Abstract
The development of eco-friendly corrosion inhibitors is a subject of several investigations, especially natural polymers. Aimed at suppressing the corrosion of L80 steel in 1 mol/L hydrochloric acid (HCl), a novel natural polymer inhibitor was developed based on xanthan gum (XG) and β-Cyclodextrin (β-CD) in this work. The corrosion inhibition effect of β-cyclodextrin modified xanthan gum (β-CD-XG) on L80 steel was evaluated by electrochemical methods, and surface analysis technology. Adsorption isotherm studies, Fourier transform infrared spectroscopy (FT-IR), and X-ray photoelectron spectroscopy (XPS) were used to explore the corrosion inhibition mechanism of β-CD-XG on L80 steel. The results suggested that β-CD-XG was classified as a mixed-type inhibitor, and mainly suppressed the anode metal dissolution by a tight adsorption film. The formation of the film was attributed to the chemisorption of –OH, –COO-, –CH2–O–, and –CH2–O–CH2– groups on the surface of L80 steel, which conformed to the Langmuir adsorption model. The experimental results illustrated that the maximum corrosion inhibition efficiency of 94.74% was acquired at 200 mg/L β-CD-XG at 293 K.
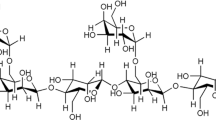
Graphic abstract


modified by β-Cyclodextrin












Similar content being viewed by others
Data availability
My research didn’t generate any data or I reused existing data.
References
Abd El-Lateef HM, Tantawy AH, Abdelhamid AA (2017) Novel quaternary ammonium-based cationic surfactants: synthesis, surface activity and evaluation as corrosion inhibitors for C1018 Carbon steel in acidic chloride solution. J Surfactants Deterg 20:735–753. https://doi.org/10.1007/s11743-017-1947-7
Abd El-Lateef HM, Shalabi K, Tantawy AH (2020a) Corrosion inhibition and adsorption features of novel bioactive cationic surfactants bearing benzenesulphonamide on C1018-steel under sweet conditions: combined modeling and experimental approaches. J Mol Liq 320:114564. https://doi.org/10.1016/j.molliq.2020.114564
Abd El-Lateef HM, Shalabi K, Tantawy AH (2020b) Corrosion inhibition of carbon steel in hydrochloric acid solution using newly synthesized urea-based cationic fluorosurfactants: experimental and computational investigations. New J Chem 44:17791–17814. https://doi.org/10.1039/d0nj04004e
Abdrabo WS, Elgendy B, Soliman KA et al (2020) Synthesis, assessment and corrosion protection investigations of some novel peptidomimetic cationic surfactants: empirical and theoretical insights. J Mol Liq 315:113672. https://doi.org/10.1016/j.molliq.2020.113672
Aghzzaf AA, Rhouta B, Steinmetz J et al (2012) Corrosion inhibitors based on chitosan-heptanoate modified beidellite. Appl Clay Sci 65–66:173–178. https://doi.org/10.1016/j.clay.2012.04.025
Al-Shihry SS, Sayed AR, Abd El-lateef HM (2020) Design and assessment of a novel poly(urethane-semicarbazides) containing thiadiazoles on the backbone of the polymers as inhibitors for steel pipelines corrosion in CO2-saturated oilfield water. J Mol Struct 1201:127223. https://doi.org/10.1016/j.molstruc.2019.127223
Amin MA, Khaled KF, Mohsen Q, Arida HA (2010) A study of the inhibition of iron corrosion in HCl solutions by some amino acids. Corros Sci 52:1684–1695. https://doi.org/10.1016/j.corsci.2010.01.019
Azmeera V, Tungala K, Adhikary P et al (2017) Solution and microwave assisted synthesis of β-Cyclodextrin grafted polyacrylamide: Water treatment and In-vitro drug release study. Int J Biol Macromol 104:1204–1211. https://doi.org/10.1016/j.ijbiomac.2017.06.111
Behpour M, Ghoreishi SM, Mohammadi N et al (2010) Investigation of some Schiff base compounds containing disulfide bond as HCl corrosion inhibitors for mild steel. Corros Sci 52:4046–4057. https://doi.org/10.1016/j.corsci.2010.08.020
Biswas A, Pal S, Udayabhanu G (2015) Experimental and theoretical studies of xanthan gum and its graft co-polymer as corrosion inhibitor for mild steel in 15% HCl. Appl Surf Sci 353:173–183. https://doi.org/10.1016/j.apsusc.2015.06.128
Cao Y, Zou C, Wang C et al (2021) Effect of TiO2 nanoparticles and SDBS on corrosion behavior of 3003 aluminum alloy in aqueous ethylene glycol containing chloride ions at high temperature. J Alloys Compd 873:159820. https://doi.org/10.1016/j.jallcom.2021.159820
Cesari A, Piras AM, Zambito Y et al (2020) 2-Methyl-β-cyclodextrin grafted ammonium chitosan: synergistic effects of cyclodextrin host and polymer backbone in the interaction with amphiphilic prednisolone phosphate salt as revealed by NMR spectroscopy. Int J Pharm 587:119698. https://doi.org/10.1016/j.ijpharm.2020.119698
Chauhan DS, Mouaden KEL, Quraishi MA, Bazzi L (2020) Aminotriazolethiol-functionalized chitosan as a macromolecule-based bioinspired corrosion inhibitor for surface protection of stainless steel in 3.5% NaCl. Int J Biol Macromol 152:234–241. https://doi.org/10.1016/j.ijbiomac.2020.02.283
Dong L, Jiao F, Qin W, Liu W (2019) Selective flotation of scheelite from calcite using xanthan gum as depressant. Miner Eng 138:14–23. https://doi.org/10.1016/j.mineng.2019.04.030
Eduok U, Ohaeri E, Szpunar J (2018) Electrochemical and surface analyses of X70 steel corrosion in simulated acid pickling medium: effect of poly (N-vinyl imidazole) grafted carboxymethyl chitosan additive. Electrochim Acta 278:302–312. https://doi.org/10.1016/j.electacta.2018.05.060
Fan B, Wei G, Zhang Z, Qiao N (2014) Characterization of a supramolecular complex based on octadecylamine and β-cyclodextrin and its corrosion inhibition properties in condensate water. Corros Sci 83:75–85. https://doi.org/10.1016/j.corsci.2014.01.043
Fu L, Lv J, Zhou L et al (2020) Study on corrosion and scale inhibition mechanism of polyaspartic acid grafted β-cyclodextrin. Mater Lett 264:127276. https://doi.org/10.1016/j.matlet.2019.127276
Gerengi H, Darowicki K, Bereket G, Slepski P (2009) Evaluation of corrosion inhibition of brass-118 in artificial seawater by benzotriazole using Dynamic EIS. Corros Sci 51:2573–2579. https://doi.org/10.1016/j.corsci.2009.06.040
Jin W, Song R, Xu W et al (2015) Analysis of deacetylated konjac glucomannan and xanthan gum phase separation by film forming. Food Hydrocoll 48:320–326. https://doi.org/10.1016/j.foodhyd.2015.02.007
Jmiai A, El Ibrahimi B, Tara A et al (2017) Chitosan as an eco-friendly inhibitor for copper corrosion in acidic medium: protocol and characterization. Cellulose 24:3843–3867. https://doi.org/10.1007/s10570-017-1381-z
Jmiai A, El Ibrahimi B, Tara A et al (2018) Alginate biopolymer as green corrosion inhibitor for copper in 1 M hydrochloric acid: experimental and theoretical approaches. J Mol Struct 1157:408–417. https://doi.org/10.1016/j.molstruc.2017.12.060
Khalaf MM, Tantawy AH, Soliman KA, Abd El-Lateef HM (2020) Cationic gemini-surfactants based on waste cooking oil as new ‘green’ inhibitors for N80-steel corrosion in sulphuric acid: a combined empirical and theoretical approaches. J Mol Struct 1203:127442. https://doi.org/10.1016/j.molstruc.2019.127442
Kistamah N, Carr CM, Rosunee S (2009) X-ray photoelectron spectroscopic study of tencel treated with a cationic β-cyclodextrin derivative. Surf Interface Anal 41:710–713. https://doi.org/10.1002/sia.3076
Li M, Xu J, Li R et al (2014) Simple preparation of aminothiourea-modified chitosan as corrosion inhibitor and heavy metal ion adsorbent. J Colloid Interface Sci 417:131–136. https://doi.org/10.1016/j.jcis.2013.11.053
Li J, Sun C, Shuang S et al (2019) Investigation on the flow-induced corrosion and degradation behavior of underground J55 pipe in a water production well in the Athabasca oil sands reservoir. J Pet Sci Eng 182:106325. https://doi.org/10.1016/j.petrol.2019.106325
Liu Y, Zou C, Yan X et al (2015) Β-Cyclodextrin modified natural chitosan as a green inhibitor for carbon steel in acid solutions. Ind Eng Chem Res 54:5664–5672. https://doi.org/10.1021/acs.iecr.5b00930
Mobin M, Rizvi M (2016) Inhibitory effect of xanthan gum and synergistic surfactant additives for mild steel corrosion in 1 M HCl. Carbohydr Polym 136:384–393. https://doi.org/10.1016/j.carbpol.2015.09.027
Pal A, Dey S, Sukul D (2016) Effect of temperature on adsorption and corrosion inhibition characteristics of gelatin on mild steel in hydrochloric acid medium. Res Chem Intermed 42:4531–4549. https://doi.org/10.1007/s11164-015-2295-8
Pan G, Shi Q, Zhang G, Huang G (2020) Selective depression of talc in chalcopyrite flotation by xanthan gum: flotation response and adsorption mechanism. Colloids Surfaces A Physicochem Eng Asp 600:124902. https://doi.org/10.1016/j.colsurfa.2020.124902
Petrović Mihajlović MB, Radovanović MB, Tasić ŽZ, Antonijević MM (2017) Imidazole based compounds as copper corrosion inhibitors in seawater. J Mol Liq 225:127–136. https://doi.org/10.1016/j.molliq.2016.11.038
Qiang Y, Zhang S, Wang L (2019) Understanding the adsorption and anticorrosive mechanism of DNA inhibitor for copper in sulfuric acid. Appl Surf Sci 492:228–238. https://doi.org/10.1016/j.apsusc.2019.06.190
Qiao K, Zeng Y (2020) Comparative study on two imidazolium-based ionic liquid surfactants as corrosion inhibitors for N80 steel in 15% hydrochloric acid solution. Mater Corros 71:1913–1926. https://doi.org/10.1002/maco.202011775
Saleh MM, Mahmoud MG, Abd El-Lateef HM (2019) Comparative study of synergistic inhibition of mild steel and pure iron by 1-hexadecylpyridinium chloride and bromide ions. Corros Sci 154:70–79. https://doi.org/10.1016/j.corsci.2019.03.048
Sayed AR, Abd El-Lateef HM (2020) Thiocarbohydrazones based on adamantane and ferrocene as efficient corrosion inhibitors for hydrochloric acid pickling of c-steel. Coatings 10:1–20. https://doi.org/10.3390/coatings10111068
Solomon MM, Umoren SA (2016) In-situ preparation, characterization and anticorrosion property of polypropylene glycol/silver nanoparticles composite for mild steel corrosion in acid solution. J Colloid Interface Sci 462:29–41. https://doi.org/10.1016/j.jcis.2015.09.057
Sudheer QMA (2013) Electrochemical and theoretical investigation of triazole derivatives on corrosion inhibition behavior of copper in hydrochloric acid medium. Corros Sci 70:161–169. https://doi.org/10.1016/j.corsci.2013.01.025
Tan P (2016) Active phase, catalytic activity, and induction period of Fe/zeolite material in nonoxidative aromatization of methane. J Catal 338:21–29. https://doi.org/10.1016/j.jcat.2016.01.027
Tan B, Zhang S, Qiang Y et al (2018) A combined experimental and theoretical study of the inhibition effect of three disulfide-based flavouring agents for copper corrosion in 0.5 M sulfuric acid. J Colloid Interface Sci 526:268–280. https://doi.org/10.1016/j.jcis.2018.04.092
Tan B, Zhang S, Liu H et al (2019) Corrosion inhibition of X65 steel in sulfuric acid by two food flavorants 2-isobutylthiazole and 1-(1,3-Thiazol-2-yl) ethanone as the green environmental corrosion inhibitors: combination of experimental and theoretical researches. J Colloid Interface Sci 538:519–529. https://doi.org/10.1016/j.jcis.2018.12.020
Tan B, Zhang S, Qiang Y et al (2020) Experimental and theoretical studies on the inhibition properties of three diphenyl disulfide derivatives on copper corrosion in acid medium. J Mol Liq 298:111975. https://doi.org/10.1016/j.molliq.2019.1119756
Tan B, Xiang B, Zhang S et al (2021) Papaya leaves extract as a novel eco-friendly corrosion inhibitor for Cu in H2SO4 medium. J Colloid Interface Sci 582:918–931. https://doi.org/10.1016/j.jcis.2020.08.093
Tan B, Zhang S, He J et al (2021) Insight into anti-corrosion mechanism of tetrazole derivatives for X80 steel in 0.5 M H2SO4 medium: Combined experimental and theoretical researches. J Mol Liq 321:114464. https://doi.org/10.1016/j.molliq.2020.114464
Tantawy AH, Soliman KA, Abd El-Lateef HM (2020) Novel synthesized cationic surfactants based on natural piper nigrum as sustainable-green inhibitors for steel pipeline corrosion in CO2–3.5%NaCl: DFT, Monte Carlo simulations and experimental approaches. J Clean Prod 250:119510. https://doi.org/10.1016/j.jclepro.2019.119510
Umoren SA, Eduok UM (2016) Application of carbohydrate polymers as corrosion inhibitors for metal substrates in different media: a review. Carbohydr Polym 140:314–341. https://doi.org/10.1016/j.carbpol.2015.12.038
Umoren SA, Li Y, Wang FH (2010) Electrochemical study of corrosion inhibition and adsorption behaviour for pure iron by polyacrylamide in H2SO4: synergistic effect of iodide ions. Corros Sci 52:1777–1786. https://doi.org/10.1016/j.corsci.2010.01.026
Umoren SA, AlAhmary AA, Gasem ZM, Solomon MM (2018) Evaluation of chitosan and carboxymethyl cellulose as ecofriendly corrosion inhibitors for steel. Int J Biol Macromol 117:1017–1028. https://doi.org/10.1016/j.ijbiomac.2018.06.014
Vimal Kumar K, Rao BVA (2019) Phosphorylated xanthan gum, an environment-friendly, efficient inhibitor for mild steel corrosion in aqueous 200 ppm NaCl. Mater Today Proc 15:155–165. https://doi.org/10.1016/j.matpr.2019.05.038
Vitaller AV, Angst UM, Elsener B (2019) Corrosion behaviour of l80 steel grade in geothermal power plants in switzerland. Metals (basel) 9:1–15. https://doi.org/10.3390/met9030331
Wang C, Chen J, Han J et al (2019) Enhanced corrosion inhibition performance of novel modified polyaspartic acid on carbon steel in HCl solution. J Alloys Compd 771:736–746. https://doi.org/10.1016/j.jallcom.2018.08.031
Wang C, Zou C, Cao Y (2021) Electrochemical and isothermal adsorption studies on corrosion inhibition performance of β-cyclodextrin grafted polyacrylamide for X80 steel in oil and gas production. J Mol Struct 1228:129737. https://doi.org/10.1016/j.molstruc.2020.129737
Yuan Q, Ge H-H, Sha J-Y et al (2018) Influence of Al2O3 nanoparticles on the corrosion behavior of brass in simulated cooling water. J Alloys Compd 764:512–522. https://doi.org/10.1016/j.jallcom.2018.06.035
Zhang G, Qiu B, Xia Y et al (2019a) Double-helix-superstructure aqueous binder to boost excellent electrochemical performance in Li-rich layered oxide cathode. J Power Sources 420:29–37. https://doi.org/10.1016/j.jpowsour.2019.02.086
Zhang W, Li HJ, Wang M et al (2019b) Tetrahydroacridines as corrosion inhibitor for X80 steel corrosion in simulated acidic oilfield water. J Mol Liq 293:111478. https://doi.org/10.1016/j.molliq.2019.111478
Zheng M, Lian F, Xiong Y et al (2019) The synthesis and characterization of a xanthan gum-acrylamide-trimethylolpropane triglycidyl ether hydrogel. Food Chem 272:574–579. https://doi.org/10.1016/j.foodchem.2018.08.083
Zou C, Zhao P, Lei Y et al (2011) Preparation and performance of a novel water-soluble cationic polymer containing β-cyclodextrin. Chem Eng Technol 34:1820–1826. https://doi.org/10.1002/ceat.201100143
Zou C, Zhao P, Hu X et al (2013) β-Cyclodextrin-functionalized hydrophobically associating acrylamide copolymer for enhanced oil recovery. Energy Fuels 27:2827–2834. https://doi.org/10.1021/ef302152t
Zou C, Yan X, Qin Y et al (2014) Inhibiting evaluation of β-Cyclodextrin-modified acrylamide polymer on alloy steel in sulfuric solution. Corros Sci 85:445–454. https://doi.org/10.1016/j.corsci.2014.04.046
Acknowledgments
This work was financially supported by the National Natural Science Foundation of China (U1662106), and Open Fund (PLN201813) of State Key Laboratory of Oil and Gas Reservoir Geology and Exploitation (Southwest Petroleum University).
Author information
Authors and Affiliations
Contributions
Yixuan Cao: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Validation, Visualization, Writing—original draft, Writing—review & editing. Changjun Zou: Conceptualization, Funding acquisition, Resources, Supervision, Validation. Chengjun Wang: Data curation, Methodology, Project administration, Software. Hao Liang: Supervision, Writing—review & editing. Shuai Lin: Supervision, Writing—review & editing. Ya Liao: Supervision, Software. Lihong Shi: Supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Human and animal participants
This article does not contain any studies involving animals or human participants performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Cao, Y., Zou, C., Wang, C. et al. β-cyclodextrin modified xanthan gum as an eco-friendly corrosion inhibitor for L80 steel in 1 M HCl. Cellulose 28, 11133–11152 (2021). https://doi.org/10.1007/s10570-021-04240-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-021-04240-8