Abstract
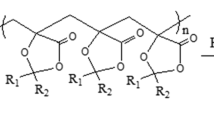
Crosslinking of cellulose fibres was obtained by inducing a substitution reaction in a cationic cellulose ether (NMM-cellulose) prepared by action of N-oxiranylmethyl-N-methylmorpholinium chloride. During the reaction the N-methylmorpholine moiety of the cellulosic ether acts as a leaving group facilitating a covalent bond formation between the ether substituent and a hydroxyl or other nucleophilic group present in the cellulose chain. In order to provide additional evidence of the suggested crosslinking route and investigate its possibilities, different reaction conditions have been investigated and assessed in terms of the obtained fibre properties. The crosslinked fibres were characterized by means of elemental analysis and structure accessibility studies, including accessibility to water, anions and nitrogen gas. According to these investigations heating at 105 °C induces a significant crosslinking. Pre-treatment with acetone restricts it mainly to formation of intra-fibre crosslinks, whereas heating from water suppresses the reactivity but results nevertheless in highly crosslinked structure with both intra- and inter-fibre crosslinks involved.









Similar content being viewed by others
References
Armitt DJ (2001) 4-(4, 6-Dimethoxy-1, 3, 5-triazin-2-yl)-4-methylmorpholinium chloride (DMTMM). Aust J Chem 54:469
Benerito RR, Webre GB, McKelvey JB (1961) A study of reactions between cotton cellulose and difunctional epoxides. Text Res J 31:757–768
Caulifield DF (1994) Ester crosslinking to improve wet performance of paper using multifunctional carboxylic acids, butanetetracarboxylic and citric acid. Tappi J 77(3):205–212
Esposito F, DeNobile MA, Mensitieri G, Nicolais L (1996) Water sorption in cellulose-based hydrogels. J Appl Polym Sci 60:2403–2407
Frick JG, Harper RJ (1982) Crosslinking cotton cellulose with aldehydes. J Appl Polym Sci 27:983–988
Guo W, Ruckenstein E (2002) Crosslinked mercerized cellulose membranes for the affinity chromatography of papain inhibitors. J Memb Sci 197:53–62
Hasani M, Westman G, Potthast A, Rosenau T (2009) Cationization of cellulose by using N- oxiranylmethyl-N-methylmorpholinium chloride and 2-oxiranylpyridine as etherification agents. J Appl Polym Sci 114:1449–1456
Heinze T, Klemm D, Loth F, Philipp B (1990) Preparation, structure and application of ionotropic gels of polysaccharides containing carboxyl groups. Review. Acta Polym 41:259–269
Kunishima M, Kawachi C, Morita J, Terao K, Iwasaki F, Tani S (1999) 4-(4, 6-Dimethoxy-1, 3, 5- triazin-2-yl)-4-methylmorpholinium chloride: an effiecient condensing agent leading to the formation of amides and esters. Tetrahedron 55:13159–13170
Morak AJ, Ward K Jr (1970) Crosslinking of linerboard to reduce stiffness loss II. Diisocyanates in iquid phase application. Tappi J 53(4):652–656
Sakamoto M, Takeda J, Yamada Y, Tonami H (1970) Reversible crosslinking in cellulose. III. Factor controlling the oxidation behaviour of mercapto groups in cotton derivatives. J Polym Sci Par A-I 8:2139–2149
Sannino A, Madaghiele M, Conversano F, Mele G, Maffezzoli A, Netti PA, Ambrosio L, Nicolais L (2004) Cellulose derivative–hyaluronic acid-based microporous hydrogels cross-linked throug divinyl sulfone (DVS) to modulate equilibrium sorption capacity and network stability. Biomacromol 5(1):92–96
Smith FR (1972) The mechanism of swollen state formaldehyde crosslinking of cellulose. Text Res 42(5):301–306
Stevens CV, Smith BF (1970) Cross-linking of cotton cellulose with ethylene urea derivatives havin varying hydrogen-bonding capabilities. Part I: Effect on the physical properties and th hydrogen-bonded structure. Text Res J 40:749–760
Stone JE, Scallan AM (1968) A structural model for the cell wall of water-swollen wood pulp fibre based on their accessibility to macromolecules. Cellulose Chem Technol 2:34–58
Tasker S, Badyal JPS (1994) Influence of crosslinking upon the macroscopic pore structure of cellulose. J Phys Chem 98(31):7599–7601
Vail LS (1972) Chemical and physical properties of cotton modified by N-methylol agents I: chemical structure of the modified cotton. Text Res J 42:360–367
Verburg GB, Snowden FW (1967) Reaction of a diisocyanate with cotton to produce wash- wear fabrics. Text Res J 37:367–371
Welch CM (1983) Glyoxal as a formaldehyde-free durable press reagent for mild curing applications. Text Res J 53:181–186
Welch CM (1988) Tetracarboxylic acids as formaldehyde-free durable press finishing agents. Part I. Catalyst, additive, and durability studies. Text Res J 58:480–486
Welch CM (1992) Formaldehyde-free durable press finishes. Rev Prog Color 22:32–41
Xu Y, Chen C, Yang CQ (1998) Applications of polymeric carboxylic acids for improving paper wet performance. Tappi J 81(11):159–164
Yang CQ, Xu Y (1998) Paper wet performance and ester crosslinking of wood pulp cellulose by poly carboxylic acids. J Appl Polym Sci 67(4):649–658
Acknowledgments
This work has been carried out within Formulosa, a joint project between Södra Cell, Tetra Pak, Korsnäs and Chalmers University of Technology, established within the framework of Avancell—Centre for Fibre Engineering. The authors would like to thank Södra Cell, Tetra Pak, Korsnäs and The Swedish Governmental Agency for Innovation Systems, Vinnova for financial support. Dr. Harald Brelid at Forest Products and Chemical Engineering Department, Chalmers University of Technology and Hannes Kanisto at Chalmers Competence Centre for Catalysis, Gothenburg are gratefully acknowledged for assistance in FSP and BET measurements, respectively.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hasani, M., Westman, G. Self-crosslinking of 2-hydroxypropyl-N-methylmorpholinium chloride cellulose fibres. Cellulose 18, 575–583 (2011). https://doi.org/10.1007/s10570-011-9531-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-011-9531-1