Abstract
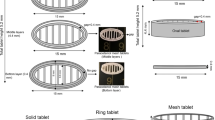
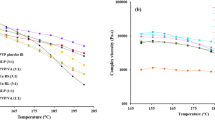
The main objective of this study was to investigate the potential of coupling hot-melt extrusion (HME) and 3D printing in order to design drug containing matrix tablets for the purpose of achieving zero order release. The effect of the blend ratio of ethyl cellulose (EC) and hydroxypropyl cellulose (HPC), carbamazepine (CBZ) as a model drug and triethyl citrate (TEC) on the mechanical and printability properties of extruded filaments was investigated. Filament formulation containing CBZ, EC and HPC (3, 64.7 and 32.3% w/w, respectively) and 20% w/w of TEC (by weight on the dry powder) showed optimum mechanical and printability properties and subsequently was printed into tablets (370 mg, 13 mm diameter, 3.5 mm thickness, cylinder-shaped) at 187 °C. The printed tablets showed good uniformity in drug content and appropriate mechanical properties. The optimum filament showed first order drug release pattern, while the 3D printed tablets showed zero-order drug release and slower drug release rate than the optimum filament. Overall, we have demonstrated that zero order release tablets can be prepared from a 2:1 ratio of EC to HPC, by combination of HME and 3D printing technologies with the capability of reducing dose frequency and adverse effects of drugs.







Similar content being viewed by others
Abbreviations
- HME:
-
Hot-melt extrusion
- 3D:
-
3 dimensional
- EC:
-
Ethyl cellulose
- HPC:
-
Hydroxypropyl cellulose
- CBZ:
-
Carbamazepine
- TEC:
-
Triethyl citrate
- SEM:
-
Scanning electron microscopy
- DSC:
-
Differential scanning calorimetry
References
Arafat B, Qinna N, Cieszynska M, Forbes RT, Alhnan MA (2018a) Tailored on demand anti-coagulant dosing: an in vitro and in vivo evaluation of 3D printed purpose-designed oral dosage forms. Eur J Pharm Biopharm 128:282–289
Arafat B, Wojsz M, Isreb A, Forbes RT, Isreb M, Ahmed W, Arafat T, Alhnan MA (2018b) Tablet fragmentation without a disintegrant: a novel design approach for accelerating disintegration and drug release from 3D printed cellulosic tablets. Eur J Pharm Sci 118:191–199
Bijarimi M, Ahmad S, Rasid R, Khushairi M, Zakir M (2016) Poly (lactic acid)/poly (ethylene glycol) blends: mechanical, thermal and morphological properties. In: AIP Conference Proceedings, AIP Publishing, p 020002
Boetker J, Water JJ, Aho J, Arnfast L, Bohr A, Rantanen J (2016) Modifying release characteristics from 3D printed drug-eluting products. Eur J Pharm Sci 90:47–52
Bruschi ML (2015) Strategies to modify the drug release from pharmaceutical systems. Woodhead Publishing, Cambridge
Chen L, Yang G, Chu X, Gao C, Wang Y, Gong W, Li Z, Yang Y, Yang M, Gao C (2019) Polymer distribution and mechanism conversion in multiple media of phase-separated controlled-release film-coating. Pharmaceutics 11(2):80
Claeys B, Vervaeck A, Hillewaere XK, Possemiers S, Hansen L, De Beer T, Remon JP, Vervaet C (2015) Thermoplastic polyurethanes for the manufacturing of highly dosed oral sustained release matrices via hot melt extrusion and injection molding. Eur J Pharm Biopharm 90:44–52
De Brabander C, Van den Mooter G, Vervaet C, Remon JP (2002) Characterization of ibuprofen as a nontraditional plasticizer of ethyl cellulose. J Pharm Sci 91(7):1678–1685
Djuris J, Nikolakakis I, Ibric S, Djuric Z, Kachrimanis K (2013) Preparation of carbamazepine–Soluplus® solid dispersions by hot-melt extrusion, and prediction of drug–polymer miscibility by thermodynamic model fitting. Eur J Pharm Biopharm 84(1):228–237
Fan W, Zhu W, Zhang X, Xu Y, Di L (2019) Application of the combination of ball-milling and hot-melt extrusion in the development of an amorphous solid dispersion of a poorly water-soluble drug with high melting point. RSC Adv 9(39):22263–22273
Farto-Vaamonde X, Auriemma G, Aquino RP, Concheiro A, Alvarez-Lorenzo C (2019) Post-manufacture loading of filaments and 3D printed PLA scaffolds with prednisolone and dexamethasone for tissue regeneration applications. Eur J Pharm Biopharm 141:100–110
Fuenmayor E, Forde M, Healy A, Devine D, Lyons J, McConville C, Major I (2018) Material considerations for fused-filament fabrication of solid dosage forms. Pharmaceutics 10(2):44
Gareb B, Eissens AC, Kosterink JG, Frijlink HW (2016) Development of a zero-order sustained-release tablet containing mesalazine and budesonide intended to treat the distal gastrointestinal tract in inflammatory bowel disease. Eur J Pharm Biopharm 103:32–42
Goyanes A, Buanz AB, Basit AW, Gaisford S (2014) Fused-filament 3D printing (3DP) for fabrication of tablets. Int J Pharm 476(1–2):88–92
Goyanes A, Wang J, Buanz A, Martínez-Pacheco R, Telford R, Gaisford S, Basit AW (2015) 3D printing of medicines: engineering novel oral devices with unique design and drug release characteristics. Mol Pharm 12(11):4077–4084
Goyanes A, Kobayashi M, Martínez-Pacheco R, Gaisford S, Basit AW (2016) Fused-filament 3D printing of drug products: microstructure analysis and drug release characteristics of PVA-based caplets. Int J Pharm 514(1):290–295
Goyanes A, Allahham N, Trenfield SJ, Stoyanov E, Gasiford S, Basit AW (2019) Direct powder extrusion 3D printing: fabrication of drug products using a novel, single-step process. Int J Pharm 567:118471
Guo F, Wu F, Mu Y, Hu Y, Zhao X, Meng W, Giesy JP, Lin Y (2016) Characterization of organic matter of plants from lakes by thermal analysis in a N 2 atmosphere. Sci Rep 6:22877
Huang S, O’Donnell KP, Keen JM, Rickard MA, McGinity JW, Williams RO (2016) A new extrudable form of hypromellose: AFFINISOL™ HPMC HME. AAPS Pharmscitech 17(1):106–119
Islam MT, Scoutaris N, Maniruzzaman M, Moradiya HG, Halsey SA, Bradley MS, Chowdhry BZ, Snowden MJ, Douroumis D (2015) Implementation of transmission NIR as a PAT tool for monitoring drug transformation during HME processing. Eur J Pharm Biopharm 96:106–116
Jamróz W, Kurek M, Łyszczarz E, Szafraniec J, Knapik-Kowalczuk J, Syrek K, Paluch M, Jachowicz R (2017) 3D printed orodispersible films with Aripiprazole. Int J Pharm 533(2):413–420
Jamróz W, Szafraniec J, Kurek M, Jachowicz R (2018) 3D printing in pharmaceutical and medical applications–recent achievements and challenges. Pharm Res 35(9):176
Kadry H, Wadnap S, Xu C, Ahsan F (2019) Digital light processing (DLP) 3D-printing technology and photoreactive polymers in fabrication of modified-release tablets. Eur J Pharm Sci 135:60–67
Kamel R, Abbas H (2018) PLGA-based monolithic filaments prepared by hot-melt extrusion: in-vitro comparative study. Annales Pharmaceutiques Francaises 76:97–106
Khaled SA, Alexander MR, Irvine DJ, Wildman RD, Wallace MJ, Sharpe S, Yoo J, Roberts CJ (2018) Extrusion 3D printing of paracetamol tablets from a single formulation with tunable release profiles through control of tablet geometry. AAPS Pharmscitech 19(8):3403–3413
Kou W, Cai C, Xu S, Wang H, Liu J, Yang D, Zhang T (2011) In vitro and in vivo evaluation of novel immediate release carbamazepine tablets: complexation with hydroxypropyl-β-cyclodextrin in the presence of HPMC. Int J Pharm 409(1–2):75–80
Kovacevic I, Parojcic J, Homšek I, Tubic-Grozdanis M, Langguth P (2008) Justification of biowaiver for carbamazepine, a low soluble high permeable compound, in solid dosage forms based on IVIVC and gastrointestinal simulation. Mol Pharm 6(1):40–47
Koester LS, Xavier CR, Mayorga P, Bassani VL (2003) Influence of β-cyclodextrin complexation on carbamazepine release from hydroxypropyl methylcellulose matrix tablets. Eur J Pharm Biopharm 55(1):85–91
Ledwon P, Andrade JR, Lapkowski M, Pawlicka A (2015) Hydroxypropyl cellulose-based gel electrolyte for electrochromic devices. Electrochim Acta 159:227–233
Li QJ, Wen HY, Jia DY, Guan XY, Pan H, Yang Y, Yu SH, Zhu ZH, Xiang RW, Pan WS (2017) Preparation and investigation of controlled-release glipizide novel oral device with three-dimensional printing. Int J Pharm 525:5–11
Lim SH, Chia SMY, Kang L, Yap KYL (2016) Three-dimensional printing of carbamazepine sustained-release scaffold. J Pharm Sci 105(7):2155–2163
Löscher W (1998) New visions in the pharmacology of anticonvulsion. Eur J Pharmacol 342(1):1–13
Lu M (2019) Novel excipients and materials used in FDM 3D printing of pharmaceutical dosage forms. 3D and 4D printing in biomedical applications: process engineering and additive manufacturing. Wiley, New York, pp 211–237
Medarević DP, Kachrimanis K, Mitrić M, Djuriš J, Djurić Z, Ibrić S (2016) Dissolution rate enhancement and physicochemical characterization of carbamazepine-poloxamer solid dispersions. Pharm Dev Technol 21(3):268–276
Mujtaba A, Kohli K (2016) In vitro/in vivo evaluation of HPMC/alginate based extended-release matrix tablets of cefpodoxime proxetil. Int J Biol Macromol 89:434–441
Norman J, Madurawe RD, Moore CM, Khan MA, Khairuzzaman A (2017) A new chapter in pharmaceutical manufacturing: 3D-printed drug products. Adv Drug Deliv Rev 108:39–50
Öblom H, Zhang J, Pimparade M, Speer I, Preis M, Repka M, Sandler N (2019) 3D-Printed Isoniazid Tablets for the Treatment and Prevention of Tuberculosis—Personalized Dosing and Drug Release. AAPS Pharmscitech 20(2):52
Palekar S, Nukala PK, Mishra SM, Kipping T, Patel K (2019) Application of 3D printing technology and quality by design approach for development of age-appropriate pediatric formulation of baclofen. Int J Pharm 556:106–116
Parfitt K (1999) Martindale: the complete drug reference. Pharmaceutical Press, London
Patil H, Tiwari RV, Repka MA (2016) Hot-melt extrusion: from theory to application in pharmaceutical formulation. AAPS Pharmscitech 17(1):20–42
Powell G, Saunders M, Rigby A, Marson AG (2010) Immediate‐release versus controlled‐release carbamazepine in the treatment of epilepsy. Cochrane Database Syst Rev 1:CD007124
Sadia M, Isreb A, Abbadi I, Isreb M, Aziz D, Selo A, Timmins P, Alhnan MA (2018) From ‘fixed dose combinations’ to ‘a dynamic dose combiner’: 3D printed bi-layer antihypertensive tablets. Eur J Pharm Sci 123:484–494
Shin TH, Ho MJ, Kim SR, Im SH, Kim CH, Lee S, Kang MJ, Choi YW (2018) Formulation and in vivo pharmacokinetic evaluation of ethyl cellulose-coated sustained release multiple-unit system of tacrolimus. Int J Biol Macromol 109:544–550
Skowyra J, Pietrzak K, Alhnan MA (2015) Fabrication of extended-release patient-tailored prednisolone tablets via fused deposition modelling (FDM) 3D printing. Eur J Pharm Sci 68:11–17
Smith DM, Kapoor Y, Klinzing GR, Procopio AT (2018) Pharmaceutical 3D printing: Design and qualification of a single step print and fill capsule. Int J Pharm 544(1):21–30
Tagami T, Fukushige K, Ogawa E, Hayashi N, Ozeki T (2017) 3D printing factors important for the fabrication of polyvinylalcohol filament-based tablets. Biol Pharm Bull 40(3):357–364
Tang B, Liu Z, Tian Z, Zhang J, Chen X, Fang G, Song H (2019) Development and evaluation of synchronized and sustained release Tripergium Wilfordii tablets based hot-melt extrusion and direct powder compression. J Drug Deliv Sci Technol 53:101208
Tiwari RV, Patil H, Repka MA (2016) Contribution of hot-melt extrusion technology to advance drug delivery in the 21st century. Expert Opin Drug Deliv 13(3):451–464
Wiranidchapong C, Ruangpayungsak N, Suwattanasuk P, Shuwisitkul D, Tanvichien S (2015) Plasticizing effect of ibuprofen induced an alteration of drug released from Kollidon SR matrices produced by direct compression. Drug Dev Ind Pharm 41(6):1037–1046
Yang Y, Wang H, Li H, Ou Z, Yang G (2018) 3D printed tablets with internal scaffold structure using ethyl cellulose to achieve sustained ibuprofen release. Eur J Pharm Sci 115:11–18
Zhang J, Feng X, Patil H, Tiwari RV, Repka MA (2017) Coupling 3D printing with hot-melt extrusion to produce controlled-release tablets. Int J Pharm 519(1–2):186–197
Zhang J, Xu P, Vo AQ, Bandari S, Yang F, Durig T, Repka MA (2019) Development and evaluation of pharmaceutical 3D printability for hot melt extruded cellulose-based filaments. J Drug Deliv Sci Technol 52:292–302
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Homaee Borujeni, S., Mirdamadian, S.Z., Varshosaz, J. et al. Three-dimensional (3D) printed tablets using ethyl cellulose and hydroxypropyl cellulose to achieve zero order sustained release profile. Cellulose 27, 1573–1589 (2020). https://doi.org/10.1007/s10570-019-02881-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-019-02881-4