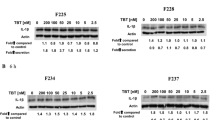
Abstract
Previous studies have shown that dibutyltin (DBT) interferes with the function of human natural killer (NK) cells, diminishing their capacity to destroy tumor cells, in vitro. DBT is a widespread environmental contaminant and has been found in human blood. As NK cells are our primary immune defense against tumor cells, it is important to understand the mechanism by which DBT interferes with their function. The current study examines the effects of DBT exposures on key enzymes in the signaling pathway that regulates NK responsiveness to tumor cells. These include several protein tyrosine kinases (PTKs), mitogen-activated protein kinases (MAPKs), and mitogen-activated protein kinase kinases (MAP2Ks). The results showed that in vitro exposures of NK cells to DBT had no effect on PTKs. However, exposures to DBT for as little as 10 min were able to increase the phosphorylation (activation) of the MAPKs. The DBT-induced activations of these MAPKs appear to be due to DBT-induced activations of the immediate upstream activators of the MAPKs, MAP2Ks. The results suggest that DBT-interference with the MAPK signaling pathway is a consequence of DBT exposures, which could account for DBT-induced decreases in NK function.





Similar content being viewed by others
References
Aluoch AO, Whalen MM. Tributyltin-induced effects on MAP kinases p38 and p44/42 in human natural killer cells. Toxicology. 2005;209:263–77.
Aluoch AO, Odman-Ghazi SO, Whalen MM. Alteration of an essential NK cell signaling pathway by low doses of tributyltin in human natural killer cells. Toxicology. 2006;224:229–37.
Aluoch AO, Odman-Ghazi SO, Whalen MM. Pattern of MAP kinases p44/42 and JNK activation by non-lethal doses of tributyltin in human natural killer cells. Arch Toxicol. 2007;81:271–7.
Anderson NG, Maller JL, Tonks NK, Sturgill TW. Requirement for integration of signals from two distinct phosphorylation pathways for activation of MAP kinase. Nature. 1990;343:651–3.
Bajpai A, Brahmi Z. Target cell-induced inactivation of cytolytic lymphocytes. Role and regulation of CD45 and calyculin A-inhibited phosphatase in response to interleukin-2. J Biol Chem. 1994;269:18864–9.
Biron CA. Activation and function of natural killer cell responses during viral infections. Curr Opin Immunol. 1997;9:24–34.
Camps M, Nichols A, Arkinstall S. Dual specificity phosphatases: a gene family for control of MAP kinase function. FASEB J. 2000;14:6–16.
Catlin R, Shah H, Bankhurst AD, Whalen MM. Dibutyltin exposure decreases granzyme B and perforin in human natural killer cells. Environ Toxicol Pharmacol. 2005;20:395–403.
Chan G, Hanks T, Fisher K-D. Vav-1 regulates NK T cell development and NK cell cytotoxicity. Eur J Immunol. 2001;31:2403–10.
Derijard B, Raingeaud J, Barret T, Wu IH, Han J, Ulevitch RJ, et al. Independent human MAP-kinase signal transduction pathways defined by MEK and MKK isoforms. Science. 1995;267:682–5.
Djeu JY, Jiang K, Wei S. A view to a kill: signals triggering cytotoxicity. Clin Cancer Res. 2002;8:636–40.
Dudimah FD, Gibson C, Whalen MM. Effect of Dibutyltin (DBT) on ATP levels in human natural killer cells. Environ Toxicol. 2007;22:117–23.
Dudimah FD, Griffey D, Wang X, Whalen MM (2009). Activation of p44/42 MAPK plays a role in the TBT-induced loss of human natural killer (NK) cell function. Cell Biol Toxicol (in press)
Ema M, Iwase T, Iwase Y, Oyama N, Ogawa Y. Change of embryotoxic susceptibility to di-n-butyltin dichloride in cultured rat embryos. Arch Toxicol. 1996;70:742–8.
Epstein RL, Phillippo ET, Harr R, Koscinski W, Vosco G. Organotin residue determination in poultry and turkey sample survey in the United States. J Agric Food Chem. 1991;39:917–21.
Forsyth DS, Weber D, Barlow L. The determination of organotin compounds in fruit juices using gas chromatography-atomic absorption spectrometry. Appl Organomet Chem. 1992a;6:579–85.
Forsyth DS, Weber D, Cldroux C. Determination of butyltin, cyclohexyltin and phenyltin compounds in beers and wines. Food Addit Contam. 1992b;9:161–9.
Gismondi A, Jacobelli J, Mainiero F, Paolini R, Piccoli M, Frati L, et al. Cutting edge: functional role for proline-rich tyrosine kinase 2 in NK cell-mediated natural cytotoxicity. J Immunol. 2000;164:2272–6.
Gomez N, Cohen P. Dissection of the protein kinase cascade by which nerve growth factor activates MAP kinases. Nature. 1991;353:170–3.
Han J, Lee JD, Jiang Y, Li Z, Feng L, Ulevitch RJ. Characterization of the structure and function of a novel MAP kinase kinase (MKK6). J Biol Chem. 1996;271:2886–91.
Ishihara H, Martin BL, Brautigan DL, Karaki H, Ozaki H, Kato Y, et al. Calyculin A and okadaic acid: inhibitors of protein phosphatase activity. Biochem Biophys Res Commun. 1989;159:871–7.
Jewett A, Bonavida B. Target induced energy of natural killer cytotoxic function is restricted to the NK-target conjugate subset. Cell Immunol. 1995;160:91–7.
Kannan K, Falandyz J. Butyltin residues in sediment, fish, fish-eating birds harbor porpoise and human tissues from the Polish coast of the Baltic Sea. Mar Pollut Bull. 1997;34:203–7.
Kannan K, Guruge KS, Thomas NJ, Tanabe S, Giesy JP. Butyltin residues in southern sea utters (Enhydra lutris nereis) found dead near California coastal waters. Environ Sci Technol. 1998;32:1169–75.
Kannan K, Senthilkumar K, Giesy JP. Occurrence of butyltin compounds in human blood. Environ Sci Technol. 1999;33:1776–9.
Mao L, Yang L, Arora A, Choe ES, Zhang G, Liu Z, et al. Role of protein phosphatase 2A in mGluR5-regulated MEK/ERK phosphorylation in neurons. J Biol Chem. 2005;280:12602–10.
Merkord J, Jonas L, Weber H, Kroning G, Nizze H. Acute interstitial pancreatitis in rats induced by dibutyltin dichloride (DBDC) pathogenesis and natural cause of lesions. Pancreas. 1997;15:392–401.
Nakashima H, Hori S, Nakazawa H. Determination of dibutyltin and dioctyltin compounds in PVC food containers, wrappage and clothes by reversed phase HPLC with column switching. Eisei Kagaku. 1990;36:155–220.
Nielsen JB, Strand J. Hepatic deposition of butyltin in humans. Metal Ions Biol Med. 2002;7:185–8.
Noda T, Morita S, Bab A. Teratogenic effects of various di-n-butyltins with different anions and butyl(3-hydroxyl) tin dilaurate in rats. Toxicology. 1993;85:149–60.
Odman-Ghazi SO, Hatcher F, Whalen MM. Expression of functionally relevant cell surface markers in dibutyltin-exposed human natural killer cells. Chem Biol Interact. 2003;146:1–18.
Payne DM, Rossomando AJ, Martino P, Erickson AK, Her JH, Shabanowitz J, et al. Identification of the regulatory phosphorylation sites in pp 42/mitogen-activated protein kinase (MAP kinase). EMBO J. 1991;10:885–92.
Pearson G, Robinson F, Beers Gibson T, Xu B-E, Karandikar M, Berman K, et al. Mitogen-activated protein (MAP) kinase pathways: regulation and physiological functions. Endocr Rev. 2001;22:153–83.
Pieters RH, Bol M, Seinen W, Penninks AH. Cellular and molecular aspects of organotin-induced thymus atrophy. Hum Exp Toxicol. 1994;12:876–9.
Roper WL (1992) “Toxicological profile for tin”. U.S. department of health and human services, agency for toxic substances and disease registry
Sadiki A-I, Williams DT, Carrier R, Thomas B. Pilot study on the contamination of drinking water by organotin compounds from PVC materials. Chemosphere. 1996;32:2389–98.
Trinchieri G. Biology of natural killer cell. Adv Immunol. 1989;47:187–376.
Trotta R, Puorro KA, Paroli M, Azzoni L, Abebe B, Eisenlohr LC, et al. Dependence of both spontaneous and antibody-dependent, granule exocytosis-mediated NK cell cytotoxicity on extracellular signal-regulated kinases. J Immunol. 1998;161:6648–56.
Trotta R, Fettuciari K, Azzoni L, Abebe B, Puorro KA, Eisenlohr LC, et al. Differential role of p38 and c-Jun N-terminal kinase 1 mitogen-activated protein kinases in NK cell cytotoxicity. J Immunol. 2000;165:1782–9.
Vivier E, Nunes JA, Vely F. Natural killer cell signaling pathways. Science. 2004;306:1517–9.
Wei S, Gamero AM, Liu JH, Daulton AA, Valkov NI, Trapani JA, et al. Control of lytic function by mitogen-activated protein kinase/extracellular regulatory kinase 2 (ERK2) in a human natural killer cell line: identification of perforin and granzyme B mobilization by functional ERK2. J Exp Med. 1998;187:1753–65.
Whalen MM, Loganathan BG, Kannan K. Immunotoxicity of environmentally relevant concentrations of butyltins on human natural killer cells in vitro. Environ Res. 1999;81:108–16.
Whalen MM, Green SA, Longanathan BG. Brief butyltin exposure induces irreversible inhibition of the cytotoxic function on human natural killer cells, In vitro. Environ Res. 2002;88:19–29.
Yamada S, Fuji Y, Mikami E, Kawamura N, Hayakawa J. Small-scale survey of organotin compounds in household commodities. J AOAC Int. 1993;76:436–41.
Acknowledgment
This research was supported by Grant S06GM008092-34 from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Odman-Ghazi, S.O., Abraha, A., Isom, E.T. et al. Dibutyltin activates MAP kinases in human natural killer cells, in vitro. Cell Biol Toxicol 26, 469–479 (2010). https://doi.org/10.1007/s10565-010-9157-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-010-9157-3