Abstract
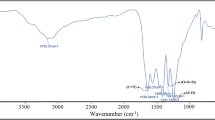
Design and synthesis of green and recyclable catalysts in order to improve the efficiency of organic chemistry reactions is a significant challenge. One of these reactions is the homocoupling reaction of terminal alkynes, known as the Glaser coupling reaction. In this study, first, graphitic carbon nitride (g-C3N4) nanosheets with high surface area and chemical stability were synthesized. Afterwards, the surface of this nano-substrate was modified with polyethyleneimine as a chelating agent to obtain g-C3N4/PEI. Finally, copper(I) was added to the nanocomposite, and the Cu(I)@g-C3N4/PEI catalyst was synthesized. The active site of copper(I) in this catalyst was calculated to be 0.16 mol in 100 g Cu(I)@g-C3N4/PEI. The synthesized catalyst was characterized by different methods such as FTIR, XRD, ICP, SEM, EDX, and mapping. Also, a deep eutectic solvent (a mixture of choline chloride and urea) was synthesized and used as a green solvent in this reaction. The reaction was performed at 100 °C for 30 min, and the yield was obtained as 90%. The advantages of this reaction include easy catalyst separation and reusability thanks to its heterogeneous nature, the use of a deep eutectic solvent as a green solvent, which results in a clean reaction with good to excellent efficiency, and finally short reaction time.
Graphical Abstract
















Similar content being viewed by others
References
Diederich F, Stang PJ, Tykwinski RR (2006) Acetylene chemistry: chemistry, biology and material science. Wiley, Hoboken
Kim YS, Jin SH, Kim SI et al (1989) Studies on the mechanism of cytotoxicities of polyacetylenes against L1210 cell. Arch Pharmacal Res 12(3):207
Mayer SF, Steinreiber A, Orru RV et al (2002) Chemoenzymatic asymmetric total syntheses of antitumor agents (3R, 9R, 10R)-and (3S, 9R, 10R)-panaxytriol and (R)-and (S)-falcarinol from panax g inseng using an enantioconvergent enzyme-triggered cascade reaction. J Org Chem 67(26):9115–9121
Tubío CR, Azuaje J, Escalante L et al (2016) 3D printing of a heterogeneous copper-based catalyst. J Catal 334:110–115
Singh M, Singh AS, Mishra N et al (2019) Benzotriazole as an efficient ligand in Cu-catalyzed Glaser reaction. ACS Omega 4(1):2418–2424
Martin RE, Diederich F (1999) Linear monodisperse π-conjugated oligomers: model compounds for polymers and more. Angew Chem Int Ed 38(10):1350–1377
Stefani HA, Costa IM, Zeni G (1999) Synthesis of polyacetylenic montiporic acids A and B. Tetrahedron Lett 40(52):9215–9217
Shi Shun AL, Tykwinski RR (2006) Synthesis of naturally occurring polyynes. Angew Chem Int Ed 45(7):1034–1057
Yun H, Chou TC, Dong H et al (2005) Total synthesis as a resource in drug discovery: the first in vivo evaluation of panaxytriol and its derivatives. J Org Chem 70(25):10375–10380
Siemsen P, Livingston RC, Diederich F (2000) Acetylenic coupling: a powerful tool in molecular construction. Angew Chem Int Ed 39(15):2632–2657
Cho EJ, Kim M, Lee D (2006) Metal-catalyzed bond reorganization of 1, 3-diynes. ChemInform. https://doi.org/10.1002/chin.200647060
Atobe S, Sonoda M, Suzuki Y et al (2013) Palladium-catalyzed oxidative homocoupling reaction of terminal acetylenes using trans-bidentatable 1-(2-pyridylethynyl)-2-(2-thienylethynyl) benzene. Res Chem Intermed 39(1):359–370
Bettanin L, Botteselle GV, Godoi M et al (2014) Green synthesis of 1, 3-diynes from terminal acetylenes under solvent-free conditions. Green Chem Lett Rev 7(2):105–112
Crowley JD, Goldup SM, Gowans ND et al (2010) An unusual nickel−copper-mediated alkyne homocoupling reaction for the active-template synthesis of [2] rotaxanes. J Am Chem Soc 132(17):6243–6248
Hilt G, Hengst C, Arndt M (2009) The unprecedented cobalt-catalysed oxidative glaser coupling under reductive conditions. Synthesis 2009(03):395–398
Cheng G, Zhang H, Cui X (2014) Copper(I)-catalyzed homo-coupling of terminal alkynes at room temperature under solvent and base free conditions using O2 as an oxidant. RSC Adv 4(4):1849–1852
Evano G, Blanchard N (2013) Copper-mediated cross-coupling reactions. Wiley, Hoboken
Hussain I, Capricho J, Yawer MA (2016) Synthesis of biaryls via ligand-free suzuki-miyaura cross-coupling reactions: a review of homogeneous and heterogeneous catalytic developments. Adv Synth Catal 358(21):3320–3349
Veisi H, Hamelian M, Hemmati S (2014) Palladium anchored to SBA-15 functionalized with melamine-pyridine groups as a novel and efficient heterogeneous nanocatalyst for Suzuki-Miyaura coupling reactions. J Mol Catal A 395:25–33
Vidal C, García-Álvarez J (2019) Metal-promoted organic transformation in DES. In: Ramón DJ, Guillena G (eds) Deep eutectic solvents: synthesis, properties, and applications. Wiley, Hoboken, pp 171–186
Climent MJ, Corma A, Iborra S (2012) Homogeneous and heterogeneous catalysts for multicomponent reactions. RSC Adv 2(1):16–58
Lu W, Sun W, Tan X et al (2019) Stabilized Cu/Cu2O nanoparticles on rGO as an efficient heterogeneous catalyst for Glaser homo-coupling. Catal Commun 125:98–102
Kumar A, Xu Q (2018) Two-dimensional layered materials as catalyst supports. ChemNanoMat 4(1):28–40
Li Y, Li YL, Sa B et al (2017) Review of two-dimensional materials for photocatalytic water splitting from a theoretical perspective. Catal Sci Technol 7(3):545–559
Deng D, Novoselov K, Fu Q et al (2016) Catalysis with two-dimensional materials and their heterostructures. Nat Nanotechnol 11(3):218–230
Reddy KR, Reddy CV, Nadagouda MN et al (2019) Polymeric graphitic carbon nitride (g-C3N4)-based semiconducting nanostructured materials: synthesis methods, properties and photocatalytic applications. J Environ Manage 238:25–40
Dadashi-Silab S, Tasdelen MA, Kiskan B et al (2014) Photochemically mediated atom transfer radical polymerization using polymeric semiconductor mesoporous graphitic carbon nitride. Macromol Chem Phys 215(7):675–681
Ansari MB, Min BH, Mo YH et al (2011) CO2 activation and promotional effect in the oxidation of cyclic olefins over mesoporous carbon nitrides. Green Chem 13(6):1416–1421
Wang X, Maeda K, Thomas A et al (2009) A metal-free polymeric photocatalyst for hydrogen production from water under visible light. Nat Mater 8(1):76–80
Chen X, Zhang J, Fu X et al (2009) Fe-g-C3N4-catalyzed oxidation of benzene to phenol using hydrogen peroxide and visible light. J Am Chem Soc 131(33):11658–11659
Goettmann F, Fischer A, Antonietti M et al (2006) Chemical synthesis of mesoporous carbon nitrides using hard templates and their use as a metal-free catalyst for Friedel-Crafts reaction of benzene. Angew Chem Int Ed 45(27):4467–4471
Wang X, Liu Q, Liu J et al (2017) 3D self-assembly polyethyleneimine modified graphene oxide hydrogel for the extraction of uranium from aqueous solution. Appl Surf Sci 426:1063–1074
Chen L, Li Z, Wu G et al (2018) Ultra-strong polyethyleneimine-graphene oxide nanocomposite film via synergistic interactions and its use for humidity sensing. Compos A 115:341–347
Peng HL, Zhong FY, Zhang JB et al (2018) Graphitic carbon nitride functionalized with polyethylenimine for highly effective capture of carbon dioxide. Ind Eng Chem Res 57(32):11031–11038
Bessa A, Henriques B, Gonçalves G et al (2020) Graphene oxide/polyethyleneimine aerogel for high-performance mercury sorption from natural waters. Chem Eng J 398:125587
Xu H, Wu K, Tian J et al (2018) Recyclable Cu/C3N4 composite catalysed homo-and cross-coupling of terminal alkynes under mild conditions. Green Chem 20(4):793–797
Hooshmand SE, Afshari R, Ramón DJ et al (2020) Deep eutectic solvents: cutting-edge applications in cross-coupling reactions. Green Chem 22(12):3668–3692
Shaabani A, Hooshmand SE (2016) Choline chloride/urea as a deep eutectic solvent/organocatalyst promoted three-component synthesis of 3-aminoimidazo-fused heterocycles via Groebke–Blackburn–Bienayme process. Tetrahedron Lett 57(3):310–313
Shishov A, Bulatov A, Locatelli M et al (2017) Application of deep eutectic solvents in analytical chemistry. Rev Microchemical J 135:33–38
Abbott AP, Capper G, Davies DL et al (2003) Novel solvent properties of choline chloride/urea mixtures. Chem Commun 1:70–71
Feiz E, Mahyari M, Ghaieni HR et al (2020) Visible-light-induced controlled ATRP by modified N-rich holey carbon nitride nanosheets in natural solvent. J Mol Liq 318:114320
Nebra N, García-Álvarez J (2020) Cu-catalyzed organic reactions in deep eutectic solvents (DESs). Copper Catal Org Syn. https://doi.org/10.1002/9783527826445.ch6
Hosseini SG, Khodadadipoor Z, Mahyari M (2018) CuO nanoparticles supported on three-dimensional nitrogen-doped graphene as a promising catalyst for thermal decomposition of ammonium perchlorate. Appl Organomet Chem 32(1):e3959
Mahyari M, Bide Y, Gavgani JN (2016) Iron (III) porphyrin supported on S and N co-doped graphene quantum dot as an efficient photocatalyst for aerobic oxidation of alcohols under visible light irradiation. Appl Catal A 517:100–109
Hajipour AR, Abrishami F (2013) Facile construction of biaryls by homocoupling of aryl halides. Org Prep Proced Int 45(3):227–231
Hajipour AR, Abrishami F (2012) Application of dimeric ortho-palladated complex of 2-methoxyphenethylamine in the Heck coupling reaction. Iranian J Catal 2(3):95–100
Yadav J, Reddy B, Reddy KB et al (2003) Glaser oxidative coupling in ionic liquids: an improved synthesis of conjugated 1, 3-diynes. Tetrahedron Lett 44(34):6493–6496
Selvarajan S, Suganthi A, Rajarajan M (2018) Fabrication of g-C3N4/NiO heterostructured nanocomposite modified glassy carbon electrode for quercetin biosensor. Ultrason Sonochem 41:651–660
Gao J (2016) The electrochemical analysis of cadmium in choline chloride/urea deep eutectic solvent electrolyte at carbon nanotubes modified electrode. Int J Electrochem Sci 11:6306–6314
Lakhi KS, Park DH, Al-Bahily K et al (2017) Mesoporous carbon nitrides: synthesis, functionalization, and applications. Chem Soc Rev 46(1):72–101
Yang L, Wang L, Xing M et al (2016) Silica nanocrystal/graphene composite with improved photoelectric and photocatalytic performance. Appl Catal B 180:106–112
Darkwah WK, Ao Y (2018) Mini review on the structure and properties (photocatalysis), and preparation techniques of graphitic carbon nitride nano-based particle, and its applications. Nanoscale Res Lett 13(1):1–15
Nabid MR, Bide Y, Ghalavand N (2015) Copper(I) ion stabilized on Fe3O4-core ethylated branched polyethyleneimine-shell as magnetically recyclable catalyst for ATRP reaction. J Appl Polym Sci. https://doi.org/10.1002/app.42337
Chen Z, Deng S, Wei H et al (2013) Polyethylenimine-impregnated resin for high CO2 adsorption: an efficient adsorbent for CO2 capture from simulated flue gas and ambient air. ACS Appl Mater Interfaces 5(15):6937–6945
Hu R, Gou H, Mo Z et al (2015) Highly selective detection of trace Cu2+ based on polyethyleneimine-reduced graphene oxide nanocomposite modified glassy carbon electrode. Ionics 21(11):3125–3133
Barceloux DG, Barceloux D (1999) Copper. J Toxicol Clin Toxicol 37(2):217–230
Li W, Jia X, Zhang B et al (2015) Fabrication of PEI grafted Fe3O4/SiO2/P (GMA-co-EGDMA) nanoparticle anchored palladium nanocatalyst and its application in Sonogashira cross-coupling reactions. New J Chem 39(4):2925–2934
Florindo C, Lima F, Ribeiro BD et al (2019) Deep eutectic solvents: overcoming 21st century challenges. Curr Opin Green Sustainable Chem 18:31–36
Yang H, Lv K, Zhu J et al (2017) Effect of mesoporous g-C3N4 substrate on catalytic oxidation of CO over Co3O4. Appl Surf Sci 401:333–340
Li JH, Liang Y, Xie YX (2005) Efficient palladium-catalyzed homocoupling reaction and Sonogashira cross-coupling reaction of terminal alkynes under aerobic conditions. J Org Chem 70(11):4393–4396
Yin L, Liebscher J (2007) Carbon-carbon coupling reactions catalyzed by heterogeneous palladium catalysts. Chem Rev 107(1):133–173
Schoergenhumer J, Waser M (2016) Transition metal-free dimerization of alkynes using hypervalent iodine reagents. Tetrahedron Lett 57(15):1678–1680
Yin K, Li CJ, Li J et al (2011) CuI-catalyzed homocoupling of terminal alkynes to 1, 3-diynes. Appl Organomet Chem 25(1):16–20
Jones CW (2010) On the stability and recyclability of supported metal-ligand complex catalysts: myths, misconceptions and critical research needs. Top Catal 53(13–14):942–952
Acknowledgements
Gratefully acknowledge the chemistry department of the Malek Ashtar University of Technology because of financial support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Abrishami, F., Soufi, A. & Mahyari, M. Cu(I)@g-C3N4/PEI: A New Heterogeneous Catalyst for Glaser Reaction in Deep Eutectic Solvent. Catal Lett 153, 2989–3002 (2023). https://doi.org/10.1007/s10562-022-04188-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-022-04188-9