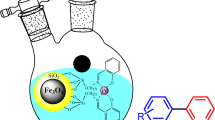
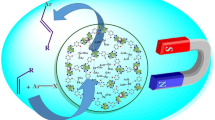
Abstract
A novel Pd-NHC functionalized magnetic Fe3O4@SBA-15@NHC-Pd was synthesized and used as an efficient heterogeneous catalyst in the Suzuki–Miyaura C–C bond formation reactions. The Fe3O4@SBA-15@NHC-Pd characterized by X-Ray Diffraction (XRD), X-ray Photoelectron Spectroscopy (XPS), Fourier Transform Infrared (FTIR) spectroscopy, Scanning Electron Microscopy (SEM), Transmission Electron Microscopy (TEM), Energy Dispersive X-ray analysis (EDX), Thermogravimetric Analysis (TGA), Differential Thermal Analysis (DTA). The Inductively Coupled Plasma-Optical emission spectroscopy (ICP-OES) analysis was used to determine the exact amount of Pd (0.33 wt%) in Fe3O4@SBA-15@NHC-Pd. The TEM images of the catalyst showed the existence of palladium nanoparticles immobilized in the catalyst's structure, while no reducing agent was used. The NHC moieties in the catalyst structure could be stabilize Pd(0) nanoparticles prevents agglomeration. The magnetic catalyst was effectively used in the Suzuki–Miyaura cross-coupling reaction of substituted phenylboronic acid derivatives with (hetero)aryl bromides in the presence of a K2CO3 at room temperature in aqueous media and magnetic catalyst could be simply extracted from the reaction mixture by an external magnet. Different aryl bromides were converted to coupled-products in excellent yields with spectacular TOFs values (up to 1,960,339 h−1); in the presence of 1 mg of Fe3O4@SBA-15@NHC-Pd catalyst (contains 3.1 × 10–6 mol% Pd) at room temperature in aqueous media. After reusability experiments, it is found that this catalyst was effectively used up to ten times in the reaction with almost consistent catalytic efficiency. A decrease in the activity of the 10th reused catalyst was found as 9%.
Graphic Abstract












Similar content being viewed by others
References
Chen Y, Chen HR, Guo LM, He QJ, Chen F, Zhou J, Feng J, Shi J (2010) ACS Nano 4:529–539. https://doi.org/10.1021/nn901398j
Shi D, Cho HS, Chen Y, Xu H, Gu H, Lian J, Wang W, Liu G, Huth C, Wang L, Ewing RC, Budko S, Pauletti GM, Dong Z (2009) Adv Mater 21:2170–2173. https://doi.org/10.1002/adma.200803159
Jafari AA, Mahmoudi H, Firouzabadi H (2015) RSC Adv 5:107474–107481. https://doi.org/10.1039/C5RA22909J
Zhang SJ, Liu XH, Zhou LP, Peng WJ (2012) Mater Lett 68:243–246. https://doi.org/10.1016/j.matlet.2011.10.070
Sing KSW (1985) Pure Appl Chem 57:603–619. https://doi.org/10.1351/pac198557040603
Yu L, Huang Y, Wei Z, Ding Y, Su C, Xu Q (2015) J Org Chem 80:8677–8683. https://doi.org/10.1021/acs.joc.5b01358
Begum T, Mondal M, Borpuzari MP, Kar R, Kalita G, Gogoi PK, Bora U (2017) Dalton Trans 46:539–546. https://doi.org/10.1039/C6DT03097A
Xu L, Liao J, Huang L, Ou D, Guo Z, Zhang H, Ge C, Gu N, Liu J (2003) Thin Solid Films 434:121–125. https://doi.org/10.1016/S0040-6090(03)00274-8
Zhang L, Li P, Li H, Wang L (2012) Catal Sci Technol 2:1859–1864. https://doi.org/10.1039/C2CY20162C
Keesara S, Parvathaneni S, Dussa G, Mandapati MR (2014) J Organomet Chem 765:31–38. https://doi.org/10.1016/j.jorganchem.2014.04.020
Marset X, De Gea S, Guillena G, Ramón DJ (2018) ACS Sustain Chem Eng 6:5743–5748. https://doi.org/10.1021/acssuschemeng.8b00598
Chen Y, Wang M, Zhang L, Liu Y, Han J (2017) RSC Adv 7:47104–47110. https://doi.org/10.1039/C7RA09947A
Nasrollahzadeh M, Issaabadi Z, Tohidi MM, Mohammad Sajadi S (2018) Chem Rec 18:165–229. https://doi.org/10.1002/tcr.201700022
Komiyama T, Minami Y, Furuya Y, Hiyama T (2018) Angew Chem 130:2005–2008. https://doi.org/10.1002/anie.201712081
Komiyama T, Minami Y, Furuya Y, Hiyama T (2018) Angew Chem Int Ed 57:1987–1990. https://doi.org/10.1002/anie.201712081
Asgari MS, Zonouzi A, Rahimi R, Rabbani M (2015) Orient J Chem 31:1537–1544. https://doi.org/10.13005/ojc/310331
Hoa ND, Duy NV, Safty SAE, Hieu NV (2015) J Nanomater 1:1–14. https://doi.org/10.1155/2015/972025
Vavsari VF, Ziarani GM, Badiei A (2015) RSC Adv 5:91686–91707. https://doi.org/10.1039/C5RA17780D
Ghorbani-Choghamarani A, Tahmasbi B, Noori N, Ghafouri-Nejad R (2017) J Iran Chem Soc 14:681–693. https://doi.org/10.1007/s13738-016-1020-x
Adimoolam MG, Amreddy N, Rao Nalam M, Sunkara MV (2018) J Magn Magn Mater 448:199–207. https://doi.org/10.1016/j.jmmm.2017.09.018
Saeedi MS, Tangestaninejad S, Moghadam M, Mirkhani V, Mohammadpoor-Baltork I, Khosropour AR (2013) Polyhedron 49:158–166. https://doi.org/10.1016/j.poly.2012.09.026
Tadic M, Kralj S, Lalatonne Y, Motte L (2019) Appl Surf Sci 476:641–646. https://doi.org/10.1016/j.apsusc.2019.01.098
Mal K, Chatterjee S, Bhaumik A, Mukhopadhyay C (2019) Chemistry Select 4:1776–1784. https://doi.org/10.1002/slct.201803708646
Nikoorazm M, Ghorbani-Choghamarani A, Noori N, Tahmasbi B (2016) Appl Organometal Chem 30:843–851. https://doi.org/10.1002/aoc.3512
Nikoorazm M, Ghorbani Choghamarani A, Panahi A, Tahmasbi B, Noori N (2018) J Iran Chem Soc 15:181–189. https://doi.org/10.1007/s13738-017-1222-x
Stevens PD, Li G, Fan J, Yen M, Gao Y (2005) Chem Commun 35:4435–4437. https://doi.org/10.1039/B505424A
Ranganath KVS, Kloesges J, Schfer A, Glorius F (2010) Angew Chem 122:7952–7956. https://doi.org/10.1002/ange.201002782
Lee JW, Cho DL, Shim WG, Moon H (2004) Korean J Chem Eng 21:246–251. https://doi.org/10.1007/BF02705405
Corma A, Kumar D (1998) Stud Surf Sci Catal 117:201–222
Sujandi S, Han SC, Han DS, Jin MJ, Park SE (2006) J Catal 243:410–419. https://doi.org/10.1016/j.jcat.2006.08.010
Firouzabadi H, Iranpoor N, Gholinejad M, Hoseini J (2011) Adv Synth Catal 353:125–132. https://doi.org/10.1002/adsc.201000390
Tang Y-Q, Lu J-M, Shao L-X (2011) J Organomet Chem 696:3741. https://doi.org/10.1016/j.jorganchem.2011.08.042
Touj N, Gürbüz N, Hamdi N, Yasar S, Ozdemir I (2018) Inorg Chim Acta 478:187–194. https://doi.org/10.1016/j.ica.2018.04.018
Touj N, Yasar S, Ozdemir N, Hamdi N, Ozdemir I (2018) J Organomet Chem 860:59–71. https://doi.org/10.1016/j.jorganchem.2018.01.017
Boztepe C, Künkül A, Yaşar S, Gürbüz N (2018) J Organomet Chem 872:123–134. https://doi.org/10.1016/j.jorganchem.2018.07.004
Dehimat ZI, Yaşar S, Tebbani D, Ozdemir I (2018) Inorg Chim Acta 469:325–334. https://doi.org/10.1016/j.ica.2017.09.048
Yaşar S, Akkoç M, Ozdemir N, Ozdemir I (2019) Turk J Chem 43:1622–1633. https://doi.org/10.3906/kim-1907-74
Akkoç M, İmik F, Dorcet V, Roisnel T, Bruneau C, Ozdemir I (2017) ChemistrySelect 2:5729–5734. https://doi.org/10.1002/slct.201701354
Karaca EO, Akkoç M, Yaşar S, Ozdemir I (2018) Arkivoc part v:230–239. https://doi.org/10.2820/ark.5550190.p010.519
Karaca EO, Akkoç M, Tahir MN, Arıcı C, Imik F, Gürbüz N, Yaşar S, Ozdemir I (2017) Tetrahedron Lett 58:3529–3532. https://doi.org/10.1016/j.tetlet.2017.07.096
Beletskaya IP, Cheprakov AV (2000) Chem Rev 100:3009–3066. https://doi.org/10.1021/cr9903048
Imik F, Yaşar S, Ozdemir I (2019) Inorg Chim Acta 495:118969. https://doi.org/10.1016/j.ica.2019.118969
Imik F, Yaşar S, Ozdemir I (2009) J Organomet Chem 896:162–167. https://doi.org/10.1016/j.jorganchem.2019.06.019
Boubakri L, Yasar S, Dorcet V, Roisnel T, Bruneau C, Hamdib N, Ozdemir I (2017) New J Chem 41:5105–5113. https://doi.org/10.1039/C7NJ00488E
Touj N, Yaşar S, Ozdemir N, Hamdi N, Ozdemir I (2018) J Organomet Chem 860:59–71. https://doi.org/10.1016/j.jorganchem.2018.01.017
Gu Z-S, Shao L-X, Lu J-M (2012) J Organomet Chem 700:132–134. https://doi.org/10.1016/j.jorganchem.2011.11.030
Liu J, Qiao SZ, Hu QH, Lu GQ (2011) Small 7:425–443. https://doi.org/10.1002/smll.201001402
Veisi H, Hameliana M, Hemmati S (2014) J Mol Catal A 395:25–33. https://doi.org/10.1016/j.molcata.2014.07.030
Le X, Dong Z, Jin Z, Wang Q, Ma J (2014) Catal Commun 53:47–52. https://doi.org/10.1016/j.catcom.2014.04.025
Nikoorazm M, Ghorbani F, Ghorbani-Choghamarani A, Erfani Z (2017) Chin J Catal 38:1413–1422. https://doi.org/10.1016/S1872-2067(17)62865-1
Khazaei A, Khazaei M, Nasrollahzadeh M (2017) Tetrahedron 73:5624–5633. https://doi.org/10.1016/j.tet.2017.05.054
Elazab HA, Siamaki AR, Moussaa S, Guptona BF, El-Shall MS (2015) Appl Catal A 491:58–69. https://doi.org/10.1016/j.apcata.2014.11.033
Tang W, Li J, Jin X, Sun J, Huang J, Li R (2014) Catal Commun 43:75–78. https://doi.org/10.1016/j.catcom.2013.09.001
You S, Xiao R, Liu H, Cai M (2017) New J Chem 41:13862–13870. https://doi.org/10.1039/C7NJ02969A
Shiri L, Tahmasbi B (2017) Phosphorus Sulfur Silicon Relat Elem 192:53–57. https://doi.org/10.1080/10426507.2016.1224878
Mohamadi Tanuraghaj H, Farahi M (2019) New J. Chem. 43:4823–4829. https://doi.org/10.1039/C8NJ06415F
Ghasemzadeh MS, Akhlaghinia B (2019) New J Chem 43:5341–5356. https://doi.org/10.1039/C9NJ00352E
Tahmasbi B, Ghorbani-Choghamarani A (2019) New J Chem 43:14485–14501. https://doi.org/10.1039/C9NJ02727K
Rafiee F, Mehdizadeh N (2018) Catal Lett 148:1345–1354. https://doi.org/10.1007/s10562-018-2363-y
Tai Z, Isaacs MA, Parlett CMA, Lee AF, Wilson K (2017) Catal Commun 92:56–60. https://doi.org/10.1016/j.catcom.2017.01.004
Keypour H, Saremi SG, Noroozi M, Veisi H (2017) Appl Organomet Chem 31:3558. https://doi.org/10.1002/aoc.3558
Nuri A, Mansoori Y, Bezaatpour A, Shchukarev A, Mikkola JP (2019) ChemistrySelect 4:1820–1829. https://doi.org/10.1002/slct.201803798
Peng X, Zhao Y, Yang T, Yang Y, Jiang Y, Ma Z, Li X, Hou J, Xi B, Liu H (2018) Microporous Mesoporous Mater 258:26–32. https://doi.org/10.1016/j.micromeso.2017.08.050
Huang S, Yang P, Cheng Z, Li C, Fan Y, Kong D, Lin J (2008) J Phys Chem C 112:7130–7137. https://doi.org/10.1021/jp800363s
Yaşar Ş, Köprülü TK, Tekin Ş, Yaşar S (2018) Inorg Chim Acta 479:17–23. https://doi.org/10.1016/j.ica.2018.04.035
Yaşar Ş, Köprülü TK, Tekin Ş, Yaşar S (2018) Appl Organomet Chem 32:4016. https://doi.org/10.1002/aoc.4016
Akkoç M, Balcıoğlu S, Gürses C, Tok TT, Ateş B, Yaşar S (2018) J Organomet Chem 869:67–74. https://doi.org/10.1016/j.jorganchem.2018.06.003
Liu X, Ma Z, Xing J, Liu H (2004) Magn Magn Mater 270:1–6. https://doi.org/10.1016/j.jmmm.2003.07.006
Mondal J, Sreejith S, Borah P, Zhao Y, Sustain ACS (2014) Chem Eng 2:934–941. https://doi.org/10.1021/sc400530a
Choghamarani AG, Tahmasbi B, Moradi P, Havasi N (2016) Appl Organometal Chem 30:619–625. https://doi.org/10.1002/aoc.3478
Nikoorazm M, Moradi P, Noori N (2020) J Porous Mater 27:1159–1169. https://doi.org/10.1007/s10934-020-00894-0
Moradi P, Hajjami M, Tahmasbi B (2020) Polyhedron 175:114169. https://doi.org/10.1016/j.poly.2019.114169
Nikoorazm M, Tahmasbi B, Gholami S, Moradi P (2020) Appl Organomet Chem 34:e5919. https://doi.org/10.1002/aoc.591
Huang Y, Tang J, Gai L, Gong Y, Guang H, He R, Lyu H (2017) Chem Eng J 319:229–239. https://doi.org/10.1016/j.cej.2017.03.015
Wang X, Pan H, Lin Q, Wu H, Jia S, Shi Y (2019) Nanoscale Res Lett 14:259. https://doi.org/10.1186/s11671-019-3075-y
Wu LQ, Li YC, Li SQ, Li ZZ, Tang GD, Qi WH, Xue LC, Ge XS, Ding LL (2015) AIP Adv 5:097210. https://doi.org/10.1063/1.4931996
Ahmed IAM, Maher BA (2018) PNAS 115:1736–1741. https://doi.org/10.1073/pnas.1719186115
Heidari F, Hekmati M, Veisi H (2017) J Colloid Interface Sci 501:175–184. https://doi.org/10.1016/j.jcis.2017.04.054
Nikoorazm M, Ghorbani F, Ghorbani-Choghamarani A, Erfani Z (2017) Chin J Catal 38:1413–1422. https://doi.org/10.1016/S1872-2067(17)62865-1
Baran T, Acıksöz E, Mentes A (2015) J Mol Catal A 407:47–52. https://doi.org/10.1016/j.molcata.2015.06.008
Singha Roy A, Mondal J, Banerjee B, Mondal P, Bhaumik A, Manirul Islam S (2014) Appl Catal A 469:320–327. https://doi.org/10.1016/j.apcata.2013.10.017
Ghorbani-Choghamarani A, Moradi P, Tahmasbi B (2019) Polyhedron 163:98–107. https://doi.org/10.1016/j.poly.2019.02.004
Dong Y, Wu X, Chen X, Wei Y (2017) Carbohydr Polym 160:106–114. https://doi.org/10.1016/j.carbpol.2016.12.044
Sobhani S, Ghasemzadeh MS, Honarmand M, Zarifi F (2014) RSC Adv 4:44166–44174. https://doi.org/10.1039/C4RA04830J
Wang X, Hu P, Xue F, Wei Y (2014) Carbohydr Polym 114:476–483. https://doi.org/10.1016/j.carbpol.2014.08.030
Wang D, Liu W, Bian F, Yu W (2015) New J Chem 39:2052–2059. https://doi.org/10.1039/C4NJ01581A
Ke H, Chen X, Zou G (2014) Appl Organometal Chem 28:54–60. https://doi.org/10.1002/aoc.3076
Karimi B, Akhavan PF (2011) Chem Commun 47:7686–7688. https://doi.org/10.1039/C1CC00017A
Khajehzadeh M, Moghadam M (2018) J Organomet Chem 863:60–69. https://doi.org/10.1016/j.jorganchem.2018.03.030
Esmaeilpour M, Sardarian AR, Firouzabadi H (2018) J Organomet Chem 873:22–34. https://doi.org/10.1016/j.jorganchem.2018.08.002
Akkoç M, Buğday N, Altın S, Yaşar S (2021). Appl Organomet Chem. https://doi.org/10.1002/aoc.6233
Akkoç M, Buğday N, Altın S, Kiraz N, Yaşar S, Özdemir İ (2021) J Organomet Chem 943:121823. https://doi.org/10.1016/j.jorganchem.2021.121823
Acknowledgements
XPS and TEM studies performed in France (CEA LETI/DTSI/SCMC Grenoble Cedex 9 FRANCE and thanks to N. GAMBACORTI for the organization of XPS and TEM analysis) within the framework of the NFFA-Europe Transnational Access Activity (NFFA Project ID: 723).
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Akkoç, M., Buğday, N., Altın, S. et al. Highly Active Fe3O4@SBA-15@NHC-Pd Catalyst for Suzuki–Miyaura Cross-Coupling Reaction. Catal Lett 152, 1621–1638 (2022). https://doi.org/10.1007/s10562-021-03755-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-021-03755-w