Abstract
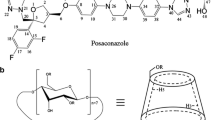
The solubilities of voriconazole, ketoconazole, and clotrimazole with and without hydroxybutenyl-β-cyclodextrin (HBenBCD) in aqueous media were examined. The solubility of these antifungal drugs was significantly improved by complexation with HBenBCD. Both the pH and the type of buffer used to adjust the medium pH had a very significant effect on drug solubilities and the apparent binding constants of the drug:cyclodextrin complexes. Additionally, the stereochemistry of tartrate buffers was found to influence both the electrostatic interaction between drug and tartrate as well as complexation of the drug-tartrate aggregate by HBenBCD. We also compared the solubilization of these antifungal drugs by HBenBCD to other cyclodextrin derivatives with different substituents under identical experimental conditions and found that the amount of drug solubilized was in some cases influenced strongly by the nature of the cyclodextrin. Solid antifungal drug:HBenBCD complexes were prepared and their dissolution profiles were obtained which showed that HBenBCD improved both the rate of dissolution and the amount of drug dissolved.
Similar content being viewed by others
References
Ahmed M.O., El-Gibaly I., Ahmed S.M. (1998). Effect of cyclodextrins on the physicochemical properties and antimycotic activity of clotrimazole. Intl. J. Pharm. 171:111–121
Buchanan C.M., Alderson S.R., Cleven C.D., Dixon D.W., Ivanyi R., Lambert J.L., Lowman D.W., Offerman R.J., Szejtli J., Szente L. (2002). Synthesis and characterization of water-soluble hydroxybutenyl cyclodextrins. Carbohydrate Res. 327(6):493–507
Buchanan C.M., Lambert J.L., Large S.E., Falling S.N., Jicsinszky L., Szejtli J. and Szente L. 2003. Cyclodextrin sulfonates, guest inclusion complexes, methods of making the same and related materials. United States Patent 6,610,671
Chen T.M., Shen H., Zhu C. (2002). Evaluation of a method for high throughput solubility determination using a multi-wavelength UV plate reader. Comb. Chem. High Throughput Screen 5(7):575–581
Connors K.A. (1997). The stability of cyclodextrin complexes in solution. Chem. Rev. 97:1325–1357
Esclusa-Diaz M.T., Guimaraens-Mendez M., Perez-Marcos M.B., Vila-Jato J.L., Torres-Labanderia J.J. (1996). Characterization and in vitro dissolution behavior of ketoconazole/β- and 2-hydroxypropyl-β-cyclodextrin inclusion compounds. Intl. J. Pharm. 143:203–210
Harding D.H. (2003). Pharmaceutical formulations containing voriconazole. United States Patent 6,632,803, B1
Higuchi T., Conners K.A. (1965). Phase-solubility techniques. Adv. Anal. Chem. Instr. 4:117–212
Owens Paul K., Fell Anthony F., Coleman Michael W. and Berridge John C. 2000. Complexation of voriconazole stereoisomers with neutral and anionic derivatized cyclodextrins. J. Incl. Phenom. Macrocyclic Chem. 38(1–4): 133–151
Pan L., Ho Q., Tsutsui K., Takahashi L. (2001). Comparison of chromatographic and spectroscopic methods used to rank compounds for aqueous solubility. J. Pharm. Sci. 90(4):521–529
Pedersen M., Bjerregaard S., Jacobsen J., Sorensen A.M. (1998). A genuine clotrimazole γ-cyclodextrin inclusion complex-isolation, antimycotic activity, toxicity and an unusual dissolution rate. Intl. J. Pharm. 176:121–131
Redenti E., Ventura P., Fronza G., Selva A., Rivara S., Plazzi P.V., Mor M. (1999). Experimental and theoretical analysis of the interaction of ( ± )-cis-ketoconazole with β-cyclodextrin in the presence of (+)-l-tartaric acid. J. Pharm. Sci. 88:599–607
Rotstein D.M., Ketesz D.J., Walker K., Swinney D.C. (1999). Stereoisomers of ketoconazole: preparation and biological activity. J. Med. Chem. 35:2818–2825
Szejtli J. (1991). Cyclodextrins in drug formulations: part II. Pharm. Technol. 15(8):24–38
Szejtli J. (1995). Selectivity/structure correlation in cyclodextrin chemistry. Supramol. Chem. 6:217–223
Szejtli J., Szente L., Redenti E. (2000). Drug/cyclodextrin/hydroxy acid multicomponent systems. Properties and pharmaceutical applications. J. Pharm. Sci. 89:1–8
Taneri F., Guneri T., Aigner Z., Kata M. (2002). Improvement in the physicochemical properties of ketoconazole through complexation with cyclodextrin derivatives. J. Incl. Phenom. Macrocyclic Chem. 44:257–260
Taneri F., Guneri T., Aigner Z., Eroes I. and Kata M. 2003. Improvement of the physicochemical properties of clotrimazole by cyclodextrin complexation. J. Incl. Phenom. Macrocyclic Chem. 46(1–2): 1–13
Taneri F., Guneri T., Aigner Z., Berkesi O., Kata M. (2003a). Thermoanalytical studies on complexes of ketoconazole with cyclodextrin derivatives. J. Thermal Anal. Calorim. 74:769–777
Taneri F., Guneri T., Aigner Z., Kata M. (2003b). Influence of cyclodextrin complexation on the physiochemical and biopharmaceutical properties of ketoconazole. J. Incl. Phenom. Macrocyclic Chem. 47:15–23
Thompson D.O. (1997). Cyclodextrins-enabling excipients: their present and future use in pharmaceuticals. Crit. Rev. Therap. Drug Carrier Sys. 14:1–104
Uekama K., Hirayama F., Irie T. (1998). Cyclodextrin drug carrier systems. Chem. Rev. 98:2045–2076
USP 28-NF 23 711 (2005). The United States Pharmacopeial Convention, Inc., Webcom Limited, Canada 2412–2414
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Buchanan, C.M., Buchanan, N.L., Edgar, K.J. et al. Solubilty and dissolution studies of antifungal drug:hydroxybutenyl-β-cyclodextrin complexes. Cellulose 14, 35–47 (2007). https://doi.org/10.1007/s10570-006-9076-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10570-006-9076-x