Abstract
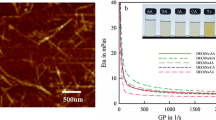
Chitin nanofibers and nanospheres were prepared by dissolution of the polymer in phosphoric acid and regeneration in water. Up to 4 % chitin was readily soluble in concentrated phosphoric acid. By varying the dissolution temperatures and times, chitin nanofibers with widths around 20 nm or chitin nanospheres with sizes around 200–500 nm were obtained with yields more than 80 %. Chemical composition analysis showed that the chemical structures of native chitin were essentially remained after the dissolution and regeneration process, and the degree of substitution of phosphorous on chitin chain was less than 1.00 %. Crystalline structure analysis revealed the crystalline structure of native chitin was not altered, but the crystallinity was decreased after regeneration. Our findings offers a facile and green process for most common labs to prepare chitin nanomaterials with tuned morphology in high yields, which may find great applications in the biomedical, pharmaceutical and food industries.











Similar content being viewed by others
References
Barber PS, Griggs CS, Bonner JR, Rogers RD (2013) Electrospinning of chitin nanofibers directly from an ionic liquid extract of shrimp shells. Green Chem 15:601–607. doi:10.1039/c2gc36582k
Brugnerotto J, Lizardi J, Goycoolea FM, Arguelles-Monal W, Desbrieres J, Rinaudo M (2001) An infrared investigation in relation with chitin and chitosan characterization. Polymer 42:3569–3580. doi:10.1016/s0032-3861(00)00713-8
Chen R-H, Domard A, Muzzarelli RAA, Tokura S, Wang D-M (2011) Advances in chitin/chitosan science and their applications. Carbohydr Polym 84:695. doi:10.1016/j.carbpol.2010.11.049
Chen X, Chew SL, Kerton FM, Yan N (2014) Direct conversion of chitin into a N-containing furan derivative. Green Chem 16:2204–2212. doi:10.1039/c3gc42436g
Chi S, Zivanovic S, Penfield MP (2006) Application of chitosan films enriched with oregano essential oil on bologna—active compounds and sensory attributes. Food Sci Technol Int 12:111–117. doi:10.1177/1082013206063845
Desai K, Kit K, Li J, Davidson PM, Zivanovic S, Meyer H (2009) Nanofibrous chitosan non-wovens for filtration applications. Polymer 50:3661–3669. doi:10.1016/j.polymer.2009.05.058
Fan Y, Saito T, Isogai A (2008a) Chitin nanocrystals prepared by TEMPO-mediated oxidation of α-chitin. Biomacromolecules 9:192–198. doi:10.1021/bm700966g
Fan Y, Saito T, Isogai A (2008b) Preparation of chitin nanofibers from squid pen β-chitin by simple mechanical treatment under acid conditions. Biomacromolecules 9:1919–1923. doi:10.1021/bm800178b
Focher B, Beltrame PL, Naggi A, Torri G (1990) Alkaline N-deacetylation of chitin enhanced by flash treatments—reaction kinetics and structure modifications. Carbohydr Polym 12:405–418. doi:10.1016/0144-8617(90)90090-f
Goodrich JD, Winter WT (2007) α-Chitin nanocrystals prepared from shrimp shells and their specific surface area measurement. Biomacromolecules 8:252–257. doi:10.1021/bm0603589
Hsieh Y-L (2013) Cellulose nanocrystals and self-assembled nanostructures from cotton, rice straw and grape skin: a source perspective. J Mater Sci 48:7837–7846. doi:10.1007/s10853-013-7512-5
Ifuku S, Nogi M, Abe K, Yoshioka M, Morimoto M, Saimoto H, Yano H (2009) Preparation of chitin nanofibers with a uniform width as α-chitin from crab shells. Biomacromolecules 10:1584–1588. doi:10.1021/bm900163d
Ifuku S, Nogi M, Abe K, Yoshioka M, Morimoto M, Saimoto H, Yano H (2011) Simple preparation method of chitin nanofibers with a uniform width of 10–20 nm from prawn shell under neutral conditions. Carbohydr Polym 84:762–764. doi:10.1016/j.carbpol.2010.04.039
Jang MK, Kong BG, Jeong YI, Lee CH, Nah JW (2004) Physicochemical characterization of α-chitin, β-chitin, and γ-chitin separated from natural resources. J Polym Sci A Polym Chem 42:3423–3432. doi:10.1002/pola.20179
Jia X, Chen Y, Shi C, Ye Y, Wang P, Zeng X, Wu T (2014) Preparation and characterization of cellulose regenerated from phosphoric acid. J Agric Food Chem 61:12405–12414. doi:10.1021/jf4042358
Klemm D, Kramer F, Moritz S, Lindstrom T, Ankerfors M, Gray D, Dorris A (2011) Nanocelluloses: a new family of nature-based materials. Angew Chem Int Ed 50:5438–5466. doi:10.1002/anie.201001273
Kumirska J, Czerwicka M, Kaczynski Z, Bychowska A, Brzozowski K, Thoeming J, Stepnowski P (2010) Application of spectroscopic methods for structural analysis of chitin and chitosan. Marine Drugs 8:1567–1636. doi:10.3390/md8051567
Lu P, Hsieh Y-L (2010) Preparation and properties of cellulose nanocrystals: rods, spheres, and network. Carbohydr Polym 82:329–336. doi:10.1016/j.carbpol.2010.04.073
Minke R, Blackwell J (1978) The structure of α-chitin. J Mol Biol 120:167–181. doi:10.1016/0022-2836(78)90063-3
No HK, Cho YI, Kim HR, Meyers SP (2000) Effective deacetylation of chitin under conditions of 15 psi/121 °C. J Agric Food Chem 48:2625–2627. doi:10.1021/jf990842l
Qin Y, Lu X, Sun N, Rogers RD (2010) Dissolution or extraction of crustacean shells using ionic liquids to obtain high molecular weight purified chitin and direct production of chitin films and fibers. Green Chem 12:968–971. doi:10.1039/c003583a
Sikorski P, Hori R, Wada M (2009) Revisit of α-chitin crystal structure using high resolution X-ray diffraction data. Biomacromolecules 10:1100–1105. doi:10.1021/bm801251e
Tzoumaki MV, Moschakis T, Biliaderis CG (2010) Metastability of nematic gels made of aqueous chitin nanocrystal dispersions. Biomacromolecules 11:175–181. doi:10.1021/bm901046c
Tzoumaki MV, Moschakis T, Kiosseoglou V, Biliaderis CG (2011) Oil-in-water emulsions stabilized by chitin nanocrystal particles. Food Hydrocoll 25:1521–1529. doi:10.1016/j.foodhyd.2011.02.008
Vincendon M (1997) Regenerated chitin from phosphoric acid solutions. Carbohydr Polym 32:233–237. doi:10.1016/s0144-8617(97)00005-2
Wu T, Zivanovic S (2008) Determination of the degree of acetylation (DA) of chitin and chitosan by an improved first derivative UV method. Carbohydr Polym 73:248–253. doi:10.1016/j.carbpol.2007.11.024
Wu T, Zivanovic S, Draughon FA, Sams CE (2004) Chitin and chitosan—value-added products from mushroom waste. J Agric Food Chem 52:7905–7910. doi:10.1021/jf0492565
Wu T, Zivanovic S, Draughon FA, Conway WS, Sams CE (2005) Physicochemical properties and bioactivity of fungal chitin and chitosan. J Agric Food Chem 53:3888–3894. doi:10.1021/jf048202s
Yuan Y et al (2014) Chitin microfibers reinforce soy protein gels cross-linked by transglutaminase. J Agric Food Chem 62:4434–4442. doi:10.1021/jf500922n
Zeng J-B, He Y-S, Li S-L, Wang Y-Z (2012) Chitin whiskers: an overview. Biomacromolecules 13:1–11. doi:10.1021/bm201564a
Zhang YHP, Cui JB, Lynd LR, Kuang LR (2006) A transition from cellulose swelling to cellulose dissolution by o-phosphoric acid: evidence from enzymatic hydrolysis and supramolecular structure. Biomacromolecules 7:644–648. doi:10.1021/bm050799c
Zhong C, Cooper A, Kapetanovic A, Fang Z, Zhang M, Rolandi M (2010) A facile bottom-up route to self-assembled biogenic chitin nanofibers. Soft Matter 6:5298–5301. doi:10.1039/c0sm00450b
Zivanovic S, Chi S, Draughon AF (2005) Antimicrobial activity of chitosan films enriched with essential oils. J Food Sci 70:M45–M51
Acknowledgments
This work is supported by the Priority Academic Program Development of Jiangsu Higher Education Institutions (PAPD), Qing Lan Project, the General Program of National Natural Science Foundation of China (31271829), and the Natural Science Foundation of Jiangsu Province (BK2012770).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, T., Wang, G., Gao, C. et al. Phosphoric acid-based preparing of chitin nanofibers and nanospheres. Cellulose 23, 477–491 (2016). https://doi.org/10.1007/s10570-015-0829-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-015-0829-2