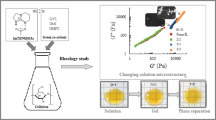
Abstract
The study proposes using the distillable ionic liquid 5-methyl-1,5,7-triaza-bicyclo-[4.3.0]non-6-enium acetate, [mTBNH][OAc], for cellulose dissolution, making it an environmentally friendly alternative to conventional solvents in the transesterification of cellulose with vinyl esters. This ionic liquid (IL) has high dissolving power for cellulose and durability for recycling. However, its high viscosity limits cellulose concentration and its expensiveness hinders commercialization. The addition of naturally derived, low-cost, and low-viscous co-solvents can reduce overall cost and viscosity. In this study, various green co-solvents, including γ-valerolactone (GVL), dimethyl isosorbide (DMI), sulfolane (SLF), and N,N′-dimethylpropyleneurea (DMPU), were mixed with [mTBNH][OAc] to test their ability to enhance cellulose transesterification. Cellulose esters with a degree of substitution (DS) up to 1.6 have been synthesized. The chemical composition of the materials was confirmed by FTIR and NMR. Green co-solvents alter the solubility and flow activation energy of cellulose in binary solvents. The produced cellulose esters become more amorphous, and their viscosity and complex moduli decrease because of the DS changing in the following order: DMPU < SLF < DMI < GVL. The efficiency of internal plasticization of cellulose esters was studied through melt flow rheology, which indicated that it increases with increasing DS. All tested cellulose esters have almost identical degradation temperatures, as detected by TGA.












Similar content being viewed by others
Data availability
No datasets were generated or analysed during the current study.
References
Asakawa M, Shrotri A, Kobayashi H, Fukuoka A (2019) Solvent basicity controlled deformylation for the formation of furfural from glucose and fructose. Green Chem 21(22):6146–6153. https://doi.org/10.1039/C9GC02600B
Asim N, Badiei M, Mohammad M (2022) Recent advances in cellulose-based hydrophobic food packaging. Emergent Mater 5(3):703–718. https://doi.org/10.1007/s42247-021-00314-2
Ass BAP, Ciacco GT, Frollini E (2006) Cellulose acetates from linters and sisal: Correlation between synthesis conditions in DMAc/LiCl and product properties. Biores Technol 97(14):1696–1702. https://doi.org/10.1016/j.biortech.2005.10.009
Barnes HA, Hutton JF, Walters K (1989) An Introduction to Rheology. Elsevier
Bering E, Torstensen JØ, Lervik A, de Wijn AS (2022) Computational study of the dissolution of cellulose into single chains: the role of the solvent and agitation. Cellulose 29(3):1365–1380. https://doi.org/10.1007/s10570-021-04382-9
Bistany KL, Kokini JL (1983) Dynamic Viscoelastic Properties of Foods in Texture Control. J Rheol 27(6):605–620. https://doi.org/10.1122/1.549732
Brandrup J, Immergut EH, Grulke EA (1999) Polymer handbook, 4th edn. Wiley
Bryan MC, Dunn PJ, Entwistle D, Gallou F, Koenig SG, Hayler JD, Hickey MR, Hughes S, Kopach ME, Moine G, Richardson P, Roschangar F, Steven A, Weiberth FJ (2018) Key Green Chemistry research areas from a pharmaceutical manufacturers’ perspective revisited. Green Chem 20(22):5082–5103. https://doi.org/10.1039/C8GC01276H
Budtova T, Navard P (2015) Viscosity-temperature dependence and activation energy of cellulose solutions. Nord Pulp Paper Res J 30(1):99–104. https://doi.org/10.3183/npprj-2015-30-01-p099-104
Cai J, Zhang L (2005) Rapid Dissolution of Cellulose in LiOH/Urea and NaOH/Urea Aqueous Solutions. Macromol Biosci 5(6):539–548. https://doi.org/10.1002/mabi.200400222
Ciacco GT (2003) Application of the solvent dimethyl sulfoxide/tetrabutyl-ammonium fluoride trihydrate as reaction medium for the homogeneous acylation of Sisal cellulose. Cellulose 10(2):125–132. https://doi.org/10.1023/A:1024064018664
Constable DJC, Dunn PJ, Hayler JD, Humphrey GR, Leazer JL Jr, Linderman RJ, Lorenz K, Manley J, Pearlman BA, Wells A, Zaks A, Zhang TY (2007) Key green chemistry research areas—a perspective from pharmaceutical manufacturers. Green Chem 9(5):411–420. https://doi.org/10.1039/B703488C
Crépy L, Chaveriat L, Banoub J, Martin P, Joly N (2009) Synthesis of cellulose fatty esters as plastics-influence of the degree of substitution and the fatty chain length on mechanical properties. ChemSusChem 2(2):165–170. https://doi.org/10.1002/cssc.200800171
Duchatel-Crépy L, Joly N, Martin P, Marin A, Tahon JF, Lefebvre JM, Gaucher V (2020) Substitution degree and fatty chain length influence on structure and properties of fatty acid cellulose esters. Carbohydr Polym 234:1. https://doi.org/10.1016/j.carbpol.2020.115912
Duereh A, Sato Y, Smith RL, Inomata H (2015) Replacement of Hazardous Chemicals Used in Engineering Plastics with Safe and Renewable Hydrogen-Bond Donor and Acceptor Solvent-Pair Mixtures. ACS Sustain Chem Eng 3(8):1881–1889. https://doi.org/10.1021/acssuschemeng.5b00474
Feng L, Chen Z (2008) Research progress on dissolution and functional modification of cellulose in ionic liquids. J Mol Liq 142(1–3):1–5. https://doi.org/10.1016/j.molliq.2008.06.007
Fink H-P, Weigel P, Purz HJ, Ganster J (2001) Structure formation of regenerated cellulose materials from NMMO-solutions. Prog Polym Sci 26(9):1473–1524. https://doi.org/10.1016/S0079-6700(01)00025-9
French AD (2014) Idealized powder diffraction patterns for cellulose polymorphs. Cellulose 21(2):885–896. https://doi.org/10.1007/s10570-013-0030-4
Fundador NGV, Enomoto-Rogers Y, Takemura A, Iwata T (2012) Syntheses and characterization of xylan esters. Polymer 53(18):3885–3893. https://doi.org/10.1016/J.POLYMER.2012.06.038
Gericke M, Schlufter K, Liebert T, Heinze T, Budtova T (2009) Rheological properties of cellulose/ionic liquid solutions: From dilute to concentrated states. Biomacromolecules 10(5):1188–1194. https://doi.org/10.1021/bm801430x
Ghasemi M (2018) Fundamental Understanding of Cellulose Dissolution Can Improve the Efficiency of Biomass Processing. Agric Res Technol: Open Access J 16(2):1–5. https://doi.org/10.19080/artoaj.2018.15.555985
Gleissle W, Hochstein B (2003) Validity of the Cox–Merz rule for concentrated suspensions. J Rheol 47(4):897–910. https://doi.org/10.1122/1.1574020
Gören A, Mendes J, Rodrigues HM, Sousa RE, Oliveira J, Hilliou L, Costa CM, Silva MM, Lanceros-Méndez S (2016) High performance screen-printed electrodes prepared by a green solvent approach for lithium-ion batteries. J Power Sources 334:65–77. https://doi.org/10.1016/j.jpowsour.2016.10.019
Han S, Li J, Zhu S, Chen R, Wu Y, Zhang X, Yu Z (2009) Potential applications of ionic liquids in wood related industries. BioResources 4(2):825–834. https://doi.org/10.15376/biores.4.2.825-834
Hansen CM (2007) Hansen Solubility Parameters. CRC Press. https://doi.org/10.1201/9781420006834
Hawkins JE, Liang Y, Ries ME, Hine PJ (2021) Time temperature superposition of the dissolution of cellulose fibres by the ionic liquid 1-ethyl-3-methylimidazolium acetate with cosolvent dimethyl sulfoxide. Carbohydr Polym Technol Appl 2:100021. https://doi.org/10.1016/j.carpta.2020.100021
Heinze T, Liebert T (2001) Unconventional methods in celulose functionalization. Prog Polym Sci 26:1689–1762. https://doi.org/10.1016/S0079-6700(01)00022-3
Henderson RK, Jiménez-González C, Constable DJC, Alston SR, Inglis GGA, Fisher G, Sherwood J, Binks SP, Curzons AD (2011) Expanding GSK’s solvent selection guide – embedding sustainability into solvent selection starting at medicinal chemistry. Green Chem 13(4):854. https://doi.org/10.1039/c0gc00918k
Ilyin SO, Makarova VV, Polyakova MY, Kulichikhin VG (2020) Phase behavior and rheology of miscible and immiscible blends of linear and hyperbranched siloxane macromolecules. Mater Today Commun 22:100833. https://doi.org/10.1016/j.mtcomm.2019.100833
Katsuhara S, Sunagawa N, Igarashi K, Takeuchi Y, Takahashi K, Yamamoto T, Li F, Tajima K, Isono T, Satoh T (2023) Effect of degree of substitution on the microphase separation and mechanical properties of cellooligosaccharide acetate-based elastomers. Carbohydr Polym 316:120976. https://doi.org/10.1016/j.carbpol.2023.120976
Köhler S, Liebert T, Schöbitz M, Schaller J, Meister F, Günther W, Heinze T (2007) Interactions of Ionic Liquids with Polysaccharides 1. Unexpected Acetylation of Cellulose with 1-Ethyl-3-methylimidazolium Acetate. Macromol Rapid Commun 28(24):2311–2317. https://doi.org/10.1002/marc.200700529
Kostag M, Gericke M, Heinze T, El Seoud OA (2019) Twenty-five years of cellulose chemistry: innovations in the dissolution of the biopolymer and its transformation into esters and ethers. Cellulose 26(1):139–184. https://doi.org/10.1007/s10570-018-2198-0
Kramar A, Rodríguez Ortega I, González-Gaitano G, González-Benito J (2023) Solution casting of cellulose acetate films: influence of surface substrate and humidity on wettability, morphology and optical properties. Cellulose 30(4):2037–2052. https://doi.org/10.1007/s10570-022-05026-2
Lefroy KS, Murray BS, Ries ME (2021) Rheological and NMR Studies of Cellulose Dissolution in the Ionic Liquid BmimAc. J Phys Chem B 125(29):8205–8218. https://doi.org/10.1021/acs.jpcb.1c02848
Lowman DW (1998) Characterization of cellulose esters by solution-state and solid-state NMR spectroscopy. ACS Symp Ser 688(10):131–162. https://doi.org/10.1021/bk-1998-0688.ch010
Malkin A (1994) Rheology Fundamentals. In Fundamental Topics in Rheology. ChemTec Publishing 153–165
Marrucci G (1996) Dynamics of entanglements: A nonlinear model consistent with the Cox-Merz rule. J Non-Newtonian Fluid Mech 62(2–3):279–289. https://doi.org/10.1016/0377-0257(95)01407-1
Martins MAR, Sosa FHB, Kilpeläinen I, Coutinho JAP (2022) Physico-chemical characterization of aqueous solutions of superbase ionic liquids with cellulose dissolution capability. Fluid Phase Equilib 556:113414. https://doi.org/10.1016/j.fluid.2022.113414
Mead DW (2011) Analytic derivation of the Cox-Merz rule using the MLD “toy” model for polydisperse linear polymers. Rheol Acta 50(9–10):837–866. https://doi.org/10.1007/s00397-011-0550-5
Mohan M, Banerjee T, Goud VV (2016) Effect of Protic and Aprotic Solvents on the Mechanism of Cellulose Dissolution in Ionic Liquids: A Combined Molecular Dynamics and Experimental Insight. ChemistrySelect 1(15):4823–4832. https://doi.org/10.1002/slct.201601094
Ostonen A, Bervas J, Uusi-Kyyny P, Alopaeus V, Zaitsau DH, Emelyanenko VN, Schick C, King AWT, Helminen J, Kilpeläinen I, Khachatrian AA, Varfolomeev MA, Verevkin SP (2016) Experimental and Theoretical Thermodynamic Study of Distillable Ionic Liquid 1,5-Diazabicyclo[4.3.0]non-5-enium Acetate. Ind Eng Chem Res 55(39):10445–10454. https://doi.org/10.1021/acs.iecr.6b02417
Parviainen A, Wahlström R, Liimatainen U, Liitiä T, Rovio S, Helminen JKJ, Hyväkkö U, King AWT, Suurnäkki A, Kilpeläinen I (2015) Sustainability of cellulose dissolution and regeneration in 1,5-diazabicyclo[4.3.0]non-5-enium acetate: a batch simulation of the IONCELL-F process. RSC Adv 5(85):69728–69737. https://doi.org/10.1039/C5RA12386K
Phadagi R, Singh S, Hashemi H, Kaya S, Venkatesu P, Ramjugernath D, Ebenso EE, Bahadur I (2021) Understanding the role of Dimethylformamide as co-solvents in the dissolution of cellulose in ionic liquids: Experimental and theoretical approach. J Mol Liq 328:115392. https://doi.org/10.1016/j.molliq.2021.115392
Pinkert A, Marsh KN, Pang S, Staiger MP (2009) Ionic liquids and their interaction with cellulose. Chem Rev 109(12):6712–6728. https://doi.org/10.1021/cr9001947
Qiu X, Hu S (2013) “Smart” materials based on cellulose: A review of the preparations, properties, and applications. Materials 6(3):738–781. https://doi.org/10.3390/ma6030738
Quintana SE, Machacon D, Marsiglia RM, Torregroza E, Garcia-Zapateiro LA (2018) Steady and shear dynamic rheological properties of squash (Cucurbita moschata) pulp. Contemp Eng Sci 11(21):1013–1024. https://doi.org/10.12988/ces.2018.8386
Rasool MA, Vankelecom IFJ (2021) γ-Valerolactone as Bio-Based Solvent for Nanofiltration Membrane Preparation. Membranes 11(6):418. https://doi.org/10.3390/membranes11060418
Rinaldi R (2011) Instantaneous dissolution of cellulose in organic electrolyte solutions. Chem Commun 47(1):511–513. https://doi.org/10.1039/C0CC02421J
Russo F, Galiano F, Pedace F, Aricò F, Figoli A (2020) Dimethyl Isosorbide As a Green Solvent for Sustainable Ultrafiltration and Microfiltration Membrane Preparation. ACS Sustain Chem Eng 8(1):659–668. https://doi.org/10.1021/acssuschemeng.9b06496
Shafiei-Sabet S, Hamad WY, Hatzikiriakos SG (2012) Rheology of nanocrystalline cellulose aqueous suspensions. Langmuir 28(49):17124–17133. https://doi.org/10.1021/la303380v
Strappaveccia G, Luciani L, Bartollini E, Marrocchi A, Pizzo F, Vaccaro L (2015) γ-Valerolactone as an alternative biomass-derived medium for the Sonogashira reaction. Green Chem 17(2):1071–1076. https://doi.org/10.1039/C4GC01728E
Tarasova E, Savale N, Krasnou I, Kudrjašova M, Rjabovs V, Reile I, Vares L, Kallakas H, Kers J, Krumme A (2023) Preparation of Thermoplastic Cellulose Esters in [mTBNH][OAC] Ionic Liquid by Transesterification Reaction. Polymers 15(19):3979. https://doi.org/10.3390/polym15193979
Tarasova E, Savale N, Ausmaa P-M, Krasnou I, Krumme A (2024) Rheology and dissolution capacity of cellulose in novel [mTBNH][OAc] ionic liquid mixed with green co-solvents. Rheol Acta. https://doi.org/10.1007/s00397-024-01433-3
Tilstam U (2012) Sulfolane: A Versatile Dipolar Aprotic Solvent. Org Process Res Dev 16(7):1273–1278. https://doi.org/10.1021/op300108w
Wen X, Wang H, Wei Y, Wang X, Liu C (2017) Preparation and characterization of cellulose laurate ester by catalyzed transesterification. Carbohydr Polym 168:247–254. https://doi.org/10.1016/j.carbpol.2017.03.074
Wenzel RN (1936) Resistance of solid surfaces to wetting by water. Ind Eng Chem 28(8):988–994. https://doi.org/10.1021/ie50320a024
Willberg-Keyriläinen P, Vartiainen J, Harlin A, Ropponen J (2017) The effect of side-chain length of cellulose fatty acid esters on their thermal, barrier and mechanical properties. Cellulose 24(2):505–517. https://doi.org/10.1007/s10570-016-1165-x
Wilson K, Murray J, Sneddon H, Jamieson C, Watson A (2018) Dimethylisosorbide (DMI) as a Bio-Derived Solvent for Pd-Catalyzed Cross-Coupling Reactions. Synlett 29(17):2293–2297. https://doi.org/10.1055/s-0037-1611054
Wu S, Qin X, Li M (2014) The structure and properties of cellulose acetate materials: A comparative study on electrospun membranes and casted films. J Ind Text 44(1):85–98. https://doi.org/10.1177/1528083713477443
Xu C, Cheng Z (2021) Thermal Stability of Ionic Liquids: Current Status and Prospects for Future Development. Processes 9(2):337. https://doi.org/10.3390/pr9020337
Yuan C, Shi W, Chen P, Chen H, Zhang L, Hu G, Jin L, Xie H, Zheng Q, Lu S (2019) Dissolution and transesterification of cellulose in γ-valerolactone promoted by ionic liquids. New J Chem 43(1):330–337. https://doi.org/10.1039/c8nj03505a
Acknowledgements
NMR spectra were acquired on instrumentation of the Estonian Center of Analytical Chemistry (https://www.akki.ee, TT4).
Funding
This research was funded by the Estonian Research Council, RESTA10.
Author information
Authors and Affiliations
Contributions
Conceptualization, E.T.; Validation, E.T.; Investigation, E.T., N.S., I.K., M.K., A.M., I.R., T.K., V.M., R.S.; Writing—original draft, E.T. and N.S.; Writing—review & editing, A.K.; Funding acquisition, A.K. The initial draft of the manuscript was written by E. Tarasova, and all authors reviewed and provided feedback on earlier versions. Finally, all authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tarasova, E., Savale, N., Trifonova, L. et al. Effect of green co-solvents on properties and synthesis of cellulose esters in superbase ionic liquid. Cellulose (2024). https://doi.org/10.1007/s10570-024-05920-x
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10570-024-05920-x