Abstract
Uterine deficiency of Dnmt3b impairs decidualization and consequent embryo implantation defects. Recent advances in molecular technologies have allowed the unprecedented mapping of epigenetic modifications during embryo implantation. DNA methyltransferase 3a (DNMT3A) and DNMT3B are responsible for establishing DNA methylation patterns produced through their de novo-type DNA methylation activity in implantation stage embryos and during germ cell differentiation. It was reported that conditional knockout of Dnmt3a in the uterus does not markedly affect endometrial function during embryo implantation, but the tissue-specific functions of Dnmt3b in the endometrium during embryo implantation remain poorly understood to investigate the role of Dnmt3b during peri-implantation period. Here, we generated Dnmt3b conditional knockout (Dnmt3bd/d) female mice using progesterone receptor-Cre mice and examined the role of Dnmt3b during embryo implantation. Dnmt3bd/d female mice exhibited compromised fertility, which was associated with defective decidualization, but not endometrial receptivity. Furthermore, results showed loss of Dnmt3b did not lead to altered genomic methylation patterns of the decidual endometrium during early pregnancy. Transcriptome sequencing analysis of uteri from day 6 pregnant mice identified phosphoglycerate kinase 1 (Pgk1) as one of the most variable genes in Dnmt3bd/d decidual endometrium. Potential roles of PGK1 in the decidualization process during early pregnancy were confirmed. Lastly, the compromised decidualization upon the downregulation of Dnmt3b could be reversed by overexpression of Pgk1. Collectively, our findings indicate that uterine deficiency of Dnmt3b impairs decidualization and consequent embryo implantation defects.
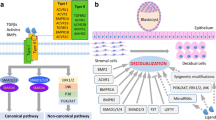
Graphical abstract

Graphical highlights
1. Dnmt3bd/d female mice exhibited compromised fertility, which was associated with defective decidualization, but not endometrial receptivity. 2. Losing Dnmt3b did not alter genomic methylation patterns of the decidual endometrium during early pregnancy. 3. Pgk1 was one of the most variable genes in Dnmt3bd/d decidual endometrium and the compromised decidualization upon the downregulation of Dnmt3b was related to the overexpression of Pgk1.








Similar content being viewed by others
Availability of data and material
All data generated or analyzed during this study are included either in this article or in the supplemental materials.
References
Aikawa S, Kano K, Inoue A, Wang J, Saigusa D, et al. Autotaxin-lysophosphatidic acid-LPA3 signaling at the embryo-epithelial boundary controls decidualization pathways. EMBO J. 2017;36:2146–60. https://doi.org/10.15252/embj.201696290.
Attwood JT, Yung RL, Richardson BC. DNA methylation and the regulation of gene transcription. Cellular and Molecular Life Sciences : CMLS. 2002;59:241–57. https://doi.org/10.1007/s00018-002-8420-z.
Bishop KS, Ferguson LR. The interaction between epigenetics, nutrition and the development of cancer. Nutrients. 2015;7:922–47. https://doi.org/10.3390/nu7020922.
Bulmer JN, Innes BA, Levey J, Robson SC, Lash GE. The role of vascular smooth muscle cell apoptosis and migration during uterine spiral artery remodeling in normal human pregnancy. FASEB Journal : Official Publication of the Federation of American Societies for Experimental Biology. 2012;26:2975–85. https://doi.org/10.1096/fj.12-203679.
Cha J, Sun X, Dey SK. Mechanisms of implantation: strategies for successful pregnancy. Nat Med. 2012;18:1754–67. https://doi.org/10.1038/nm.3012.
Chen Z, Zhang Y. Role of mammalian DNA methyltransferases in development. Annu Rev Biochem. 2020;89:135–58. https://doi.org/10.1146/annurev-biochem-103019-102815.
Colicchia M, Campagnolo L, Baldini E, Ulisse S, Valensise H, Moretti C. Molecular basis of thyrotropin and thyroid hormone action during implantation and early development. Hum Reprod Update. 2014;20:884–904. https://doi.org/10.1093/humupd/dmu028.
Croy BA, van den Heuvel MJ, Borzychowski AM, Tayade C. Uterine natural killer cells: a specialized differentiation regulated by ovarian hormones. Immunol Rev. 2006;214:161–85. https://doi.org/10.1111/j.1600-065X.2006.00447.x.
Deb K, Reese J, Paria BC. Methodologies to study implantation in mice. Methods Mol Med. 2006;121:9–34. https://doi.org/10.1385/1-59259-983-4:007.
Diao H, Li R, El Zowalaty AE, Xiao S, Zhao F, Dudley EA, Ye X. Deletion of Lysophosphatidic acid receptor 3 (Lpar3) disrupts fine local balance of progesterone and estrogen signaling in mouse uterus during implantation. Biol Reprod. 2015;93:123. https://doi.org/10.1095/biolreprod.115.131110.
Ding YB, Long CL, Liu XQ, Chen XM, Guo LR, Xia YY, He JL, Wang YX. 5-aza-2’-deoxycytidine leads to reduced embryo implantation and reduced expression of DNA methyltransferases and essential endometrial genes. PLoS ONE. 2012;7: e45364. https://doi.org/10.1371/journal.pone.0045364.
Dorostghoal M, Ghaffari HO, Marmazi F, Keikhah N. Overexpression of endometrial estrogen receptor-alpha in the window of implantation in women with unexplained infertility. International Journal of Fertility & Sterility. 2018;12:37–42. https://doi.org/10.22074/ijfs.2018.5118.
Gagliardi M, Strazzullo M, Matarazzo MR. DNMT3B functions: novel insights from human disease. Frontiers in Cell and Developmental Biology. 2018;6:140. https://doi.org/10.3389/fcell.2018.00140.
Gao J, Mazella J, Tseng L. Hox proteins activate the IGFBP-1 promoter and suppress the function of hPR in human endometrial cells. DNA Cell Biol. 2002;21:819–25. https://doi.org/10.1089/104454902320908469.
Gao F, Bian F, Ma X, Kalinichenko VV, Das SK. Control of regional decidualization in implantation: role of FoxM1 downstream of Hoxa10 and cyclin D3. Sci Rep. 2015;5:13863. https://doi.org/10.1038/srep13863.
Gao L, Anteneh H, Song J. Dissect the DNMT3A- and DNMT3B-mediated DNA Co-methylation through a Covalent Complex Approach. J Mol Biol. 2020;432:569–75. https://doi.org/10.1016/j.jmb.2019.11.004.
Gellersen B, Brosens IA, Brosens JJ. Decidualization of the human endometrium: mechanisms, functions, and clinical perspectives. Seminars in Reproductive Medicine. 2007;25:445–53. https://doi.org/10.1055/s-2007-991042.
Geng Y, Gao R, Chen X, Liu X, Liao X, Li Y, Liu S, Ding Y, Wang Y, He J. Folate deficiency impairs decidualization and alters methylation patterns of the genome in mice. Mol Hum Reprod. 2015;21:844–56. https://doi.org/10.1093/molehr/gav045.
Hansen RS, Wijmenga C, Luo P, Stanek AM, Canfield TK, Weemaes CM, Gartler SM. The DNMT3B DNA methyltransferase gene is mutated in the ICF immunodeficiency syndrome. Proc Natl Acad Sci USA. 1999;96:14412–7. https://doi.org/10.1073/pnas.96.25.14412.
He, Y., Luo, Y., Zhang, D., Wang, X., Zhang, P., Li, H., Ejaz, S., and Liang, S. (2019). PGK1-mediated cancer progression and drug resistance. American journal of cancer research 9, 2280–2302.doi:
Hui Y, Zhang Y, Wang K, Pan C, Chen H, Qu L, Song X, Lan X. Goat DNMT3B: An indel mutation detection, association analysis with litter size and mRNA expression in gonads. Theriogenology. 2020;147:108–15. https://doi.org/10.1016/j.theriogenology.2020.02.025.
Iurlaro M, von Meyenn F, Reik W. DNA methylation homeostasis in human and mouse development. Curr Opin Genet Dev. 2017;43:101–9. https://doi.org/10.1016/j.gde.2017.02.003.
Kamae C, Imai K, Kato T, Okano T, Honma K, et al. Clinical and Immunological Characterization of ICF Syndrome in Japan. J Clin Immunol. 2018;38:927–37. https://doi.org/10.1007/s10875-018-0559-y.
Lessey BA, Palomino WA, Apparao KB, Young SL, Lininger RA. Estrogen receptor-alpha (ER-alpha) and defects in uterine receptivity in women. Reproductive Biology and Endocrinology : RB&E. 2006;4(Suppl 1):S9. https://doi.org/10.1186/1477-7827-4-S1-S9.
Li N, Lu S, Ding Y, Chen X, He J, et al. Uterine Dnmt3a is not required for mouse embryo implantation. Curr Mol Med. 2020. https://doi.org/10.2174/1566524020666200225115526.
Liu JL, Wang TS. Systematic analysis of the molecular mechanism underlying decidualization using a text mining approach. PLoS ONE. 2015;10: e0134585. https://doi.org/10.1371/journal.pone.0134585.
Ma WG, Song H, Das SK, Paria BC, Dey SK. Estrogen is a critical determinant that specifies the duration of the window of uterine receptivity for implantation. Proc Natl Acad Sci USA. 2003;100:2963–8. https://doi.org/10.1073/pnas.0530162100.
Maekawa R, Tamura I, Shinagawa M, Mihara Y, Sato S, Okada M, Taketani T, Tamura H, Sugino N. Genome-wide DNA methylation analysis revealed stable DNA methylation status during decidualization in human endometrial stromal cells. BMC Genomics. 2019;20:324. https://doi.org/10.1186/s12864-019-5695-0.
Nagashima T, Li Q, Clementi C, Lydon JP, DeMayo FJ, Matzuk MM. BMPR2 is required for postimplantation uterine function and pregnancy maintenance. J Clin Investig. 2013;123:2539–50. https://doi.org/10.1172/JCI65710.
Nowialis P, Lopusna K, Opavska J, Haney SL, Abraham A, et al. Catalytically inactive Dnmt3b rescues mouse embryonic development by accessory and repressive functions. Nat Commun. 2019;10:4374. https://doi.org/10.1038/s41467-019-12355-7.
Ren W, Gao L, Song J (2018) Structural basis of DNMT1 and DNMT3A-mediated DNA methylation. Genes 9.https://doi.org/10.3390/genes9120620
Rinaldi L, Datta D, Serrat J, Morey L, Solanas G, et al. Dnmt3a and Dnmt3b associate with enhancers to regulate human epidermal stem cell homeostasis. Cell Stem Cell. 2016;19:491–501. https://doi.org/10.1016/j.stem.2016.06.020.
Rinaldi, L., Avgustinova, A., Martin, M., Datta, D., Solanas, G., Prats, N., and Benitah, S.A. (2017). Loss of Dnmt3a and Dnmt3b does not affect epidermal homeostasis but promotes squamous transformation through PPAR-gamma. eLife 6.doi:https://doi.org/10.7554/eLife.21697
Salker MS, Christian M, Steel JH, Nautiyal J, Lavery S, et al. Deregulation of the serum- and glucocorticoid-inducible kinase SGK1 in the endometrium causes reproductive failure. Nat Med. 2011;17:1509–13. https://doi.org/10.1038/nm.2498.
Salker MS, Steel JH, Hosseinzadeh Z, Nautiyal J, Webster Z, Singh Y, Brucker S, Lang F, Brosens JJ. Activation of SGK1 in endometrial epithelial cells in response to PI3K/AKT Inhibition impairs embryo implantation. Cellular Physiology and Biochemistry : International Journal of Experimental Cellular Physiology, Biochemistry, and Pharmacology. 2016;39:2077–87. https://doi.org/10.1159/000447903.
Salleh N, Sayem ASM, Giribabu N, Khaing SL. Expression of proteins related to thyroid hormone function in the uterus is down-regulated at the day of implantation in hypothyroid pregnant rats. Cell Biol Int. 2019;43:486–94. https://doi.org/10.1002/cbin.11114.
Shariati MBH, Niknafs B, Seghinsara AM, Shokrzadeh N, Alivand MR. Administration of dexamethasone disrupts endometrial receptivity by alteration of expression of miRNA 223, 200a, LIF, Muc1, SGK1, and ENaC via the ERK1/2-mTOR pathway. J Cell Physiol. 2019;234:19629–39. https://doi.org/10.1002/jcp.28562.
Shui LJ, Meng Y, Huang C, Qian Y, Liu JY. Aminopeptidase N expression in the endometrium could affect endometrial receptivity. Biochem Biophys Res Commun. 2019;514:469–74. https://doi.org/10.1016/j.bbrc.2019.04.174.
Singh M, Chaudhry P, Asselin E. Bridging endometrial receptivity and implantation: network of hormones, cytokines, and growth factors. J Endocrinol. 2011;210:5–14. https://doi.org/10.1530/JOE-10-0461.
Su Y, Guo S, Liu C, Li N, Zhang S, et al. Endometrial pyruvate kinase M2 is essential for decidualization during early pregnancy. J Endocrinol. 2020;245:357–68. https://doi.org/10.1530/JOE-19-0553.
Tan Y, Li M, Cox S, Davis MK, Tawfik O, Paria BC, Das SK. HB-EGF directs stromal cell polyploidy and decidualization via cyclin D3 during implantation. Dev Biol. 2004;265:181–95. https://doi.org/10.1016/j.ydbio.2003.09.019.
Tapia A, Vilos C, Marin JC, Croxatto HB, Devoto L. Bioinformatic detection of E47, E2F1 and SREBP1 transcription factors as potential regulators of genes associated to acquisition of endometrial receptivity. Reproductive Biology and Endocrinology : RB&E. 2011;9:14. https://doi.org/10.1186/1477-7827-9-14.
Wang S, Jiang B, Zhang T, Liu L, Wang Y, et al. Insulin and mTOR pathway regulate HDAC3-mediated deacetylation and activation of PGK1. PLoS Biol. 2015;13: e1002243. https://doi.org/10.1371/journal.pbio.1002243.
Wang L, Bo X, Yi X, Xiao X, Zheng Q, Ma L, Li B. Exosome-transferred LINC01559 promotes the progression of gastric cancer via PI3K/AKT signaling pathway. Cell Death Dis. 2020;11:723. https://doi.org/10.1038/s41419-020-02810-5.
Xu GL, Bestor TH, Bourc’his D, Hsieh CL, Tommerup N, Bugge M, Hulten M, Qu X, Russo JJ, Viegas-Pequignot E. Chromosome instability and immunodeficiency syndrome caused by mutations in a DNA methyltransferase gene. Nature. 1999;402:187–91. https://doi.org/10.1038/46052.
Xu T, Wang C, Shen J, Tong P, O’Keefe R. Ablation of Dnmt3b in chondrocytes suppresses cell maturation during embryonic development. J Cell Biochem. 2018;119:5852–63. https://doi.org/10.1002/jcb.26775.
Yan B, Guo Q, Nan XX, Wang Z, Yin Z, Yi L, Wei YB, Gao YL, Zhou KQ, Yang JR. Micro-ribonucleic acid 29b inhibits cell proliferation and invasion and enhances cell apoptosis and chemotherapy effects of cisplatin via targeting of DNMT3b and AKT3 in prostate cancer. Onco Targets Ther. 2015;8:557–65. https://doi.org/10.2147/OTT.S76484.
Yu M, Qin H, Wang H, Liu J, Liu S, Yan Q. N-glycosylation of uterine endometrium determines its receptivity. J Cell Physiol. 2020;235:1076–89. https://doi.org/10.1002/jcp.29022.
Zhang C, Yang C, Li N, Liu X, He J, et al. Elevated insulin levels compromise endometrial decidualization in mice with decrease in uterine apoptosis in early-stage pregnancy. Arch Toxicol. 2019;93:3601–15. https://doi.org/10.1007/s00204-019-02601-8.
Zuo RJ, Gu XW, Qi QR, Wang TS, Zhao XY, Liu JL, Yang ZM. Warburg-like glycolysis and lactate shuttle in mouse decidua during early pregnancy. J Biol Chem. 2015;290:21280–91. https://doi.org/10.1074/jbc.M115.656629.
Funding
This work was supported by the National Key Research and Development Program of China [2018YFC1004401], the Natural Science Foundation of Chongqing [cstc2018jcyjAX0309], and the Science and Technology Research Program of Chongqing Municipal Education Commission [KJQN201900413].
Author information
Authors and Affiliations
Contributions
YW and RG designed and directed the study. JL and MC performed the analyses and interpretation of data. RG and JL wrote and critically reviewed the manuscript. YW and YD conceived the study. JL and WL performed experiments, the analyses and interpretation of data. MC performed experiments and analyzed data. CT provided technical and study design support. XC, CP, FL provided technical and material support. NL, XL, JH, YG, TL, and XM provided technical support.
Corresponding authors
Ethics declarations
Ethics approval
All procedures involving animal were approved by the Ethics Committee of Chongqing Medical University.
Consent to participate and consent for publication
Informed consent was obtained from all individual participants included in the study.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Long, J., Li, W., Chen, M. et al. Uterine deficiency of Dnmt3b impairs decidualization and causes consequent embryo implantation defects. Cell Biol Toxicol 39, 1077–1098 (2023). https://doi.org/10.1007/s10565-021-09664-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-021-09664-3