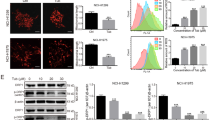
Abstract
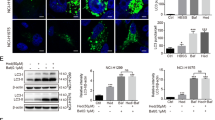
Autophagy is typically activated in cancer cells as a rescue strategy in response to cellular stress (e.g., chemotherapy). Herein, we found that Berbamine Hydrochloride (Ber) can act as an effective inhibitor of the late stage of autophagic flux, thereby potentiating the killing effect of chemotherapy agents. Lung carcinoma cells exposed to Ber exhibited increased autophagosomes, marked by LC3-II upregulation. The increased level of p62 after Ber treatment indicated that the autophagic flux was blocked at the late stage. The lysosome staining assay and cathepsin maturation detection indicated impaired lysosomal acidification. We found that Nox2 exhibited intensified co-localization with lysosomes in Ber-treated cells. Nox2 is a key enzyme for superoxide anion production capable of transferring electrons into the lysosomal lumen, thereby neutralizing the inner protons; this might explain the aberrant acidification. This hypothesis is further supported by the observed reversal of lysosomal cathepsin maturation by Nox2 inhibitors. Finally, Ber combined with cisplatin exhibited a synergistic killing effect on lung carcinoma cells. Further data suggested that lung carcinoma cells co-treated with Ber and cisplatin accumulated excessive reactive oxygen species (ROS), which typically activated MAPK-mediated mitochondria-dependent apoptosis. The enhanced anti-cancer effect of Ber combined with cisplatin was also confirmed in an in vivo xenograft mouse model. These findings indicate that Ber might be a promising adjuvant for enhancing the cancer cell killing effect of chemotherapy via the inhibition of autophagy. In this process, Nox2 might be a significant mediator of Ber-induced aberrant lysosomal acidification.
Graphical abstract







Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Code availability
Not applicable.
Abbreviations
- Ber:
-
Berbamine Hydrochloride;
- Cis:
-
Cisplatin
- PTX:
-
Paclitaxel
- ATCC:
-
American Type Culture Collection
- DMEM:
-
Dulbecco’s Modified Eagle Medium
- CCK8:
-
Cell Counting Kit-8
- Baf:
-
Bafilomycin A1; CQ, chloroquine
- Rapa:
-
Rapamycin
- HBSS:
-
Hank’s Balanced Salt Solution
- AO:
-
Acridine orange
- Cat L:
-
Cathepsin L
- Cat D:
-
Cathepsin D
- V-ATPase:
-
Vacuolar H + -ATPase
- ROS:
-
Reactive oxygen species
- Ras-related C3 botulinum toxin substrate 2:
-
Rac2
- NADPH oxidase:
-
Nox
- NAC:
-
N-Acetylcysteine
- PARP:
-
Poly (ADP-ribose) polymerase
- PI:
-
Propidium iodide
- cyto-c:
-
Cytochrome c
- mito:
-
Mitochondria
- cyto:
-
Cytosol
References
Ashrafi G, Schwarz TL. The pathways of mitophagy for quality control and clearance of mitochondria. Cell Death Differ. 2013;20(1):31–42. https://doi.org/10.1038/cdd.2012.81.
Bedard K, Krause KH. The NOX family of ROS-generating NADPH oxidases: physiology and pathophysiology. Physiol Rev. 2007;87(1):245–313. https://doi.org/10.1152/physrev.00044.2005.
Dai J, Liang K, Zhao S, et al. Chemoproteomics reveals baicalin activates hepatic CPT1 to ameliorate diet-induced obesity and hepatic steatosis. Proc Natl Acad Sci USA. 2018;115(26):E5896-e5905. https://doi.org/10.1073/pnas.1801745115.
Drake LE, Springer MZ, Poole LP, Kim CJ, Macleod KF. Expanding perspectives on the significance of mitophagy in cancer. Semin Cancer Biol. 2017;47:110–24. https://doi.org/10.1016/j.semcancer.2017.04.008.
Duan F, Yu Y, Guan R, Xu Z, Liang H, Hong L. Vitamin K2 Induces Mitochondria-Related Apoptosis in Human Bladder Cancer Cells via ROS and JNK/p38 MAPK Signal Pathways. PLoS ONE. 2016;11(8):e0161886. https://doi.org/10.1371/journal.pone.0161886.
Edelstein CL, Venkatachalam MA, Dong Z. Autophagy inhibition by chloroquine and hydroxychloroquine could adversely affect acute kidney injury and other organ injury in critically ill patients with COVID-19. Kidney Int. 2020;98(1):234–5. https://doi.org/10.1016/j.kint.2020.05.001.
El Far O, Seagar M. A role for V-ATPase subunits in synaptic vesicle fusion? J Neurochem. 2011;117(4):603–12. https://doi.org/10.1111/j.1471-4159.2011.07234.x.
Fan J, Ren D, Wang J, et al. Bruceine D induces lung cancer cell apoptosis and autophagy via the ROS/MAPK signaling pathway in vitro and in vivo. Cell Death Dis. 2020;11(2):126. https://doi.org/10.1038/s41419-020-2317-3.
Figueira TR, Barros MH, Camargo AA, et al. Mitochondria as a source of reactive oxygen and nitrogen species: from molecular mechanisms to human health. Antioxid Redox Signal. 2013;18(16):2029–74. https://doi.org/10.1089/ars.2012.4729.
Fu R, Deng Q, Zhang H, et al. A novel autophagy inhibitor berbamine blocks SNARE-mediated autophagosome-lysosome fusion through upregulation of BNIP3. Cell Death Dis. 2018;9(2):243. https://doi.org/10.1038/s41419-018-0276-8.
Gu J, Gui Y, Chen L, Yuan G, Lu HZ, Xu X. Use of natural products as chemical library for drug discovery and network pharmacology. PLoS ONE. 2013;8(4):e62839. https://doi.org/10.1371/journal.pone.0062839.
Hoang HM, Johnson HE, Heo J. Rac-dependent feedforward autoactivation of NOX2 leads to oxidative burst. J Biol Chem. 2021;297(2):100982. https://doi.org/10.1016/j.jbc.2021.100982.
Jin ZJ. About the evaluation of drug combination. Acta Pharmacol Sin. 2004;25(2):146–7.
Kawai A, Uchiyama H, Takano S, Nakamura N, Ohkuma S. Autophagosome-lysosome fusion depends on the pH in acidic compartments in CHO cells. Autophagy. 2007;3(2):154–7. https://doi.org/10.4161/auto.3634.
Kim KH, Lee MS. Autophagy–a key player in cellular and body metabolism. Nat Rev Endocrinol. 2014;10(6):322–37. https://doi.org/10.1038/nrendo.2014.35.
Kitchen DB, Decornez H, Furr JR, Bajorath J. Docking and scoring in virtual screening for drug discovery: methods and applications. Nat Rev Drug Discovery. 2004;3(11):935–49. https://doi.org/10.1038/nrd1549.
Kleih M, Böpple K, Dong M, et al. Direct impact of cisplatin on mitochondria induces ROS production that dictates cell fate of ovarian cancer cells. Cell Death Dis. 2019;10(11):851. https://doi.org/10.1038/s41419-019-2081-4.
Klionsky DJ, Abdel-Aziz AK, Abdelfatah S, et al. Guidelines for the use and interpretation of assays for monitoring autophagy (4th edition)(1). Autophagy. 2021;17(1):1–382. https://doi.org/10.1080/15548627.2020.1797280.
Kris MG, Gaspar LE, Chaft JE, et al. Adjuvant Systemic Therapy and Adjuvant Radiation Therapy for Stage I to IIIA Completely Resected Non-Small-Cell Lung Cancers: American Society of Clinical Oncology/Cancer Care Ontario Clinical Practice Guideline Update. J Clin Oncol. 2017;35(25):2960–74. https://doi.org/10.1200/jco.2017.72.4401.
Levy JMM, Towers CG, Thorburn A. Targeting autophagy in cancer. Nat Rev Cancer. 2017;17(9):528–42. https://doi.org/10.1038/nrc.2017.53.
Li W, Li Y, Tian W, et al. 2-methylbenzoyl berbamine, a multi-targeted inhibitor, suppresses the growth of human osteosarcoma through disabling NF-κB ERK and AKT Signaling Networks. Aging. 2020a;12(14):15037–49. https://doi.org/10.18632/aging.103565.
Li X, He S, Ma B. Autophagy and autophagy-related proteins in cancer. Mol Cancer. 2020b;19(1):12. https://doi.org/10.1186/s12943-020-1138-4.
Liu L, Sakakibara K, Chen Q, Okamoto K. Receptor-mediated mitophagy in yeast and mammalian systems. Cell Res. 2014;24(7):787–95. https://doi.org/10.1038/cr.2014.75.
Lou X, Zhou Q, Yin Y, Zhou C, Shen Y. Inhibition of the met receptor tyrosine kinase signaling enhances the chemosensitivity of glioma cell lines to CDDP through activation of p38 MAPK pathway. Mol Cancer Ther. 2009;8(5):1126–36. https://doi.org/10.1158/1535-7163.mct-08-0904.
Meng XY, Zhang HX, Mezei M, Cui M. Molecular docking: a powerful approach for structure-based drug discovery. Curr Comput Aided Drug Des. 2011;7(2):146–57. https://doi.org/10.2174/157340911795677602.
Mindell JA. Lysosomal acidification mechanisms. Annu Rev Physiol. 2012;74:69–86. https://doi.org/10.1146/annurev-physiol-012110-142317.
Mizushima N, Yoshimori T, Levine B. Methods in mammalian autophagy research. Cell. 2010;140(3):313–26. https://doi.org/10.1016/j.cell.2010.01.028.
Morrison DK. MAP kinase pathways. Cold Spring Harbor Perspect Biol. 2012;4(11):a011254. https://doi.org/10.1101/cshperspect.a011254
Ni HM, Williams JA, Ding WX. Mitochondrial dynamics and mitochondrial quality control. Redox Biol. 2015;4:6–13. https://doi.org/10.1016/j.redox.2014.11.006.
Pickrell AM, Youle RJ. The roles of PINK1, parkin, and mitochondrial fidelity in Parkinson’s disease. Neuron. 2015;85(2):257–73. https://doi.org/10.1016/j.neuron.2014.12.007.
Pinzi L, Rastelli G. Molecular Docking: Shifting Paradigms in Drug Discovery. Int J Mol Sci. 2019;20(18):4331. https://doi.org/10.3390/ijms20184331
Rastogi R, Geng X, Li F, Ding Y. NOX Activation by Subunit Interaction and Underlying Mechanisms in Disease. Front Cell Neurosci. 2016;10:301. https://doi.org/10.3389/fncel.2016.00301.
Ruan S, Xie R, Qin L, et al. Aggregable Nanoparticles-Enabled Chemotherapy and Autophagy Inhibition Combined with Anti-PD-L1 Antibody for Improved Glioma Treatment. Nano Lett. 2019;19(11):8318–32. https://doi.org/10.1021/acs.nanolett.9b03968.
Rybicka JM, Balce DR, Chaudhuri S, Allan ER, Yates RM. Phagosomal proteolysis in dendritic cells is modulated by NADPH oxidase in a pH-independent manner. EMBO J. 2012;31(4):932–44. https://doi.org/10.1038/emboj.2011.440.
Savina A, Jancic C, Hugues S, et al. NOX2 controls phagosomal pH to regulate antigen processing during crosspresentation by dendritic cells. Cell. 2006;126(1):205–18. https://doi.org/10.1016/j.cell.2006.05.035.
Savina A, Peres A, Cebrian I, et al. The small GTPase Rac2 controls phagosomal alkalinization and antigen crosspresentation selectively in CD8(+) dendritic cells. Immunity. 2009;30(4):544–55. https://doi.org/10.1016/j.immuni.2009.01.013.
Schröder K. NADPH oxidases: Current aspects and tools. Redox Biol. 2020;34:101512. https://doi.org/10.1016/j.redox.2020.101512.
Sharma A, Anand SK, Singh N, Dwarkanath A, Dwivedi UN, Kakkar P. Berbamine induced activation of the SIRT1/LKB1/AMPK signaling axis attenuates the development of hepatic steatosis in high-fat diet-induced NAFLD rats. Food Funct. 2021;12(2):892–909. https://doi.org/10.1039/d0fo02501a.
Siegel RL, Miller KD, Jemal A. Cancer statistics 2020. CA Cancer J Clin. 2020;70(1):7–30. https://doi.org/10.3322/caac.21590.
Trivedi PC, Bartlett JJ, Pulinilkunnil T. Lysosomal Biology and Function: Modern View of Cellular Debris Bin. Cells. 2020;9(5):1131. https://doi.org/10.3390/cells9051131
Violi F, Oliva A, Cangemi R, et al. Nox2 activation in Covid-19. Redox Biol. 2020;36:101655. https://doi.org/10.1016/j.redox.2020.101655.
Wang K, Gong Q, Zhan Y, et al. Blockage of Autophagic Flux and Induction of Mitochondria Fragmentation by Paroxetine Hydrochloride in Lung Cancer Cells Promotes Apoptosis via the ROS-MAPK Pathway. Front Cell Dev Biol. 2019;7:397. https://doi.org/10.3389/fcell.2019.00397.
Wang K, Zhan Y, Chen B, et al. Tubeimoside I-induced lung cancer cell death and the underlying crosstalk between lysosomes and mitochondria. Cell Death Dis. 2020;11(8):708. https://doi.org/10.1038/s41419-020-02915-x.
Xi G, Hu X, Wu B, et al. Autophagy inhibition promotes paclitaxel-induced apoptosis in cancer cells. Cancer Lett. 2011;307(2):141–8. https://doi.org/10.1016/j.canlet.2011.03.026.
Xie J, Ma T, Gu Y, et al. Berbamine derivatives: a novel class of compounds for anti-leukemia activity. Eur J Med Chem. 2009;44(8):3293–8. https://doi.org/10.1016/j.ejmech.2009.02.018.
Yan Y, Chen X, Wang X, et al. The effects and the mechanisms of autophagy on the cancer-associated fibroblasts in cancer. J Exp Clin Cancer Res: CR. 2019;38(1):171. https://doi.org/10.1186/s13046-019-1172-5.
Yao Y, Cui L, Ye J, et al. Dioscin facilitates ROS-induced apoptosis via the p38-MAPK/HSP27-mediated pathways in lung squamous cell carcinoma. Int J Biol Sci. 2020;16(15):2883–94. https://doi.org/10.7150/ijbs.45710.
Yu Q, Wang P, Yang L, et al. Novel synthetic tosyl chloride-berbamine regresses lethal MYC-positive leukemia by targeting CaMKIIγ/Myc axis. Biomed Pharmacother. 2019;117:109134. https://doi.org/10.1016/j.biopha.2019.109134.
Zhang H, Zhu T, Fu R, et al. Combination of Detoxified Pneumolysin Derivative ΔA146Ply and Berbamine as a Treatment Approach for Breast Cancer. Mol Therapy Oncol. 2020;18:247–61. https://doi.org/10.1016/j.omto.2020.06.015.
Zhang SF, Wang XY, Fu ZQ, et al. TXNDC17 promotes paclitaxel resistance via inducing autophagy in ovarian cancer. Autophagy. 2015;11(2):225–38. https://doi.org/10.1080/15548627.2014.998931.
Zhang Y, Wang J, Hui B, et al. Pristimerin enhances the effect of cisplatin by inhibiting the miR-23a/Akt/GSK3β signaling pathway and suppressing autophagy in lung cancer cells. Int J Mol Med. 2019;43(3):1382–94. https://doi.org/10.3892/ijmm.2019.4057.
Zheng K, Jiang Y, Liao C, et al. NOX2-Mediated TFEB Activation and Vacuolization Regulate Lysosome-Associated Cell Death Induced by Gypenoside L, a Saponin Isolated from Gynostemma pentaphyllum. J Agric Food Chem. 2017;65(31):6625–37. https://doi.org/10.1021/acs.jafc.7b02296.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 82074070, 82104571), the Guangdong Natural Science Foundation (Grant No. 2020A1515110632), the Science and Technology Plan Project of Guangzhou of China (Grant No. 202102020721), the Guangdong Provincial Bureau of Traditional Chinese Medicine Research Foundation (Grant No. 20222043, 20221119), 2019 characteristic innovation projects of Guangdong colleges and Universities (Grant No. 2019GKTSCX045), the Guangdong Medical Science and Technology Research Foundation (Grant No. A2020293), and the Bethune Quest-Pharmaceutical Research Capacity Building Project (Grant No. B-19-H-20200622).
Author information
Authors and Affiliations
Contributions
KW and JYX were involved in conceptualization. YJZ, QGC, and YS helped in methodology. YJZ and QGC contributed to validation and formal analysis. XLW was involved in resources and funding acquisition. TXZ and WBZ helped in project administration and data curation. BNC and YS contributed to visualization and software. CXL and YJJ was involved in investigation. YHT and BYD helped in supervision. YJZ was involved in writing—original draft preparation. JYX and KW helped in writing—review and editing and funding acquisition.
Corresponding authors
Ethics declarations
Ethics approval
The animal experiment was approved by the Institutional Animal Care and Use Committee at Guangzhou University of Chinese Medicine (Project No. 20190927002).
Consent for publication
Not applicable.
Consent to participate
Not applicable.
Competing interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Headlights
• Berbamine Hydrochloride (Ber) accelerated the assembly of the Nox2 complex by promoting the activity of Rac2, thereby impairing lysosomal acidification in lung cancer cells.
• Ber potentiated the killing effect of cisplatin through the ROS-MAPK signaling pathway in lung cancer cells.
• The enhancive anti-cancer effect of Ber combined with cisplatin was also confirmed in an in vivo xenograft mouse model.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhan, Y., Chen, Q., Song, Y. et al. Berbamine Hydrochloride inhibits lysosomal acidification by activating Nox2 to potentiate chemotherapy-induced apoptosis via the ROS-MAPK pathway in human lung carcinoma cells. Cell Biol Toxicol 39, 1297–1317 (2023). https://doi.org/10.1007/s10565-022-09756-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-022-09756-8