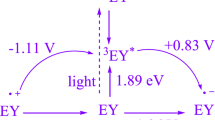
Abstract
A visible light promoted, photoredox catalysed, green one-pot approach for the synthesis of cyclohexyloxyphenethylpyridinoneshas been developed. New synthetic approach for the preparation of tailor-made synthesis of cyclohexyloxyphenethylpyridinones compounds are in extremely high demand as they display significant potent activity against wildtype HIV1 strains as well as on mutant strains. Herein, we have designed an efficient photo-induced synthetic strategy to obtain the target compound with higher yield.
Graphical Abstract





Similar content being viewed by others
References
Sinoussi FB, Chermann JC, Rey F, Nugeyre MT, Chamaret S, Gruest J, Dauguet C, Blin CA, Brun FV, Rouzioux C, Rozenbaum W, Montagnier L (1983) Science 220:868–871
Schupbach J, Popovic M, Gilden RV, Gonda MA, Sarngadharan MG, Gallo RC (1984) Science 224:503–505
Tang J, Maddali K, Dreis CD, Sham YY, Vince R, Pommier Y, Wang Z (2011) Bioorg Med Chem Lett 21:2400–2402
Kyenda NLK, Lubbe MS, Serfontein JH, Truter I (2008) J Clin Pharm Ther 33:393–400
Tronchet JM, Seman M (2003) Curr Top Med Chem 3:1496–1511
Tarby CM (2004) Curr Top Med Chem 4:1045–1057
Kelly TA, Proudfoot JR, McNeil DW, Patel UR, David E, Hargrave DK, Grob PM, Cardozo M, Agarwal A, Adam J (1995) J Med Chem 38:4839–4847
Romero DL, Olmsted RA, Poel TJ, Morge RA, Biles C, Keiser BJ, Kopta LA, Friis JM, Hosley JD, Stefanski KJ, Wishka DG, Evans DB, Morris J, Stehle RG, Sharma SK, Yagi Y, Voorman RL, Adams WL, Tarpley WG, Thomas RC (1996) J Med Chem 39:3769–3789
Das K, Clark AD, Lewi PJ, Heeres J, de Jonge MR, Koymans, Lucien MH, Vinkers HM, Daeyaert F, Ludovici DW, Kukla MJ, Corte BD, Kavash RW, Ho CY, Ye H, Lichtenstein MA, Andries K, Pauwels R, de Béthune MP, Boyer PL, Clark P, Hughes SH, Janssen, Paul AJ, Arnold E (2004) J. Med. Chem47: 2550–2560.
De Corte BL (2005) J Med Chem 48:1689–1696
Lansdon EB, Brendza KM, Hung M, Wang R, Mukund S, Jin D, Birkus G, Kutty N, Liu XH (2010) Med Chem 53:4295–4299
Benjahad A, Guillemont J, Andries K, Nguyen CH, Grierson DS (2003) Bioorg Med Chem Lett 13:4309–4312
Benjahad A, Courte K, Guillemont J, Mabire D, Coupa S, Poncelet A, Csoka I, Andries K, Pauwels R, De Bethune MP, Monneret C, Bisagni E, Nguyen CH, Grierson DS (2004) J Med Chem 47:5501–5514
Benjahad A, Croisy M, Monneret C, Bisagni E, Mabire D, Coupa S, Poncelet A, Csoka I, Guillemont J, Meyer C, Andries K, Pauwels R, De Be ́thune MP, Himmel DM, Das K, Arnold E, Nguyen CH, Grierson DS (2005) J. Med. Chem 48: 1948–1964.
Xu B, Lumb JP, Arndtsen BA (2015) Angew. Chem., Int. Ed54: 4208.
Tiwari J, Saquib M, Singh S, Tufail F, Singh M, Singh J, Singh J (2016) Green Chem 18:3221–3231
Sun X, Li X, Song S, Zhu Y, Liang YF, Jiao N (2015) J Am Chem Soc 137:6059
Prier CK, Rankic DA, Macmillan DWC (2013) Chem Rev 113:5322–5363
Twilton J, Le C, Zhang P (2017) Nat Rev Chem 1:0052
Toth BL, Tischler O, Novak Z (2016) Tetrahedron Lett 57:4505
Wang Y, Liu A, Ma D, Li S, Lu C, Li T, Chen C (2018) Catalysts 8:355
Yang HM, Liu ML, Tu JW, Stempel EM, Campbell MG, Chuang GJ (2020) J Org Chem 85:2040
Graml A, GhoshI, Konig B (2017) J. Org. Chem 82: 3552.
Zhang X, Zhu P, Zhang R, Li X, Yao T (2020) J Org Chem 85:9503
Cheng WM, Shang R (2020) ACS Catal 10:9170
Tang J, Grampp G, Liu Y, Wang BX, Tao FF, Wang LJ, Liang XZ, Xiao HQ, Shen YMJ (2015) Org Chem 80:2724
Kancherla R, Muralirajan K, Sagadevan A, Rueping M (2019) Trends Chem 1:510–638
Chen YZ, Wang DH, Chen B, Zhong JJ, Tung CH, Wu LZ (2012) J Org Chem 77:6773–6777
Parasram M, Gevorgyan V (2017) Chem Soc Rev 46:6227–6240
Yasu Y, Koike T, Akita M (2013) Chem Commun 49:2037–2039
Fan XZ, Rong JW, Wu HL, Zhou Q, Deng HP, Tan JD, Xue CW, Wu LZ, Tao HR, Wu J (2018) Angew Chem 57:8514–8518
Li X, Gu X, Li Y, Li P (2014) ACS Catal 4:1897–1900
Fan W, Yang Q, Xu F, Li P (2014) J Org Chem 79:10588–10592
Kaur S, Zhao G, Busch E, Wang T (2019) Org Biomol Chem 17:1955–1961
Ni C, Chen W, Jiang C, Lu H (2020) New J Chem 44:313–316
Zhang W, Xiang XX, Chen J, Yang C, Pan YL, Cheng JP, Meng Q, Li X (2020) Nat Commun 11:638
Teng L, Liu X, Guo P, Yu Y, Cao H (2020) Org Lett 22:3841–3845
Zhang Y, Ye C, Li S, Ding A, Gu G, Guo H (2017) RSC Adv 7:13240–13243
Srivastava V, Singh PK, Singh PP (2014) Croat Chem Acta 87:91–95
Srivastava V, Singh PK, Singh PP (2014) Chem Heterocycl Compd 50:573–578
Srivastava V, Singh PK, Singh PP (2015) Croat Chem Acta 88:59–65
Srivastava V, Singh PK, Singh PP (2015) Croat Chem Acta 88:227–233
Srivastava V, Singh PK, Singh PP (2016) Asian J Chem 28:2159–2163
Srivastava V, Singh PK, Singh PP (2016) Rev Roum Chem 61:755–761
Srivastava V, Singh PK, Singh PP (2017) Croat Chem Acta 90:435–441
Srivastava V, Singh PK, Kanaujia S, Singh PP (2018) New J Chem 42:688–691
Singh PK, Singh PP, Srivastava V (2018) Croat Chem Acta 91:383–387
Srivastava V, Singh PK, Singh PP (2019) Tetrahedron Lett 60:40–43
Srivastava V, Singh PK, Singh PP (2019) Tetrahedron Lett 60:1333–1336
Srivastava V, Singh PK, Singh PP (2019) Tetrahedron Lett 60:151041
Srivastava V, Singh PK, Singh PP (2020) Rev Roum Chem 65:221–226
Tivari S, Singh PK, Singh PP, Srivastava V (2022) RSC Adv 12:35221–35226
Singh SP, Srivastava V, Singh PK, Singh PP (2023) Tetrahedron 132:133245
Mishara M, Srivastava V, Singh PK, Singh PP (2023). Croat Chem Acta. https://doi.org/10.5562/cca3927
Beg MZ, Singh PK, Singh PP, Srivastava M, Srivastava V (2023). Mol Diversity. https://doi.org/10.1007/s11030-022-10595-2
Srivastava V, Singh PK, Srivastava A, Singh PP (2020) RSC Adv 10:20046
Srivastava A, Singh PK, Ali A, Singh PP, Srivastava V (2020) RSC Adv 10(2020):39495
Srivastava V, Singh PK, Srivastava A, Singh PP (2020) RSC Adv 11:14251–14259
Srivastava V, Singh PP (2021) Org Biomol Chem 19:313–321
Singh PP, Singh PK, Beg MZ, Kashyap A, Srivastava V (2021) Synth Commun 51:3033–3058
Srivastava V, Singh PK, Srivastava A, Sinha S, Singh PP (2021) Photochemistry 1:237–246
Srivastava V, Singh PK, Tivari S, Singh PP (2022) Org Chem Front 9:1485–1507
Srivastava V, Singh PK, Singh PP (2022) J Photochem Photobiol C 50:100488
Srivastava V, Singh PP (2022) RSC Adv 12:18245–18265
Singh PP, Sinha S, Pandey G, Srivastava V (2022) RSC Adv 12:29826–29839
Singh PP, Singh PK, Srivastava V (2022) Org Chem Front 10:216–236
Huang X, Qin F, Chen X, Liu Y, Zou H (2004) J Chromatogr B 804:13–18
Meyers AI, Slade J, Smith RK, Mihelich ED (1979) J Org C’hem 44(13):2247–2249
Udoikono AD, Louis H, Eno EA, Agwamba EC, Unimuke TO, Igbalagh AT, Edet HO, Odey JO, Adeyinka AS (2022) J Photochem Photobiol 10:100116
Mujwar S (2021) Comput Biol Med 136:104748
Pandey AK, Shukla DV, Mishra VN, Singh V, Yadav OP, Dwivedi A (2022) J Indian Chem Soc 99:100396
Mujwar S, Kumar V (2020) Assay Drug Dev Technol 18:318–327
Douche D, Sert Y, Brandán SA, Kawther AA, Bilmez B, Dege N, Louzi AE, Bougrin K, Karrouchi K, Himmi B (2021) J Mol Struct 1232:130005
Patterson AW, Wood WJ, Hoensby M, Lesley S, Spraggon G, Ellman JA (2006) J Med Chem 49:6298–6307
Roy DR, Parthasarathi R, Maity B, Subramanian V, Chattaraj PK (2005) Bioorg Med Chem 13:3405–3412
Pettersen EF, Goddard TD, Huang CC, Couch GS, Greenblatt DM, Meng MC, Ferrin TE (2005) J Comput Chem 25:1605–1612
Li H, Wang H, Kang S, Silverman RB, Poulos TL (2016) Biochemistry 55:3702–3707
Daina A, Michielin O, Zoete V (2014) J Chem Inf Model 54:3284–3301
Lipinski CA, Lombardo F, Dominy BW, Feeney PJ (1997) Adv Drug Delivery Rev 23:3–25
Acknowledgements
The authors are grateful for financial support provided by University Grants Commission India (UGC start-up Project No. F.30-461/2019, BSR).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest among them.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Srivastava, V., Tivari, S., Singh, P.K. et al. Photocatalysed Synthesis and Structure Activity Evaluation of Cyclohexyloxyphenethylpyridinones as Potent HIV-1 Inhibitor. Catal Lett 154, 771–779 (2024). https://doi.org/10.1007/s10562-023-04345-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-023-04345-8