Abstract
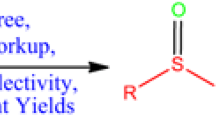
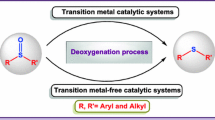
Metal-free aerobic oxidation of aryl sulfides to sulfoxides and sulfones has been developed in the presence of aliphatic aldehydes with excellent selectivity and yields. The reaction proceeded under mild conditions with the catalysis of N-hydroxyphthalimide (NHPI). Control experiments indicated that the reaction underwent a free radical pathway with the acylperoxyl radicals generated from aldehydes in situ as the key intermediates. The aromatic aldehydes were less efficient in the sulfide oxidation, which might be explained by the fact that the aromatic ring dispersed the electrons of free radicals and thus weakened the attacking ability of peroxy free radicals.
Graphic Abstract







Similar content being viewed by others
References
Carreno MC (1995) Applications of sulfoxides to asymmetric synthesis of biologically active compounds. Chem Rev 95:1717–1760. https://doi.org/10.1021/cr00038a002
Fernández I, Khiar N (2003) Recent developments in the synthesis and utilization of chiral sulfoxides. Chem Rev 103:3651–3705. https://doi.org/10.1021/cr990372u
Otocka S, Kwiatkowska M, Madalińska L, Kiełbasiński P (2017) Chiral organosulfur ligands/catalysts with a stereogenic sulfur atom: applications in asymmetric synthesis. Chem Rev 117:4147–4181. https://doi.org/10.1021/acs.chemrev.6b00517
Yu H, Li Z, Bolm C (2018) Transition-metal-free arylations of in-situ generated sulfenates with diaryliodonium salts. Org Lett 20:7104–7106. https://doi.org/10.1021/acs.orglett.8b03046
Jia T, Bellomo A, Montel S, Zhang M, El Baina K, Zheng B, Walsh PJ (2014) Diaryl sulfoxides from aryl benzyl sulfoxides: a single palladium-catalyzed triple relay process. Angew Chem Int Ed 53:260–264. https://doi.org/10.1002/anie.201307172
Izquierdo F, Chartoire A, Nolan SP (2013) Direct S-arylation of unactivated arylsulfoxides using [Pd(IPr*)(cin)Cl]. ACS Catal 3:2190–2193. https://doi.org/10.1021/cs400533e
Liu M, Shi S, Zhao L, Wang M, Zhu G, Zheng X, Gao J, Xu J (2017) Effective utilization of in situ generated hydroperoxide by a Co–SiO2@Ti–Si core-shell catalyst in the oxidation reactions. ACS Catal 8:683–691. https://doi.org/10.1021/acscatal.7b03259
Li B, Liu A-H, He L-N, Yang Z-Z, Gao J, Chen K-H (2012) Iron-catalyzed selective oxidation of sulfides to sulfoxides with the polyethylene glycol/O2 system. Green Chem 14:130–135. https://doi.org/10.1039/c1gc15821j
Rezaeifard A, Jafarpour M, Naeimi A, Haddad R (2012) Aqueous heterogeneous oxygenation of hydrocarbons and sulfides catalyzed by recoverable magnetite nanoparticles coated with copper(II) phthalocyanine. Green Chem 14:3386–3394. https://doi.org/10.1039/c2gc35837a
Neveselý T, Svobodová E, Chudoba J, Sikorski M, Cibulka R (2016) Efficient metal-free aerobic photooxidation of sulfides to sulfoxides mediated by a vitamin B2 derivative and visible light. Adv Synth Catal 358:1654–1663. https://doi.org/10.1002/adsc.201501123
Iwahama T, Sakaguchi S, Ishii Y (1998) Selective oxidation of sulfides to sulfoxides with molecular oxygen catalyzed by N-hydroxyphthalimide (NHPI) in the presence of alcohols. Tetrahedron Lett 39:9059–9062. https://doi.org/10.1016/S0040-4039(98)02054-1
Liu K-J, Deng J-H, Yang J, Gong S-F, Lin Y-W, He J-Y, Cao Z, He W-M (2020) Selective oxidation of (hetero) sulfides with molecular oxygen under clean conditions. Green Chem 22:433–438. https://doi.org/10.1039/C9GC03713F
Cheng Z, Sun P, Tang A, Jin W, Liu C (2019) Switchable synthesis of aryl sulfones and sulfoxides through solvent-promoted oxidation of sulfifides with O2/air. Org Lett 21:8925–8929. https://doi.org/10.1021/acs.orglett.9b03192
Shapiro N, Kramer M, Goldberg I, Vigalok A (2010) Straightforward radical organic chemistry in neat conditions and “on water.” Green Chem 12:582–584. https://doi.org/10.1039/B922475K
Vanoye L, Abdelaal M, Grundhauser K, Guicheret B, Fongarland P, Bellefon CD, Favre-Reguillon A (2019) Reinvestigation of the organocatalyzed aerobic oxidation of aldehydes to acids. Org Lett 21:10134–10138. https://doi.org/10.1021/acs.orglett.9b04193
Murahashi S-I, Oda Y, Naota T (1992) Iron-and ruthenium-catalyzed oxidations of alkanes with molecular oxygen in the presence of aldehydes and acids. J Am Chem Soc 114:7914–7916. https://doi.org/10.1021/ja00046a048
Murahashi S-I, Oda Y, Naota T, Komiya N (1993) Aerobic oxidations of alkanes and alkenes in the presence of aldehydes catalysed by copper salts. Chem Commun 2:139–140. https://doi.org/10.1039/c39930000139
Li X, Wang F, Lu X, Song G, Zhang H (1997) A novel method for epoxidation of cyclohexene catalyzed by Fe2O3 with molecular oxygen and aldehydes. Synth Commun 27:2075–2079. https://doi.org/10.1080/00397919708006813
Li X, Wang F, Zhang H, Wang C, Song G (1996) Baeyer-villiger oxidation of cyclohexanone with molecular oxygen in the presence of benzaldehyde. Synth Commun 26:1613–1616. https://doi.org/10.1080/00397919608003530
Wang L, Wang Y, Du R, Dao R, Yuan H, Liang C, Yao J, Li H (2018) N-hydroxyphthalimide (NHPI) promoted aerobic baeyer-villiger oxidation in the presence of aldehydes. ChemCatChem 10:4947–4952. https://doi.org/10.1002/cctc.201801165
Wang L, Zhang Y, Du R, Yuan H, Wang Y, Yao J, Li H (2019) Selective one-step aerobic oxidation of cyclohexane to ε-caprolactone mediated by N-hydroxyphthalimide (NHPI). ChemCatChem 11:2260–2264. https://doi.org/10.1002/cctc.201900282
Minisci F, Gambarotti C, Pierini M, Porta O, Punta C, Recupero F, Lucarini M, Mugnaini V (2006) Molecule-induced homolysis of N-hydroxyphthalimide (NHPI) by peracids and dioxirane: a new, simple, selective aerobic radical epoxidation of alkenes. Tetrahedron Lett 47:1421–1424. https://doi.org/10.1016/j.tetlet.2005.12.089
Rao TV, Sain B, Kumar K, Murthy PS, Rao TSRP, Joshi GC (1998) Oxidation of sulphides by molecular oxygen-aldehyde system in the absence of metal catalyst. Synth Commun 28:319–326. https://doi.org/10.1080/00397919808005725
Rao TV, Sain B, Kafola S, Nautiyal BR, Sharma YK, Nanoti SM, Garg MO (2007) Oxidative desulfurization of HDS diesel using the aldehyde-molecular oxygen oxidation system. Energy Fuels 21:3420–3424. https://doi.org/10.1021/ef700245g
Murata S, Murata K, Kidena K, Nomura M (2004) A novel oxidative desulfurization system for diesel fuels with molecular oxygen in the presence of cobalt catalysts and aldehydes. Energy Fuels 18:116–121. https://doi.org/10.1021/ef034001z
Maity N, Wattanakit C, Muratsugu S, Ishiguro N, Yang Y, Ohkoshi S-I, Tada M (2012) Sulfoxidation on a SiO2-supported Ru complex using O2/aldehyde system. Dalton Trans 41:4558–4565. https://doi.org/10.1039/c2dt12133f
Zhang P, Wang Y, Li H, Antonietti M (2012) Metal-free oxidation of sulfides by carbon nitride with visible light illumination at room temperature. Green Chem 14:1904–1908. https://doi.org/10.1039/C2GC35148J
Wang J-Q, He L-N, Miao C-X, Gao J (2009) The free-radical chemistry of polyethylene glycol: organic reactions in compressed carbon dioxide. ChemSusChem 2:755–760. https://doi.org/10.1002/cssc.200900060
Wertz JE, Bolton JR (1972) Elementary theory and practical applications. McGraw-Hill, New York
Acknowledgements
This work was supported by the National Natural Science Foundation of China (Grant No. 22073081), and the Fundamental Research Funds for the Central Universities. L. Wang gratefully acknowledge the financial support for this research by the Zhejiang Province Postdoctoral Science Foundation (Grant No. ZJ2020159).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Wang, L., Zhang, Y., Yao, J. et al. Metal-Free Synthesis of Sulfones and Sulfoxides through Aldehyde-Promoted Aerobic Oxidation of Sulfides. Catal Lett 152, 1131–1139 (2022). https://doi.org/10.1007/s10562-021-03706-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-021-03706-5