Abstract
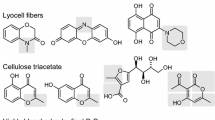
In the present work, aged cotton linters have been analyzed for their chromophore content according to the CRI (“chromophore release & identification”) method. Despite the very low contents in the ppb range, nine chromophores have been unambiguously identified, which makes this account the first one on defined chromophoric structures isolated from cotton. A common feature of the chromophores are 2-hydroxy-[1,4]benzoquinone, 2-hydroxyacetophenone and 5,8-dihydroxynaphthoquinone moieties, which resemble chromophoric structures found in other cellulosic substrates, such as bleached pulps or fibers. The finding of these compounds in lignin-free cotton linters confirms the previous hypothesis that those chromophores are formed from (oxidized) carbohydrate structures rather than from lignin fragments.






Similar content being viewed by others
References
Adorjan I, Potthast A, Rosenau T, Sixta H, Kosma P (2004) Discoloration of cellulose solutions in N-methylmorpholine-N-oxide (Lyocell). Part 1: studies on model compounds and pulps. Cellulose 12(1):51–57
Bader RFW (1990) Atoms in molecules: a quantum theory (International series of monographs on chemistry 22). Oxford University Press, Oxford
Brockmann H, Greve H, Waldmüller W (1971) Dimerisierung und cyclo-Trimerisierung von Naphthazarin und 5.6.7.8-Tetrahydro-chinizarin. Chem Ber 104:1436–1454
Crosby AH, Lutz RE (1956) A study of an oxidative-amination method for the synthesis of aminoquinones. J Am Chem Soc 78(6):1233–1235
Gamble GR (2007) The effect of bale ageing on cotton fiber chemistry, processing performance, and yarn quality. J Cotton Sci 11:98–103
Gamble GR (2008) Method for the prediction of the rate of +b color change in upland cotton (Gossypium hirsutum L.) as a function of storage temperatures. J Cotton Sci 12:171–177
Granovsky AA (2010) Firefly version 7.1.G (2010). http://classic.chem.msu.su/gran/firefly/index.html
Jene PG, Pernin CG, Ibers JA (2001) Comparison of conventional and synchrotron X-ray structure determinations of the adduct 1, 2, 4, 5-tetrahydroxybenzene-2, 5-dihydroxy-1, 4-benzoquinone (1/1) and a conventional X-ray structure determination of 1, 2, 4, 5-tetrahydroxybenzene monohydrate. Acta Cryst C 57:730–734
Keith TA (2010) AIMAll package. http://aim.tkgristmill.com/references.html
Krainz K, Potthast A, Suess U, Dietz T, Nimmerfroh N, Rosenau T (2009) Effects of selected key chromophores on cellulose integrity upon bleaching. Holzforschung 63(6):647–656
Lagercrantz C (1964) Formation of stable radicals in alkaline solution of some monosaccharides. Acta Chem Scand 18(5):1321–1324
Ledl F, Schleicher E (1990) New aspects of the Maillard reaction in foods and in the human body. Angew Chem Int Ed Engl 29(6):565–706
Manzocco L, Calligaris S, Mastrocola D, Nicoli MC, Lerici CR (2000) Review of non-enzymatic browning and antioxidant capacity in processed foods. Trends Food Sci Technol 11(9–10):340–346
Martins SIFS, Jongen WMF, van Boekel MAJS (2000) A review of Maillard reaction in food and implications to kinetic modeling. Trends Food Sci Technol 11(9–10):364–373
Olsson K, Pernemalm PA, Theander O (1978) Formation of aromatic compounds from carbohydrates. Part 7. Acta Chem Scand B 32:249–256
Popoff T, Theander O (1972) Formation of aromatic compounds from carbohydrates. Part 1. Carbohydr Res 22:135–149
Popoff T, Theander O (1976a) Formation of aromatic compounds from carbohydrates. Part 3. Acta Chem Scand B 30:397–402
Popoff T, Theander O (1976b) Formation of aromatic compounds from carbohydrates. Part 4. Acta Chem Scand B 30:705–710
Popoff T, Theander O, Westerlund E (1978) Formation of aromatic compounds from carbohydrates. Part 6. Acta Chem Scand B 32:1–7
Rao TVP, Venkateswarlu V (1965) Chemical eximination of Embelia ribes-VII. Synthesis of some new N-bis(anhydrobenzoquinones). Tetrahedron 21:389–390
Rokita S (ed) (2009) Ortho-Quinone methides in Wiley series on reactive intermediates in chemistry and biology, vol 1. Wiley, New York
Rosenau T, Potthast A, Milacher W, Adorjan I, Hofinger A, Kosma P (2004a) Discoloration of cellulose solutions in N-methylmorpholine-N-oxide (Lyocell). Part 2: isolation and identification of chromophores. Cellulose 12(2):197–208
Rosenau T, Potthast A, Milacher W, Hofinger A, Kosma P (2004b) Isolation and identification of residual chromophores in cellulosic materials. Polymer 45(19):6437–6443
Rosenau T, Adorjan I, Potthast A, Kosma P (2005) Isolation and identification of residual chromomophores in cellulosic materials. Macromol Symp 223(1):239–252
Rosenau T, Potthast A, Kosma P, Suess H-U, Nimmerfroh N (2007) First isolation and identification of residual chromophores from aged bleached pulp samples. Holzforschung 61(6):656–661
Semmingsen D (1977) The crystal and molecular structure of 2, 5-Dihydroxy-1, 4-benzoquinone at −162 °C. Acta Chem Scand B 31:11–14
Suess HU, Filho CL (2003) How to improve brightness stability of Eucalyptus Kraft pulp. Proceedings ABTCP 2003, 36° Congresso Internacional de Celulose e Papel, São Paulo, Brazil
Theander O, Nelson DA (1978) Aqueous, high-temperature transformation of carbohydrates relative to utilization of biomass. Adv Carbohydr Chem Biochem 46:273–326
Theander O, Westerlund E (1980) Formation of aromatic compounds from carbohydrates. Part 8. Acta Chem Scand B 34:701–705
Acknowledgments
We would like to thank the Austrian Christian-Doppler-Society for financial support through the “Christian Doppler Laboratory for Advanced Cellulose Chemistry and Analytics”.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rosenau, T., Potthast, A., Krainz, K. et al. Chromophores in cellulosics, VI. First isolation and identification of residual chromophores from aged cotton linters. Cellulose 18, 1623–1633 (2011). https://doi.org/10.1007/s10570-011-9585-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-011-9585-0