Abstract
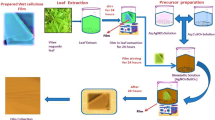
TEMPO nanofibrillated cellulose (TNFC) was used in different concentrations (0, 30, 50, and 70 wt%, all based on the mass of cellulose-copper hybrid material) as template to synthesize copper nanoparticles from copper sulfate; the resulting hybrid material was mixed with polyvinyl alcohol to prepare films. The final composite films were evaluated effectively in terms of their antimicrobial properties against Escherichia coli DH5α and in terms of the copper release from the films on deionized water. Copper concentration was determined by inductively coupled plasma optical emission spectroscopy. The results indicate that increasing the cellulosic material concentration will facilitate the control of copper release from the films. Data regarding to copper release rate was modeled by empirical models; the results indicate that copper release obeys the power law when TNFC is at a low concentration (≤30 %), and presents an exponential behavior when TNFC concentration increase up to 70 % (coefficient R2 is 0.9951).







Similar content being viewed by others
Abbreviations
- TEMPO:
-
2,2,6,6-tetramethylpiperidine-1-oxyl radical
- TNFC:
-
TEMPO nanofibrillated cellulose
- PVA:
-
Polyvinyl alcohol
- CuNPs:
-
Copper nanoparticles
- DI:
-
Deionized water
References
AOAC (1995) Official methods of analysis, 16th edn. In: Association of Official Analytical Chemists International, Gaithersburg, MD
Black JL, Jaczynski J (2006) Temperature effect on inactivation kinetics of Escherichia coli O157: H7 by electron beam in ground beef, chicken breast meat, and trout fillets. J Food Sci 71(6):M221–M227
Buonocore GG, Del Nobile MA, Panizza A, Corbo MR, Nicolais L (2003) A general approach to describe the antimicrobial agent release from highly swellable films intended for food packaging applications. J Control Release 90(1):97–107
Cady NC, Behnke JL, Strickland AD (2011) Copper-based nanostructured coatings on natural cellulose: nanocomposites exhibiting rapid and efficient inhibition of a multi-drug resistant wound pathogen, a. baumannii, and mammalian cell biocompatibility in vitro. Adv Funct Mater 21(13):2506–2514
Cioffi N, Torsi L, Ditaranto N, Tantillo G, Ghibelli L, Sabbatini L, Bleve-Zacheo T, D’Alessio M, Zambonin PG, Traversa E (2005) Copper nanoparticle/polymer composites with antifungal and bacteriostatic properties. Chem Mater 17(21):5255–5262
Duncan TV, Pillai K (2014) Release of engineered nanomaterials from polymer nanocomposites: diffusion, dissolution, and desorption. ACS Appl Mater Interfaces 7(1):2–19
Feng J, Shi Q, Li W, Shu X, Chen A, Xie X, Huang X (2014) Antimicrobial activity of silver nanoparticles in situ growth on TEMPO-mediated oxidized bacterial cellulose. Cellulose 21(6):4557–4567
Fernandes SCM, Freire CSR, Silvestre AJD, Neto CP, Gandini A, Berglund LA, Salmén L (2010) Transparent chitosan films reinforced with a high content of nanofibrillated cellulose. Carbohydr Polym 81(2):394–401
Hahn A, Brandes G, Wagener P, Barcikowski S (2011) Metal ion release kinetics from nanoparticle silicone composites. J Control Release 154(2):164–170
Hanahan D (1985) Techniques for transformation of E. coli. In: Glover DM (ed) DNA cloning, a practical approach, vol I. IRL Press, Oxford, pp 109–135
Isogai A, Saito T, Fukuzumi H (2011) TEMPO-oxidized cellulose nanofibers. Nanoscale 3:71–85
Kumar R, Münstedt H (2005) Silver ion release from antimicrobial polyamide/silver composites. Biomaterials 26(14):2081–2088
Levanduski L, Jaczynski J (2008) Increased resistance of Escherichia coli O157: H7 to electron beam following repetitive irradiation at sub-lethal doses. Int J Food Microbiol 121(3):328–334
López-Carballo G, Higueras L, Gavara R, Hernández-Muñoz P (2012) Silver ions release from antibacterial chitosan films containing in situ generated silver nanoparticles. J Agric Food Chem 61(1):260–267
Raffi M, Mehrwan S, Bhatti TM, Akhter JI, Hameed A, Yawar W, ul Hasan MM (2010) Investigations into the antibacterial behavior of copper nanoparticles against Escherichia coli. Ann Microbial 60(1):75–80
Ritger PL, Peppas NA (1987) A simple equation for description of solute release I. Fickian and non-fickian release from non-swellable devices in the form of slabs, spheres, cylinders or discs. J Control Release 5(1):23–36
Ruparelia JP, Chatterjee AK, Duttagupta SP, Mukherji S (2008) Strain specificity in antimicrobial activity of silver and copper nanoparticles. Acta Biomater 4(3):707–716
Silva AR, Unali G (2011) Controlled silver delivery by silver–cellulose nanocomposites prepared by a one-pot green synthesis assisted by microwaves. Nanotechnology 22(31):315605
Silva FM, Pinto RJ, Daniel-da-Silva AL, Trindade T (2014) Cationic release behavior of antimicrobial cellulose/silver nanocomposites. Cellulose 21(5):3551–3560
Smetana AB, Klabunde KJ, Marchin GR, Sorensen CM (2008) Biocidal activity of nanocrystalline silver powders and particles. Langmuir 24(14):7457–7464
Usman MS, Ibrahim NA, Shameli K, Zainuddin N, Yunus WM (2012) Copper nanoparticles mediated by chitosan: synthesis and characterization via chemical methods. Molecules 17:14928–14936
Usman MS, Zowalaty MEE, Shameli K, Zainuddin N, Salama M, Ibrahim NA (2013) Synthesis, characterization, and antimicrobial properties of copper nanoparticles. Int J Nanomed 8:4467–4479
Xia X, Xie C, Cai S, Yang Z, Yang X (2006) Corrosion characteristics of copper microparticles and copper nanoparticles in distilled water. Corros Sci 48(12):3924–3932
Zhong T, Oporto GS, Jaczynski J, Tesfai AT, Armstrong J (2013) Antimicrobial properties of the hybrid copper nanoparticles-carboxymethyl cellulose. Wood Fiber Sci 45(2):215–222
Zhong T, Oporto GS, Peng Y, Xie X, Gardner DJ (2015a) Drying cellulose-based materials containing copper nanoparticles. Cellulose. doi:10.1007/s10570-015-0646-7
Zhong T, Oporto GS, Jaczynski J, Jiang C (2015b) Nanofibrillated cellulose and copper nanoparticles embedded in polyvinyl alcohol films for antimicrobial applications. Biomed Res Int 2015:1–8
Acknowledgments
Funding for this work has been provided by the USDA NIFA Grant No. 2013-34638-21481 “Development of novel hybrid cellulose nanocomposite film with potent biocide properties utilizing low quality Appalachian hardwoods” and NIFA McStennis WVA00098 “Efficient utilization of biomass for biopolymers in central Appalachia”. The authors also thank Dr. Ronald Sabo from USDA Forest Product Laboratory (FPL) for supplying TEMPO nanofibrillated cellulose gel.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Jiang, C., Oporto, G.S., Zhong, T. et al. TEMPO nanofibrillated cellulose as template for controlled release of antimicrobial copper from PVA films. Cellulose 23, 713–722 (2016). https://doi.org/10.1007/s10570-015-0834-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-015-0834-5