toshioka, T., and Hidaka, H. (1990). Inhibition of forskolin-induced neurite outgrowth and protein phosphorylation by a newly synthesized selective inhibitor of cyclic AMP-dependent protein kinase, N-[2-(p-bromocinnamylamino)ethyl]-5-isoquinolinesulfonamide (h-89), of PC12D pheochromocytoma cells. J. Biol. Chem. 265:5267–5272.
Davis, P. J., Shih, A., Lin, H. Y., Martino, L. J., and Davis, F. B. (2000). Thyroxine promotes association of mitogen-activated protein kinase and nuclear thyroid hormones receptor (TR) and causes serine phosphorylation of TR. J. Biol. Chem. 275:38032–38039.
Davis, P. J., Tillmann, H. C., Davis, F. B., and Wehling, M. (2002). Comparison of the mechanisms of nongenomic actions of thyroid hormone and steroid hormones. J. Endocrinol. Invest. 25:377–388.
Eng, L. F., Ghirnikar, R. S., and Lee, Y. L. (2000). Glial fibrillary acidic protein: GFAP-thirty-one years (1969–2000). Neurochem. Res. 25:1439–1451.
Freitas, M. S., de Mattos, A. G., Camargo, M. M., Wannmacher, C. M. D., and Pessoa-Pureur, R. (1995). Cytoskeletal-associated protein kinase and phosphatase activities from cerebral cortex of young rats. Neurochem. Res. 20:951–956.
Funchal, C., Vieira de Almeida, L. M., Oliveira Loureiro, S., Vivian, L., de Lima Pelaez, P., Dall Bello Pessutto, F., Rosa, A. M., Wajner, M., and Pessoa-Pureur, R. (2003). In vitro phosphorylation of cytoskeletal proteins from cerebral cortex of rats. Brain Res. Protein 11:111–118.
Gaiarsa, J.-L., Caillard, O., and Ben-Ari, Y. (2002). Long-term plasticity at GABAergic and glycinergic synapses: Mechanisms and functional significance. Trends Neurosci. 25:564–570.
Gotow, T., Tanaka, T., Nakamura, Y., and Takeda, M. (1994). Dephosphorylation of the largest neurofilament subunit protein influences the structure of crossbridges in reassembled neurofilaments. J. Cell Sci. 107:1949–1957.
Guidato, S., Tsai, L. H., Woodgett, J., and Miller, C. C. (1996). Differential cellular phosphorylation of neurofilament heavy side arms by glycogen synthase kinase-3 and cyclin-dependent kinase-5. J. Neurochem. 66:1698–1706.
Harrison, N. L. (1990). On the presynaptic action of baclofen at inhibitory synapses between cultured rat hippocampal neurones. J. Physiol. (Lond.) 422:433–446.
Hashimoto, R., Nakamura, Y., Goto, H., Wada, Y., Sakoda, S., and Kaibuchi, K. (1998). Domain- and site- specific phosphorylation of bovine NF-L by Rho-associated kinase. Biochem. Biophys. Res. Commun. 245:407–411.
Hashimoto, H., Walker, C. H., Prange, A. J. Jr., and Mason, G. A. (1991). The effects of thyroid hormones on potassium-stimulated release of 3H-GABA by synaptosomes of rat cerebral cortex. Neuropsychopharmacology 5:49–54.
Hisanaga, S., Yasugawa, S., Yamakawa, T., Miyamoto, E., Ikebe, M., Uchiyama, M., and Kishimoto, T. (1993). Dephosphorylation of microtubule-binding sites at the neurofilament-H tail domain by alkaline, acid, and protein phosphatases. J. Biochem. (Tokyo) 113:705–709.
Ho, W. H., Wang, S. M., and Yin, H. S. (2001). Regulation of the subcellular distribution and gene expression of GABA(A) receptor by microtubules and microfilaments in cultured brain neurons. J. Cell. Biochem. 83:291–303.
Hsieh, S. T., Kidd, G. J., Crawforg, T. O., Xu, Z., Lin, W. M., Trapp, B. D., Cleveland, D. W., and Griffin, J. W. (1994). Regional modulation of neurofilament organization by myelination in normal axons. J. Neurosci. 14:6392–6401.
Huang, C. J., Geller, H. M., Green, W. L., and Craelius, W. (1999). Acute effect of thyroid hormone analogs on sodium currents in neonatal rat myocytes. J. Mol. Cell. Cardiol. 31:881–893.
Inada, H., Goto, H., Tanabe, K., Nishi, Y., Kaibuchi, K., and Inagaki, M. (1998). Rho-associated kinase phosphorylates desmin, the myogenic intermediate filament protein, at unique amino-terminal sites. Biochem. Biophys. Res. Commun. 253:21–25.
Inagaki, M., Gonda, Y., Nishizawa, K., Kitamura, S., Sato, C., and Ando, S. (1990). Phosphorylation sites linked to glial filament disassembly in vitro located in a non-alpha-helical head domain. J. Biol. Chem. 265:4722–4729.
Inagaki, M., Nishi, Y., Nishizawa, K., Matsuyama, M., and Sato, C. (1987). Site-specific phosphorylation induces disassembly of vimentin filaments in vitro. Nature 328:649–652.
Incerpi, S., De Vito, P., Luly, P., Spagnuolo, S., and Leoni, S. (1999a). Short-term effect of tyroid hormones and 3,5-diiodothyronine on membrane transport systems in chick embryo hepatocytes. Endocrinology 143:1660–1668.
Incerpi, S., Luly, P., De Vito, P., and Farias, R. N. (1999b). Short-term effects of thyroid hormones on the Na/H antiport in L-6 myoblasts: high molecular specificity for 3,3′,5-triiodo-L-thyronine. Endocrinology 140:683–689.
Johnston, G. A. R. (1996). GABAC receptors: Relatively simple transmitter-gated ion channels? Trends Pharmacol. Sci. 17:319–323.
Komatsu, Y. (1996). GABAB receptors, monoamine receptors, and postsynaptic inositol triphopshate-induced Ca2+ release are involved in the induction of long-term potentiation at visual cortical inhibitory synapses. J. Neurosci. 16:6342–6352.
Ku, N. O., and Omary, M. B. (1997). Phosphorylation of human keratin 8 in vivo at conserved head domain serine 23 and at epidermal growth factor-stimulated tail domain serine 431. J. Biol. Chem. 272:7556–7564.
Laemmli, U. K. (1970). Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 277:680–685.
Lazar, M. A. (1993). Thyroid hormone receptors: Multiple forms, multiple possibilities. Endocrinol. Rev. 14:184–193.
Leterrier, J. F., Kas, J., Hartwig, J., Vegners, R., and Janmey, P. A. (1996). Mechanical effects of neurofilament cross-bridges. Modulation by phosphorylation, lipids, and interactions with F-actin. J. Biol. Chem. 271:15687–15694.
Lin, H.-Y., Davis, F. B., Gordinier, J. K., Martino, L. J., and Davis, P. J. (1999). Thyroid hormone induces activation of mitogen-activated protein kinase in cultured cells. Am. J. Physiol. 276:C1014–C1024.
Lin, H.-Y., Thacore, H. R., Davis, F. B., and Davis, P. J. (1996). Potentiation by tyroxine of interferon-gamma-induced antiviral state requires PKA and PKC activities. Am. J. Physiol. 271:C1256–C1261.
Lin, H.-Y., Yen, P. M., Davis, F. B., and Davis, P. J. (1997). Protein synthesis-dependent potentiation of thyroxine of antiviral activity of interferon-gamma. Am. J. Physiol. 273:C1225–1232.
Lowry, O. H., Rosebrough, N. J., Farr, A. L., and Randall, R. J. (1951). Protein measurement with the Folin phenol reagent. J. Biol. Chem. 193:265–267.
MacDonald, R. L., and Olsen, R. W. (1994). GABAA receptor channels. Annu. Rev. Neurosci. 17:569–602.
Martin, J. V., Williams, D. B., Fitzgerald, R. M., Im, H. K., and Vonvoigtlander, P. F. (1996). Thyroid hormonal modulation of the binding and activity of the GABAA receptor complex of brain. Neuroscience 73:705–713.
Mason, G. A., Walker, C. H., and Prange, A. J. Jr. (1987). Modulation of gamma-aminobutyric acid uptake of rat brain synaptosomes by thyroid hormones. Neuropsychopharmacology 1:63–70.
Narihara, R., Hirouchi, M., Ichida, T., Kuriyama, K., and Roberts, E. (1994). Effects of thyroxine and its related compounds on cerebral GABA receptors: Inhibitory action on benzodiazepine recognition site in GABAA receptor complex. Neurochem. Int. 25:451–454.
Nixon, R. A., and Sihag, R. K. (1991). Neurofilament phosphorylation: A new look at regulation and function. Trends Neurosci. 14:501–506.
Olsen, R. W., and DeLorey, T. M. (1999). GABA and glicine. In Siegel, G. J., Agranoff, B. W., Albers, R. W., Fisher, S. K., and Uhler, M. D. (eds.), Basic Neurochemistry, Lippincott-Raven, New York, pp. 335–346.
Paramio, J. M., and Jorcano, J. L. (2002). Beyond structure: Do intermediate filaments modulate cell signalling? Bioessays 24:836–844.
Paul, S., Gharami, K., Das, S., and Sarkar, P. K. (1999). Thyroid hormone-induced maturation of astrocytes is associated with the expression of new variants of vimentin and their phosphorylation. J. Neurochem. 73:1964–1972.
Peter, M., Sanghera, J. S., Pelech, S. S., and Nigg, E. A. (1992). Mitogen-activated protein kinases phosphorylate nuclear lamins and display sequence specificity overlapping that of mitotic protein kinase p34 cdc2. Eur. J. Biochem. 205:287–294.
Porter, J. T., and McCarthy, K. D. (1997). Astrocytic neurotransmitter receptors in situ and in vivo. Prog. Neurobiol. 51:439–455.
Rosewater, K., and Sontheimer, H. (1994). Fibrous and protoplasmic astrocytes express GABAA receptors that differ in benzodiazepine pharmacology. Brain Res. 636:73–80.
Runquist, M., and Alonso, G. (2003). GABAergic signaling mediates the morphological organization of astrocytes in the adult rat forebrain. Glia 41:137–151.
Safran, M., Farwell, A. P., and Leonard, J. L. (1992). Thyroid hormone-dependent redistribution of the 55-kilodalton monomer of protein disulfide isomerase in cultured glial cells. Endocrinology 131:2413–2418.
Sandrini, M., Marrama, D., Vergoni, A. V., and Bertolini, A. (1991). Effects of thyroid status on the characteristics of alpha 1-, alpha 2-, beta, imipramine and GABA receptors in the rat brain. Life Sci. 48:659–666.
Segal, J. (1989). Action of the thyroid hormone at the level of the plasma membrane. Endocrinol. Res. 15:619–649.
Shetty, K. T., Kink, W. T., and Pant, H. C. (1993). Cdc-like kinase from rat spinal cord specifically phosphorylates KSPXK motifs in neurofilament proteins: Isolation and characterization. Proc. Natl. Acad. Sci. U.S.A. 90:6844–6848.
Siegrist-Kaiser, C., Juge-Aubry, C., Tranter, M., Ekenbarger, D., and Leonard, J. (1990). Thyroxine-dependent modulation of actin polymerization in cultured astrocytes. A novel, extranuclear action of thyroid hormone. J. Biol. Chem. 265:5296–5302.
Sihag, R. K., and Nixon, R. A. (1990). Phosphorylation of the amino terminal head domain of the middle molecular mass 145 kDa subunit of neurofilaments. J. Biol. Chem. 265:4166–4171.
Silva, F. R., Leite, L. D., Barreto, K. P., D’Agostini, C., and Zamoner, A. (2001). Effect of 3,5,5′-triiodo-L-thyronine on amino acid accumulation and membrane potential in Sertoli cells of the rat testis. Life Sci. 69:977–986.
Silva, F. R. M. B., Leite, L. D., and Wassermann, G. F. (2002). Rapid signal transduction in Sertoli cells. Eur. J. Endocrinol. 147:425–433.
Sun, D., Leung, C. L., and Liem, R. K. H. (1996). Phosphorylation of the high molecular weight neurofilament protein (NF-H) by Cdk5 and p35. J. Biol. Chem. 271:14245–14251.
Tanaka, J., Ogawara, M., Ando, S., Shibata, M., Yatani, R., and Kusagawa, M. (1993). Phosphorylation of a 62 kd porcine alpha-internexin, a newly identified intermediate filament protein. Biochem. Biophys. Res. Commun. 196:115–123.
Tokumitsu, H., Chijiwa, T., Hagiwara, M., Mizutani, A., Terasawa, M., and Hidaka, H. (1990). Kn62,1-N,O-Bis-[(5-isoquino-linesulfonyl)-N-methy-(-tyrisyl)]-4-phenylpiperazine, a specific inhibitor of Ca2+/calmodulin-dependent protein kinase II. J. Biol. Chem. 265:5315–5320.
Volpato, K. C., Menegaz, D., Leite, L. D., Barreto, K. P., Garcia, E. V., and Silva, F. R. M. B. (2004). Involvement of K+ channels and calcium-dependent pathways in the action of T3 on amino acid accumulation and membrane potential in Sertoli cells of immature rat testis. Life Sci. 74:1277–1288.
Wang, H., and Olsen, R. W. (2000). Binding of the GABA(A) receptor-associated protein (GABARAP) to microtubules and microfilaments suggests involvement of the cytoskeleton in GABARAPGABA(A) receptor interaction. J. Neurochem. 75:644–655.
Zamoner, A., Frasson Corbelini, P., Funchal, C., Menegaz, D., Mena Barreto Silva, F. R., and Pessoa Pureur, R. (2005). Involvement of calcium-dependent mechanisms on the action of T3 in the in vitro phosphorylation of vimentin of immature rat testis. Life Sci. 77:3321–3335.
Zhu, X. G., Hanover, J., Hager, G, and Cheng, S. Y. (1998). Hormone-induced translocation of thyroid hormone receptors in living cells visualized under a receptor green fluorescent protein chimera. J. Biol. Chem. 273:27058–27063.
ACKNOWLEDGMENTS
This work was supported by Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq), Fundação de Amparo à Pesquisa do Estado do Rio Grande do Sul (FAPERGS), and PROPESq-UFRGS.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zamoner, A., Funchal, C., Heimfarth, L. et al. Short-Term Effects of Thyroid Hormones on Cytoskeletal Proteins Are Mediated by GABAergic Mechanisms in Slices of Cerebral Cortex from Young Rats. Cell Mol Neurobiol 26, 209–224 (2006). https://doi.org/10.1007/s10571-006-9027-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10571-006-9027-y