Abstract
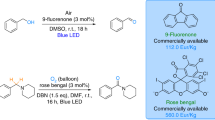
A novel photocatalytic cyclization reaction was developed for the synthesis of quinazolinones from o-aminobenzamides and in-situ generated aldehydes from alcohols using 9-fluorenone as the photocatalyst through a "one-pot" process. Furthermore, alcohols are perfect alternatives to aldehydes due to some unique advantages, such as being green, less toxic, available, and economical. The present protocol showed good tolerance for various substrates and could afford a range of quinazolinones (29 examples) up to 91% under ambient conditions.
Graphical Abstract






Similar content being viewed by others
References
Tietze LF, Modi A (2000) Med Res Rev 20(4):304–322
Godula K, Sames D (2006) Science 312(5770):67–72
Belenkii LI, Gramenitskaya VN, Evdokimenkova YB (2011). In: Katritzky AR (ed) Advances in Heterocyclic Chemistry. Academic press, Cambridge
Cao SL, Feng YP, Jiang YY, Liu SY, Ding GY, Li RT (2005) Bioorg Med Chem Lett 15(7):1915–1917
Kumar D, Jacob MR, Reynolds MB, Kerwin SM (2002) Bioorg Med Chem 10(12):3997–4004
Rida SM, Ashour FA, El-Hawash SAM, Badr H, Shalaby MA (2005) Eur J Med Chem 40(9):949–959
Murty MSR, Ram KR, Rao RV, Yadav JSU, Murty SN, Kumar KP (2011) Med Chem Res 20(5):626–636
Horton DA, Bourne GT, Smythe ML (2003) Chem Rev 103(3):893–930
Pawar OB, Chavan FR, Sakate SS, Shinde ND (2010) Chinese J Chem 28(1):69–71
Zhao D, Wang T, Li JX (2014) Chem Commun 50(49):6471–6474
Tavakoli-Hoseini N, Davoodnia AA (2011) Chinese J Chem 29(8):1685–1688
Mohammadi AA, Sadat-Hossini SS (2011) Chinese J Chem 29(9):1982–1984
Hioki H, Matsushita K, Nakamura S, Horiuchi H, Kubo M, Harada K, Fukuyama YJ (2008) J Comb Chem 10(5):620–623
Cheng R, Guo T, Daisy ZN, Du YF, Zhao K (2013) Synthesis 45(21):998–3006
Chen JX, Su WK, Wu HY, Liu MC, Jin C (2007) Green Chem 9(9):972–975
Zhan D, Li T, Wei H, Weng W, Ghandi K, Zeng QA (2013) RSC Adv 3(24):9325–9329
Roy AD, Subramanian A, Roy R (2006) J Org Chem 71(1):382–385
Rachakonda S, Pratap PS, Rao MVB (2012) Synthesis 44(13):2065–2069
Abdel-Jalil RJ, Voelter W, Saeed M (2004) Tetrahedron Lett 45(17):3475–3476
Li F, Lu L, Ma J (2015) Org Chem Front 2(12):1589–1597
Gong W, Chen X, Jiang H, Chu DD, Cui Y, Liu Y (2019) J Am Chem Soc 141(18):7498–7508
Prakash M, Jayakumar S, Kesavan V (2013) Synthesis 45(16):2265–2272
Zhou J, Fang J (2011) J Org Chem 76(19):7730–7736
Hikawa H, Ino Y, Suzuki H, Yokoyama Y (2012) J Org Chem 77(16):7046–7051
Watson JAA, Maxwell C, Williams JMJ (2012) Org Biomol Chem 10(2):240–243
Zhang Z, Wang M, Zhang C, Zhang Z, Lu J, Wang F (2015) Chem Commun 51(44):9205–9207
Hu Y, Chen L, Li B (2016) RSC Adv 6(69):65196–65204
Das S, Sinha S, Samanta D, Chakraborty G, Brandao P, Paul ND (2019) J Org Chem 84(16):10160–10171
Ge W, Zhu X, Wei Y (2013) RSC Adv 3(27):10817–10822
Xie Z, Lan J, Zhu H, Lei G, Jiang G, Le Z (2021) Chinese Chem Lett 32(4):1427–1431
Hamad H, Bailon-Garcia E, Maldonado-Hodar FJ, Perez-Cadenas AF, Carrasco-Marin F, Morales-Torres S (2019) Appl Catal B: Environ 241:385–392
Hamad H, Bailón-García E, Morales-Torres S, Carrasco-Marín F, Pérez-Cadenas AF, Maldonado-Hódar FJ (2020) Nanomaterials Basel. 10(4):729
Qi MY, Conte M, Anpo M, Tang ZR, Xu YJ (2021) Chem Rev 121(21):13051–13085
Narayanam JMR, Stephenson CRJ (2011) Chem Soc Rev 40(1):102–113
Zhu C, Yue H, Chu L, Rueping M (2020) Chem Sci 11(16):4051–4064
Ou W, Zou R, Han M, Yu L, Su C (2020) Chinese Chem Lett 31(7):1899–1902
Zhou R, Goh YY, Liu H, Tao H, Li L, Wu J (2017) Angew Chem Int Ed 56(52):16621–16625
Han C, Tang ZR, Liu J, Jin S, Xu YJ (2019) Chem Sci 10(12):3514–3522
Lina Q, Lia YH, Qi MY, Li JY, Tang ZR, Anpo M, Yamada YMA, Xu YJ (2020) Appl Catal B-Environ 271:118946
Yang MQ, Xu YJ (2013) Chem Chem Phys 15(44):19102–19118
Li YH, Zhang F, Chen Y, Li JY, Xu YJ (2020) Green Chem 22(1):163–169
Zhou R, Ma L, Yang X, Cao J (2021) Org Chem Front 8(3):426–444
Qi MY, Conte M, Tang ZR, Xu YJ (2022) ACS Nano 16(10):17444–17453
Li JY, Li YH, Qi MY, Lin Q, Tang ZR, Xu YJ (2020) ACS Catal 10(11):6262–6280
Li YH, Qi MY, Tang ZR, Xu YJ (2022) J Phys Chem C 126(4):1872–1880
Qi MY, Li YH, Anpo M, Tang ZR, Xu YJ (2020) ACS Catal 10(23):14327–14335
Yang MQ, Xu YJ (2013) Phys Chem Chem Phys 15(44):19102–19118
Chan AY, Perry IB, Bissonnette NB, Buksh BF, Edwards GA, Frye LI, Garry OL, Lavagnino MN, Li BX, Liang YF, Mao E, Millet A, Oakley JV, Reed NL, Sakai HA, Seath CP, MacMillan DWC (2021) Chem Rev 122(2):485–1542
Lang X, Ma W, Chen C, Ji H, Zhao J (2014) Acc Chem Res 47(2):355–363
Chen YZ, Wang ZU, Lu HJ, Yu SH, Jiang HL (2017) J Am Chem Soc 139(5):2035–2044
Zhao QQ, Hu XQ, Yang MN, Chen JR, Xiao WJ (2016) Chem Commun 52(86):12749–12752
Meng C, Yang K, Fu XZ, Yuan RS (2015) ACS Catal 5(6):3760–3766
Zhou R, Liu R, Zhang K, Han L, Zhang H, Gao W, Li R (2017) Chem Commun 53(51):6860–6863
Ravelli D, Fagnoni M, Albini A (2013) Chem Soc Rev 42(1):97–113
Romero NA, Nicewicz DA (2016) Chem Rev 116(17):10075–10166
Srivastava V, Singh PP (2017) RSC Adv 7(50):31377–31392
Peng JB, Qi X, Wu XF (2016) Chemsuschem 9(17):2279–2283
Wang YF, Xu WG, Sun B, Yu QQ, Li TJ, Zhang FL (2019) J Org Chem 84(20):13104–13111
Xia JB, Zhu Z, Chen C (2013) J Am Chem Soc 135(46):17494–17500
Okamoto H, Yamaji M, Gohda S, Kubozono Y, Komura N, Sato K, Sugino H, Satake K (2011) Org Lett 13(10):2758–2761
Margrey KA, Nicewicz DA (2016) Acc Chem Res 49(9):1997–2006
Ali El-Remaily MA, Hamad HA, Soliman AM, Elhady OM (2021) Appl Organomet Chem 35(7):e6238
Fathy M, Hamad H (2016) RSC adv 6(9):7310–7316
Hamad H, Abd El-latif M, Kashyout AEH, Sadik W, Feteha M (2015) Process Saf Environ 98:390–398
Hamad H, Castelo-Quibén J, Morales-Torres S, Carrasco-Marín F, Pérez-Cadenas AF, Maldonado-Hódar FJ (2018) Materials 11(9):1766
Hamad H, Bailon-Garcia E, Morales-Torres S, Carrasco-Marin F, Perez-Cadenas AF, Maldonado-Hodar FJ (2018) J Environ Chem Eng 6(4):5032–5041
Jiang X, Tang T, Wang JM, Chen Z, Zhu YM, Ji SJ (2014) J Org Chem 79(11):5082–5087
Xu W, Jin Y, Liu H, Jiang Y, Fu AH (2011) Org Lett 13(6):1274–1277
Pieterse L, Van der Walt MM, Terre’Blanche G (2020) Bioorg Med Chem Lett 30(16):127274
Balaji S, Balamurugan G, Ramesh R, Semeril D (2021) Organometallics 40(6):725–734
Heravi M, Montazeri N, Rahimzadeh M, Bakavoli M, Ghassemzadeh M (2004) Pol Jo Chem 78:2101–2103
Sylvain L, Marine H, Julien G, Isabelle SA, Laurent B, Christophe H, Vincent L, Corinne F, Thierry B (2015) Org Lett 17(7):1700–1703
Yao D, Jiang J, Zhang H, Huang Y, Huang J, Wang J (2021) Bioorg Med Chem Lett 47:128204
Abdullaha M, Mohammed S, Ali M, Kumar A, Vishwakarma RA, Bharate SB (2019) J Org Chem 84(9):5129–5140
Hao S, Yang J, Liu P, Xu J, Yang C, Li F (2021) Org Lett 23(7):2553–2558
Parua S, Das S, Sikari R, Sinha S, Paul ND (2017) J Org Chem 82(14):7165–7175
Ma B, Wang Y, Peng J, Zhu Q (2011) J Org Chem 76(15):6362–6366
Lee S, Sim J, Jo H, Viji M, Srinu L, Lee K, Lee H, Manjunatha V, Jung JK (2019) Org Biomole Chem 17(35):8067–8070
Feng Y, Li Y, Cheng G, Wang L, Cui X (2015) J Org Chem 80(14):7099–7107
Fang J, Zhou J (2012) Org Biomol Chem 10(12):2389–2391
Hikawa H, Nakayama T, Takahashi M, Kikkawa S, Azumaya I (2021) Adv Synth Catal 363(16):4075–4084
Hamad H, Elsenety MM, Sadik W, El-Demerdash AG, Nashed A, Mostafa A, Elyamny S (2022) Sci Rep-UK 12(1):1–20
Ali El-Remaily MA, Kamel MS, Halim SA, Shokr EK, Abdel-Ghany H, Hamad H (2023) Mater Chem Phys 293:126972
Ghosh I, Mukhopadhyay A, Koner AL, Samanta S, Nau WM, Moorthy JN (2014) Chem Chem Phys 16(31):16436–16445
Bains AK, Ankit Y, Adhikari D (2021) Org Lett 23(6):2019–2023
Acknowledgements
We are grateful for the financial support from the National Natural Science Foundation of China (22005179), and Natural Science Foundation of Shandong Province (ZR2020QB113, ZR2020MB018 and, ZR2021QB049).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Ren, J., Xiao, Q. et al. Photocatalytic One-Pot Synthesis of Quinazolinone Under Ambient Conditions. Catal Lett 153, 3771–3782 (2023). https://doi.org/10.1007/s10562-022-04266-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-022-04266-y