Abstract
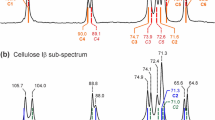
The local structure on the surface of plate-like cellulose II oligomer crystals (CIIOCs) was investigated by NMR spectroscopy. CIIOCs with reducing ends that were selectively labeled with 13C were enzymatically synthesized using cellodextrin phosphorylase. The solid-state 13C cross polarization/magic angle spinning spectra of the 13C-labeled CIIOCs clearly exhibited two resonance peaks (labeled as C1Rα and C1Rβ) derived from the C1 atoms of the reducing ends. The 2D 13C double quantum/13C single quantum homonuclear correlation spectrum indicated that two magnetically nonequivalent glucopyranose rings (Rα and Rβ units) coexisted at the reducing end units. The 1H/13C heteronuclear correlation spectrum suggested that there was a large difference between the local environments around the anomeric carbons of Rα and Rβ units. The abundance ratio of C1Rβ to C1Rα in the solid state was 4:1, whereas that of C1α and C1β in solution was 1:1. When the oligomer chains are packed in the cellulose II crystal, the reducing end units located on the surface of the plate-like crystal may tend to have β-anomeric structure, which would be more sterically stable than the α-anomeric structure.







Similar content being viewed by others
References
Arai M, Tanaka K, Kawaguchi T (1994) Purification and properties of cellodextrin phosphorylase from Clostridium thermocellum. J Ferment Bioeng 77:239–242. https://doi.org/10.1016/0922-338X(94)90226-7
Atalla RH, Gast JC, Sindorf DW et al (1980) 13C NMR spectra of cellulose polymorphs. J Am Chem Soc 102:3249–3251. https://doi.org/10.1021/ja00529a063
Brown SP (2007) Probing proton–proton proximities in the solid state. Prog Nucl Magn Reson Spectrosc 50:199–251. https://doi.org/10.1016/j.pnmrs.2006.10.002
Dudley RL, Fyfe CA, Stephenson PJ et al (1983) High-resolution 13C CP/MAS NMR spectra of solid cellulose oligomers and the structure of cellulose II. J Am Chem Soc 105:2469–2472. https://doi.org/10.1021/ja00346a059
Fyfe CA, Stephenson PJ, Veregin RP et al (1984) Insights into the lattice structure of cellulose II from the high resolution CP/MAS solid state 13C NMR spectrum of cellotetraose. J Carbohydr Chem 3:663–673. https://doi.org/10.1080/07328308408057923
Hiraishi M, Igarashi K, Kimura S et al (2009) Synthesis of highly ordered cellulose II in vitro using cellodextrin phosphorylase. Carbohydr Res 344:2468–2473. https://doi.org/10.1016/j.carres.2009.10.002
Hohwy M, Jakobsen HJ, Edén M et al (1998) Broadband dipolar recoupling in the nuclear magnetic resonance of rotating solids: a compensated C7 pulse sequence. J Chem Phys 108:2686–2694. https://doi.org/10.1063/1.475661
Ishii Y, Yesinowski JP, Tycko R (2001) Sensitivity enhancement in solid-state 13C NMR of synthetic polymers and biopolymers by 1H NMR detection with high-speed magic angle spinning. J Am Chem Soc 123:2921–2922. https://doi.org/10.1021/ja015505j
Isogai A, Usuda M, Kato T et al (1989) Solid-state CP/MAS 13C NMR study of cellulose polymorphs. Macromolecules 22:3168–3172. https://doi.org/10.1021/ma00197a045
Kadokawa J (2011) Precision polysaccharide synthesis catalyzed by enzymes. Chem Rev 111:4308–4345. https://doi.org/10.1021/cr100285v
Kobayashi S, Shoda S, Uyama H (1995) Enzymatic polymerization and oligomerization. Adv Polym Sci 121:1–30
Kobayashi S, Sakamoto J, Kimura S (2001) In vitro synthesis of cellulose and related polysaccharides. Prog Polym Sci 26:1525–1560. https://doi.org/10.1016/S0079-6700(01)00026-0
Kono H, Numata Y (2004) Two-dimensional spin-exchange solid-state NMR study of the crystal structure of cellulose II. Polymer 45:4541–4547. https://doi.org/10.1016/j.polymer.2004.04.025
Krishnareddy M, Kim Y-K, Kitaoka M et al (2002) Cellodextrin phosphorylase from Clostridium thermocellum YM4 strain expressed in Escherichia coli. J Appl Glycosci 49:1–8. https://doi.org/10.5458/jag.49.1
Moulthrop JS, Swatloski RP, Moyna G, Rogers RD (2005) High-resolution 13C NMR studies of cellulose and cellulose oligomers in ionic liquid solutions. Chem Commun 40:1557–1559. https://doi.org/10.1039/b417745b
Pfeffer PE, Hicks KB, Frey MH, Opella SJ, Earl WL (1984) Complete solid state 13C NMR chemical shift assignments for α-d-Glucose, α-d-Glucose-H2O and β-d-Glucose. J Carbohydr Chem 3:197–217. https://doi.org/10.1080/07328308408058815
Reichenbecher M, Lottspeich F, Bronnenmeier K (1997) Purification and properties of a cellobiose phosphorylase (CepA) and a cellodextrin phosphorylase (CepB) from the cellulolytic thermophile Clostridium stercorarium. Eur J Biochem 247:262–267. https://doi.org/10.1111/j.1432-1033.1997.00262.x
Samain E, Lancelon-Pin C, Férigo F et al (1995) Phosphorolytic synthesis of cellodextrins. Carbohydr Res 271:217–226. https://doi.org/10.1016/0008-6215(95)00022-L
Schnell I (2004) Dipolar recoupling in fast-MAS solid-state NMR spectroscopy. Prog Nucl Magn Reson Spectrosc 45:145–207. https://doi.org/10.1016/j.pnmrs.2004.06.003
Schnell I, Spiess HW (2001) High-resolution 1H NMR spectroscopy in the solid state: very fast sample rotation and multiple-quantum coherences. J Magn Reson 151:153–227. https://doi.org/10.1006/jmre.2001.2336
Sheth K, Alexander JK (1969) Purification and properties of β-1,4-oligoglucan:orthophosphate glucosyltransferase from Clostridium thermocellum. J Biol Chem 244:457–464
Silverstein R, Voet J, Reed D, Abeles RH (1967) Purification and mechanism of action of sucrose phosphorylase. J Biol Chem 242:1338–1346
Wang T, Williams JK, Schmidt-Rohr K, Hong M (2015) Relaxation-compensated difference spin diffusion NMR for detecting 13C–13C long-range correlations in proteins and polysaccharides. J Biomol NMR 61:97–107. https://doi.org/10.1007/s10858-014-9889-0
Wiench JW, Bronnimann CE, Lin VSY, Pruski M (2007) Chemical shift correlation NMR spectroscopy with indirect detection in fast rotating solids: studies of organically functionalized mesoporous silicas. J Am Chem Soc 129:12076–12077. https://doi.org/10.1021/ja074746+
Yataka Y, Sawada T, Serizawa T (2015) Enzymatic synthesis and post-functionalization of two-dimensional crystalline cellulose oligomers with surface-reactive groups. Chem Commun 51:12525–12528. https://doi.org/10.1039/C5CC04378F
Acknowledgments
This work was partially supported by a research grant for Exploratory Research on Sustainable Humanosphere Science to R.K. from the Research Institute for Sustainable Humanosphere (RISH) of Kyoto University, a Grant-in-Aid for Scientific Research C (No. 17K05882) to R.K. from the Japan Society for the Promotion of Science (JSPS), and the Advanced Low Carbon Technology Research and Development Program (ALCA; No. JPMJAL1502) from the Japan Science and Technology Agency (JST).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Kita, Y., Kusumi, R., Kimura, T. et al. Surface structural analysis of selectively 13C-labeled cellulose II by solid-state NMR spectroscopy. Cellulose 27, 1899–1907 (2020). https://doi.org/10.1007/s10570-019-02896-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-019-02896-x