Abstract
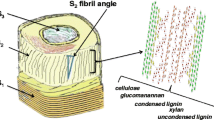
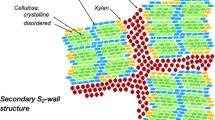
The mechanical and physical properties of wood fibres are dependent on the organisation of their constituent polymers (cellulose, hemicellulose and lignin). Fourier Transform Infrared (FTIR) microscopy was used to examine the anisotropy of the main wood polymers in isolated cell wall fragments from branches of maple and Serbian spruce. Polarised FTIR measurements indicated an anisotropy, i.e. orientation of the cellulose microfibrils that was more or less parallel to the longitudinal axis of the cell wall. The hemicelluloses, glucomannan and xylan appeared to have a close link to the orientation of the cellulose and, thus, an orientation more parallel to the axis of the cell wall. An important result is that, in both maple and spruce samples, lignin was found to be organised in a parallel way in relation to the longitudinal cell wall axis, as well as to the cellulose. The results show that, despite the different lignin precursors and the different types of hemicelluloses in these two kinds of wood, lignin has a similar orientation, when it comes to the longitudinal axis of the cell wall.






Similar content being viewed by others
References
Åkerholm M, Salmén L (2001) Interactions between wood polymers studied by dynamic FT-IR spectroscopy. Polymer 42:963–969
Åkerholm M, Hinterstoisser B, Salmén L (2004) Characterization of the crystalline structure of cellulose using statistic and dynamic FT-IR spectroscopy. Carbohydr Res 339:569–578
Aspinall GO (1980) Chemistry of cell wall polysaccharides. In: Priess J (ed) The biochemistry of plants: a comprehensive treatise. Academic Press, New York, pp 477–500
Atalla RH, Agarwal UP (1985) Raman microprobe evidence for lignin orientation in the cell walls of native woody tissue. Science 227:636–638
Baskin TI (2005) Anisotropic expansion of the plant cell wall. Annu Rev Cell Dev Biol 21:203–222
Bergander A, Salmén L (2002) Cell wall properties and their effects on the mechanical properties of fibres. J Mater Sci 37:151–156
Bogdanovic J, Djikanovic D, Maksimovic V, Tufegdzic S, Djokovic D, Isajev V, Radotic K (2006) Phenolics, lignin content and peroxidase activity in Picea omorika lines. Biol Plant 50:461–464
Bogdanovic J, Milosavic N, Prodanovic R, Ducic T, Radotic K (2007) Variability of antioxidant enzyme activity and isoenzyme profile in needles of Serbian spruce (Picea omorika (Panc.) Purkinye). Biochem System Ecol 35:263–273
Chen M, Sommer AJ, McClure JW (2000) Fourier transform–IR determination of protein contamination in thioglycolic acid lignin from radish seedlings and improved methods for extractive-free cell wall preparation. Phytochem Anal 11:153–159
Faix O (1991) Classification of lignins from different botanical origins by FTIR spectroscopy. Holzforschung 45:21–27
Gierlinger N, Goswami L, Schmidt M, Burgert I, Coutand C, Rogge T, Schwanninger M (2008) In situ FT-IR microscopic study on enzymatic treatment of poplar wood cross-sections. Biomacromolecules 9:2194–2201
Harris PJ (1983) Cell walls. In: Hall JL, Moore AL (eds) Isolation of membranes and organelles from plant cells. Academic Press, London, pp 25–53
Hatfield R, Vermeris W (2001) Lignin formation in plants. The dilemma of linkage specificity. Plant Physiol 126:1351–1357
Houtman CJ, Atalla RH (1995) Cellulose: lignin interactions. A computational study. Plant Physiol 107:977–984
Liang CY, Basset KH, McGinnes EA, Marchessault RH (1960) Infrared spectra of crystalline polysaccharides; VII. Thin wood sections. Tappi 43:232–235
Marchessault RH (1962) Application of infra-red spectroscopy to cellulose and woody polysaccharides. Pure Appl Chem 5:107–129
Melton LD, Smith BG (2001) Current protocols in food analytical chemistry. Wiley, New York
Micic M, Jeremic M, Radotic K, Mavers M, Leblanc R (2000) Visualization of artificial lignin supramolecular structures. Scanning 22:288–294
Micic M, Orbulescu J, Radotic K, Jeremic M, Sui G, Zheng Yu, Leblanc RM (2002) ZL-DHP lignin model compound at the air–water interface. Biophys Chem 99:55–62
Micic M, Radotic K, Jeremic M, Leblanc RM (2003) Study of self-assembly of the lignin model compound on cellulose model substrate. Macromol Biosci 3:100–106
Monties B (1998) Novel structures and properties of lignins in relation to their natural and induced variability in ecotypes, mutants and transgenic plants. Polym Degrad Stabil 59:53–64
Olsson A-M, Bjurhager I, Gerber L, Sundberg B, Salmén L (2011) Ultra-structural organisation of cell wall polymers in normal- and tension wood of aspen revealed by polarisation FT-IR microscopy. Planta 233:1277–1286
Radotic K, Simic-Krstic J, Jeremic M, Trifunovic M (1994) A study of lignin formation at the molecular level by scanning tunneling microscopy. Biophys J 66:1763–1767
Salmon S, Hudson SM (1997) Crystal morphology, biosynthesis and physical assembly of cellulose chitin and chitosan. J Macromol Sci C37:199–263
Sarkanen KV, Hergert HL (1971) Classification and distribution. In: Sarkanen KV, Ludwig CH (eds) Lignins: occurrence, formation, structure and reactions. Wiley, New York, pp 43–94
Steinbach G, Pomozi I, Zsiros O, Páy A, Horváth GV, Garab G (2008) Imaging fluorescence detected linear dichroism of plant cell walls in laser scanning confocal microscope. Cytometry 73A:202–208
Stevanic J, Salmén L (2009) Orientation of the wood polymers in the cell wall of spruce wood fibres. Holzforschung 63:497–503
Strack D, Heilemann J, Wray V, Dirks H (1988) Cell wall: conjugated phenolics from coniferae leaves. Phytochemistry 28:2071–2078
Acknowledgments
This work was supported by the Ministry of Science and Technology of the Republic of Serbia (Project #173017) and a Short Time Scientific Mission in COST Action FP0802 by Jasna Simonović at Innventia, Stockholm, Sweden. The Wallenberg Wood Science Center is acknowledged for funding of Jasna Stevanic and Lennart Salmén. We also acknowledge Gabor Stein and Krzysztof Pawlak, Biology Research Center, Szeged, Hungary, for obtaining images of the cell wall fragments.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Simonović, J., Stevanic, J., Djikanović, D. et al. Anisotropy of cell wall polymers in branches of hardwood and softwood: a polarized FTIR study. Cellulose 18, 1433–1440 (2011). https://doi.org/10.1007/s10570-011-9584-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-011-9584-1