Abstract
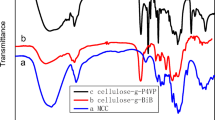
Ion pair self-assembly (IPSAM) responsive to temperature and oxidation was prepared using quaternium hydroxyethyl cellulose ethoxylate (QHECE) as a cationic hydrophilic polymer and (phenylthio) acetic acid (PTA) as a hydrophobic counter ion. The IPSAM was spontaneously formed when the quaternium to carboxylic group molar ratio was 1/9–2/8. QHECE/PTA IPSAM was found as sphere-like nanoparticles whose diameter was tens of nanometers on the TEM micrograph. The ion pair showed the upper critical solution temperature (UCST) that increased with increasing the PTA content and decreased when the PTA of the ion pair was oxidized by H2O2. The ion pair was interface-active due to its amphiphilic property and the interface activity was decreased upon the PTA oxidation. The critical micelle concentration of the ion pair was about 10.5 mg/ml, determined by a fluorospectroscopic method, and it decreased upon oxidation. The ionic interaction between QHECE and PTA and the oxidation of PTA were confirmed by FT-IR spectroscopy, and the oxidation was re-confirmed by X-ray photoelectron microscopy. The release of a payload (i.e. nile red) in IPSAM was restrained below the UCST but it was triggered above the phase transition temperature possibly due to the disintegration of the IPSAM. The release was expedited by the PTA oxidation, possibly because the UCST shifted downward below the release medium temperature. The release was somewhat dependent on the pH value but it was not affected by pH value as much as by temperature and oxidation.










Similar content being viewed by others
Data availability
All relevant data are included in the manuscript. Data can be available from the corresponding author upon request.
References
Attwood D, Florence AT (1983) Surfactant systems:their chemistry, pharmacy, and biology, London
Cho A, La Y, Jeoung S, Moon HR, Ryu JH, Shin TJ, Kim KT (2017) Mix-and-match assembly of block copolymer blends in solution. Macromolecules 50:3234–3243. https://doi.org/10.1021/acs.macromol.7b00438
Discher DE, Ortiz V, Srinivas G, Klein ML, Kim YH, Christian D, Cai S, Photos P, Ahmed F (2007) Emerging applications of polymersomes in delivery: from molecular dynamics to shrinkage of tumors. Prog Polym Sci 32:838–857. https://doi.org/10.1016/j.progpolymsci.2007.05.011
Dutta AK, Kamada K, Ohta K (1996) Spectroscopic studies of nile red in organic solvents and polymers. J Photochem Photobiol A 93:57–64. https://doi.org/10.1016/1010-6030(95)04140-0
Elsabee MZ, Morsi RE, Al-Sabagh AM (2009) Surface active properties of chitosan and its derivatives. Colloids Surf B: Biointerfaces 74:1–16. https://doi.org/10.1016/j.colsurfb.2009.06.021
Fenyves R, Schmutz M, Horner IJ, Bright FV, Rzayev J (2014) Aqueous self-assembly of giant bottlebrush block copolymer surfactants as shape-tunable building blocks. J Am Chem Soc 136:7762–7770. https://doi.org/10.1021/ja503283r
Fong C, Le T, Drummond CJ (2012) Lyotropic liquid crystal engineering–ordered nanostructured small molecule amphiphile self-assembly materials by design. Chem Soc Rev 41:1297–1322. https://doi.org/10.1039/C1CS15148G
Grzybowski BA, Wilmer CE, Kim J, Browne KP, Bishop KJM (2009) Self-assembly: from crystals to cells. Soft Matter 5:1110–1128. https://doi.org/10.1039/B819321P
Has C, Pan S (2021) Vesicle formation mechanisms: an overview. J Liposome Res 31:90–111. https://doi.org/10.1080/08982104.2020.1730401
Hickey RJ, Haynes AS, Kikkawa JM, Park SJ (2011) Controlling the self-assembly structure of magnetic nanoparticles and amphiphilic block-copolymers: from micelles to vesicles. J Am Chem Soc 133:1517–1525. https://doi.org/10.1021/ja1090113
Israelachvili JN, Mitchell DJ, Ninham BW (1976) Theory of self-assembly of hydrocarbon amphiphiles into micelles and bilayers. J Chem Soc Faraday Trans 2 72:1525–1568. https://doi.org/10.1039/F29767201525
Israelachvili JN, Marčelja S, Horn RG (1980) Physical principles of membrane organization. Q Rev Biophys 13:121–200. https://doi.org/10.1017/S0033583500001645
Jahan R, Bodratti AM, Tsianou M, Alexandridis P (2020) Biosurfactants, natural alternatives to synthetic surfactants: physicochemical properties and applications. Adv Colloid Interface Sci 275:102061. https://doi.org/10.1016/j.cis.2019.102061
Khalil RA, Zarari AA (2014) Theoretical estimation of the critical packing parameter of amphiphilic self-assembled aggregates. Appl Surf Sci 318:85–89. https://doi.org/10.1016/j.apsusc.2014.01.046
Kim TH, Alle M, Kim JC (2019) Oxidation- and temperature-responsive poly(hydroxyethyl acrylate-co-phenyl vinyl sulfide) Micelle as a potential Anticancer drug carrier. Pharmaceutics 11:462. https://doi.org/10.3390/pharmaceutics11090462
Kim TH, Alle M, Park SC, Zhao F, Long W, Samala S, Kim JC (2021) Self-assembly prepared using an ion pair of poly(ethylene imine) and (phenylthio) acetic acid as a drug carrier for oxidation, temperature, and NIR-responsive release. Chem Eng J 415:128954. https://doi.org/10.1016/j.cej.2021.128954
Kulkarni CV (2019) Calculating the ‘chain splay’ of amphiphilic molecules: towards quantifying the molecular shapes. Chem Phys Lipids 218:16–21. https://doi.org/10.1016/j.chemphyslip.2018.11.004
Kurniasih IN, Liang H, Mohr PC, Khot G, Rabe JP, Mohr A (2015) Nile red dye in aqueous surfactant and micellar solution. Langmuir 31:2639–2648. https://doi.org/10.1021/la504378m
Lee JS, Feijen J (2012) Polymersomes for drug delivery: design, formation and characterization. JCR 161:473–483. https://doi.org/10.1016/j.jconrel.2011.10.005
Li S, Wu W, Xiu K, Xu F, Li Z, Li J (2014) Doxorubicin loaded pH-responsive micelles capable of rapid intracellular drug release for potential tumor therapy. J Biomed Nanotechnol 10:1480–1489. https://doi.org/10.1166/jbn.2014.1846
Lombardo D, Kiselev MA, Magazù S, Calandra P (2015) Amphiphiles self-assembly: basic concepts and future perspectives of supramolecular approaches. Adv Condens Matter Phys 2015:151683. https://doi.org/10.1155/2015/151683
Lv Y, Yang B, Li YM, Wu Y, He F, Zhuo RX (2014) Crosslinked triblock copolymeric micelle for redox-responsive drug delivery. Colloids Surf B: Biointerfaces 122:223–230. https://doi.org/10.1016/j.colsurfb.2014.06.068
Mandal Goswami M (2016) Synthesis of micelles guided magnetite (Fe3O4) hollow spheres and their application for AC magnetic field responsive drug release. Sci Rep 6:35721. https://doi.org/10.1038/srep35721
Mosley GL, Yamanishi CD, Kamei DT (2012) Mathematical modeling of vesicle drug delivery systems 1: vesicle formation and stability along with drug loading and release. J Lab Autom 18:34–45. https://doi.org/10.1177/2211068212457161
Mukherjee S, Raghuraman H, Chattopadhyay A (2007) Membrane localization and dynamics of Nile red: effect of cholesterol. Biochim Biophys Acta Biomembr 1768:59–66. https://doi.org/10.1016/j.bbamem.2006.07.010
Nakahara Y, Kida T, Nakatsuji Y, Akashi M (2005) New fluorescence method for the determination of the critical micelle concentration by photosensitive monoazacryptand derivatives. Langmuir 21:6688–6695. https://doi.org/10.1021/la050206j
Pope RLE, Brown AM (2020) A primer on tissue pH and local anesthetic potency. Adv Physiol Educ 44:305–308. https://doi.org/10.1152/advan.00018.2020
Ramanathan M, Shrestha LK, Mori T, Ji Q, Hill JP, Arika K (2013) Amphiphile nanoarchitectonics: from basic physical chemistry to advanced applications. Phys Chem Chem Phys 15:10580–10611. https://doi.org/10.1039/C3CP50620G
Randhawa MA, Iqbal A, Nasimullah M, Akhtar M, Yousaf SM, Turner P (1995) Henderson-Hasselbalch equation is inadequate for the measurement of transmembrane diffusion of drugs and buccal drug absorption is a useful alternative. Gen Pharmacol 26:875–879. https://doi.org/10.1016/0306-3623(94)00256-M
Razavi B, Abdollahi A, Roghani-Mamaqani H, Salami-Kalajahi M (2020) Light- and temperature-responsive micellar carriers prepared by spiropyran-initiated atom transfer polymerization: investigation of photochromism kinetics, responsivities, and controlled release of doxorubicin. Polymers 187:122046. https://doi.org/10.1016/j.polymer.2019.122046
Shakeel M, Mehmood K, Siddiq M (2020) Solution and surface properties of metformin hydrochloride in aqueous solution and its interaction with amino acids and anionic surfactant, sodium dodecyl sulphate. Proc Natl Acad Sci India Sect A Phys Sci 90:389–398. https://doi.org/10.1007/s40010-018-0587-2
Shiab P, Zhang H, Lina L, Songa C, Chen Q, Li Z (2019) Molecular dynamics simulation of four typical surfactants in aqueous solution. RSC Adv 9:3224–3231. https://doi.org/10.1039/C8RA09670H
Wang MH, Jeong JH, Kim JC (2016) Thermo-triggerable self-assembly comprising cinnamoyl polymeric β cyclodextrin and cinnamoyl Pluronic F127. Colloids Surf B Biointerfaces 142:148–158. https://doi.org/10.1016/j.colsurfb.2016.02.048
Yan XY, Lin Z, Zhang W, Xu H, Guo QY, Liu Y, Luo J, Liu XY, Zhang R, Huang J, Liu T, Su Z, Zhang R, Zhang S, Liu T, Cheng SZD (2020) Magnifying the structural components of biomembranes: a prototype for the study of the self-assembly of giant lipids. Angew Chem Int Ed 59:5226–5234. https://doi.org/10.1002/anie.201916149
Yang F, Cao Z, Wang G (2015) Micellar assembly of a photo- and temperature-responsive amphiphilic block copolymer for controlled release. Polym Chem 6:7995–8002. https://doi.org/10.1039/C5PY01435B
Yu X, Zhang WB, Yue K, Li X, Liu H, Xin Y, Wang CL, Wesdemiotis C, Cheng SZD (2012) Giant molecular shape amphiphiles based on polystyrene–hydrophilic [60]fullerene conjugates: click synthesis, solution self-assembly, and phase behavior. J Am Chem Soc 134:7780–7787. https://doi.org/10.1021/ja3000529
Yuba E (2020) Development of functional liposomes by modification of stimuli-responsive materials and their biomedical applications. J Mater Chem B 8:1093–1107. https://doi.org/10.1039/C9TB02470K
Zhang H, Kim JC (2016) Reduction-responsive monoolein cubic phase containing hydrophobically modified poly(ethylene imine) and dithiodipropionic acid. Colloids Surf A Physicochem Eng Asp 506:526–534. https://doi.org/10.1016/j.colsurfa.2016.07.007
Zhao F, Kim JC (2022) Thermo-sensitive self-assembly of poly(ethylene imine)/(phenylthio) acetic acid ion pair in surfactant solutions. Drug Deliv 29:2245. https://doi.org/10.1080/10717544.2022.2027571
Funding
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (No. 2018R1A6A1A03025582) and the National Research Foundation of Korea (NRF) grant funded by the Korea government (MSIT) (No. 2022R1A2C2003353).
Author information
Authors and Affiliations
Contributions
JCK: Conceptualization, Funding acquisition, Project administration, Resources, Validation, Writing—original draft preparation, Writing—review & editing. KW: Data curation, Formal analysis, Investigation, Visualization. KR: Data curation, Formal analysis, Investigation. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Human or animal rights
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wangpimool, K., Rana, K. & Kim, JC. Temperature and oxidation-responsive ion pair self-assembly composed of hydroxyethyl cellulose ethoxylate and phenylthio acetic acid. Cellulose 30, 7591–7606 (2023). https://doi.org/10.1007/s10570-023-05375-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-023-05375-6