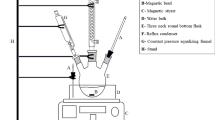
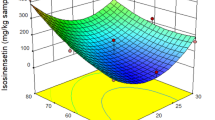
Abstract
Response surface methodology (RSM) was used to optimize the oxidizing the omeprazole sulfide to (S)-omeprazole catalyzed by environmentally friendly catalyst soybean pod peroxidase (SPP) in cetyltrimethylammonium bromide (CTAB)/isooctane/n-butyl alcohol/water water-in-oil microemulsions. With the initial concentration of SPP of 3200 U ml−1, the conversion of the omeprazole sulfide, the (S)-omeprazole yield and ee were 93.75%, 91.56% and 96.08%, respectively, under the optimal conditions: Wo of 15.85, the concentration of H2O2 of 22.44 mM and reaction temperature of 49.68 °C, respectively. The proposed mechanism of asymmetric sulfoxidations catalyzed by SPP involves three concomitant mechanisms as follows: (1) a two-electron reduction of SPP-I, (2) a single-electron transfer to SPP-I and (3) nonenzymatic reactions. Based on the proposed mechanism which is reasonable and can express the oxidations, the reaction system includes five enzymatic and two nonenzymatic reactions. With 5.44% of the average relative error, a kinetic model based on the mechanisms fitting observed data very well was established, and the SPP-catalyzed reactions including both the two-electron reduction and the single-electron transfer mechanisms obey ping-pong mechanism with substrate and product inhibition, while nonenzymatic reactions follow a power law. This study has also demonstrated the feasibility of SPP as a substitute with low cost, excellent enantioselectivity and better thermal stability.
Graphic Abstract







Similar content being viewed by others
Abbreviations
- Sa :
-
Hydrogen peroxide
- A0 :
-
Initial concentration of hydrogen peroxide, mM
- Sb :
-
5-Methoxy-2-(((4-methoxy-3,5-dimethylpyridin-2-yl)methyl)thio)-1H-benzoimidazole, Omeprazole thioether, 5-methoxy-2-(((4-methoxy-3,5-dimethylpyridin-2-yl)methyl)thio)-1H-benzo[d]imidazole
- B0 :
-
Initial concentration of Sb, mM
- Sc :
-
Cation radical of Sb, Omeprazole sulfide cation
- E0 :
-
Initial concentration of peroxidase, U ml−1
- ee:
-
Enantiomeric excess, %
- K1, K10, K19 :
-
Kinetic parameter, dimensionless
- K2, K3, K6 :
-
Kinetic parameter, mM−1
- K4, K5, K7 :
-
Kinetic parameter, mM−2
- K8 :
-
Kinetic parameter, mM−3
- K9 :
-
Kinetic parameter, h−1 U−1 ml mM1−2K10
- K12, K14, K16 :
-
Kinetic parameter, h−1 U−1 ml mM−1
- K11, K13, K15, K17 :
-
Kinetic parameter, h−1 U−1 ml mM−2
- K18 :
-
Kinetic parameter, h−1 U−1 ml mM1−K19
- KK1 KK2:
-
∑K sum of Kappa constant, dimensionless
- PR :
-
(R)-enantiomer of P
- PS :
-
(S)-enantiomer of P, esomeprazole
- P:
-
5-Methoxy-2-[(4-methoxy-3,5-dimethylpyridin-2-yl) methylsulfinyl]-1H-benzoimidazole, Omeprazole
- Q:
-
H2O
- R:
-
Dication radical of Sb
- Wo:
-
Water/CTAB, mol
- Y(i):
-
Relative residual or yield of Sa, Sb, PS, PR, C and R, for i = 1–6, respectively, dimensionless
- Ysimulij :
-
Simulated residual or yield of Sa, Sb, PS and PR, for i = 1–4, j = 1–12, dimensionless
- Yexpi j :
-
Experimental residual or yield of Sa, Sb, PS and PR, for i = 1–4, j = 1–12, dimensionless
- T:
-
Time, hour
References
Delamare M, Belot S, Caille J-C, Martinet F, Kagan HB, Henryon V (2009) A new titanate/(+)-(1R, 2S)-cis-1-amino-2-indanol system for the asymmetric synthesis of (S)-tenatoprazole. Tetrahedron Lett 50(15):1702–1704
Dembitsky VM (2003) Oxidation, epoxidation and sulfoxidation reactions catalysed by haloperoxidases. Tetrahedron 26(59):4701–4720
Thomas SM, DiCosimo R, Nagarajan V (2002) Biocatalysis: applications and potentials for the chemical industry. Trends Biotechnol 20(6):238–242
Carreño MC (1995) Applications of sulfoxides to asymmetric synthesis of biologically active compounds. Chem Rev 95(6):1717–1760
Harris RZ, Newmyer SL, De Montellano PRO (1993) Horseradish peroxidase-catalyzed two-electron oxidations. Oxidation of iodide, thioanisoles, and phenols at distinct sites. Journal of Biological Chemistry 268(3):1637–1645
Blee E, Schuber F (1989) Mechanism of S-oxidation reactions catalyzed by a soybean hydroperoxide-dependent oxygenase. Biochemistry 28(12):4962–4967
Van Deurzen MPJ, van Rantwijk F, Sheldon RA (1997) Selective oxidations catalyzed by peroxidases. Tetrahedron 53(39):13183–13220
van de Velde F, Könemann L, van Rantwijk F, Sheldon RA (2000) The rational design of semisynthetic peroxidases. Biotechnol Bioeng 67(1):87–96
Zhang Y, Wu Y-Q, Xu N, Zhao Q, Yu H-L, Xu J-H (2019) Engineering of cyclohexanone monooxygenase for the enantioselective synthesis of (S)-omeprazole. ACS Sustainable Chemistry & Engineering 7(7):7218–7226
Zhang Y, Liu F, Xu N, Wu Y-Q, Zheng Y-C, Zhao Q, Lin G, Yu H-L, Xu J-H (2018) Discovery of two native Baeyer-Villiger monooxygenases for asymmetric synthesis of bulky chiral sulfoxides. Appl Environ Microbiol 84(14):e00638-e718
Ozaki SI, Matsui T, Watanabe Y (1997) Conversion of Myoglobin into a Peroxygenase:? A Catalytic Intermediate of Sulfoxidation and Epoxidation by the F43H/H64L Mutant. J Am Chem Soc 119(28):6666–6667
Akasaka R, Mashino T, Hirobe M (1993) Cytochrome P450-like Substrate Oxidation Catalyzed by Cytochrome c and Immobilized Cytochrome c. Archives of Biochemistry & Biophysics 301(2):355–360
Singh P, Prakash R, Shah K (2012) Effect of organic solvents on peroxidases from rice and horseradish: prospects for enzyme based applications. Talanta 97:204–210
Hong ES, Kwon OY, Ryu K (2008) Strong substrate-stabilizing effect of a water-miscible ionic liquid [BMIM][BF 4] in the catalysis of horseradish peroxidase. Biotech Lett 30(3):529–533
Lopez F, Cinelli G, Colella M, De Leonardis A, Palazzo G, Ambrosone L (2014) The role of microemulsions in lipase-catalyzed hydrolysis reactions. Biotechnol Prog 30(2):360–366
Seenivasaperumal M, Federsel H-J, Szab K (2010) Mechanism of the Asymmetric Sulfoxidation in the Esomeprazole Process: Effects of the Imidazole Backbone for the Enantioselection. Adv Synth Catal 351(6):903–919
Amiri S, Shakeri A, Sohrabi MR, Khalajzadeh S, Ghasemi E (2019) Optimization of ultrasonic assisted extraction of fatty acids from Aesculus hippocastanum fruit by response surface methodology. Food Chem 271:762–766
Adachi M, Harada M, Katoh S (2000) Bioaffinity separation of chymotrypsinogen using antigen-antibody reaction in reverse micellar system composed of a nonionic surfactant. Biochem Eng J 4(2):149–151
Krieger N, Taipa MA, Melo EHM, Lima-Filho JL, Aires-Barros MR, Cabral JMS (1997) Kinetic characterization ofpenicillium citrinum lipase in AOT/lsooctane-reversed micelles. Appl Biochem Biotechnol 67(1–2):87–95
Carvalho CML, Cabral JMS (2000) Reverse micelles as reaction media for lipases. Biochimie 82(11):1063–1085
Chen N, Fan J-B, Xiang J, Chen J, Liang Y (2006) Enzymatic hydrolysis of microcrystalline cellulose in reverse micelles. Biochim Biophys Acta Proteins Proteom 1764(6):1029–1035
Bru R, Sánchez-Ferrer A, García-Carmona F (1995) Kinetic models in reverse micelles. Biochem J 310(3):721–739
Hamann H-J, Höft E, Mostowicz D, Mishnev A, Urbańczyk-Lipkowska Z, Chmielewski M (1997) New optically pure sugar hydroperoxides. Synhtesis and use for enantioselective oxygen transfer. Tetrahedron 53(1):185–192
Goodwin DC, Grover TA, Aust SD (1997) Roles of efficient substrates in enhancement of peroxidase-catalyzed oxidations. Biochemistry 36(1):139–147
Drożdż A, Erfurt K, Bielas R, Chrobok A (2015) Chemo-enzymatic Baeyer-Villiger oxidation in the presence of Candida antarctica lipase B and ionic liquids. New J Chem 39(2):1315–1321
Törnvall U, Fürst CM, Hatti-Kaul R, Hedström M (2009) Mass spectrometric analysis of peptides from an immobilized lipase: focus on oxidative modifications. Rapid Commun Mass Spectrom 23(18):2959–2964
Törnvall U, Hedström M, Schillén K, Hatti-Kaul R (2010) Structural, functional and chemical changes in Pseudozyma antarctica lipase B on exposure to hydrogen peroxide. Biochimie 92(12):1867–1875
Ji L, Franke A, Brindell M, Oszajca M, Zahl A, van Eldik R (2014) Combined experimental and theoretical study on the reactivity of Compounds I and II in horseradish peroxidase biomimetics. Chem Eur J 20(44):14437–14450
Dunford HB (1991) Horseradish peroxidase: structure and kinetic properties. In: Everse J, Everse KE, Grisham MB (eds) Perozidases in Chemistry and Biology. CRC Press, Boca Raton, pp 1–23
Samuni U, Maimon E, Goldstein S (2018) A kinetic study of the oxidation of hydroxamic acids by compounds I and II of horseradish peroxidase: Effect of transition metal ions. J Coord Chem 71(11–13):1728–1737
Harris DL, Loew GH (1996) Identification of putative peroxide intermediates of peroxidases by electronic structure and spectra calculations. J Am Chem Soc 118(43):10588–10594
Perez U, Dunford HB (1990) Transient-state kinetics of the reactions of 1-methoxy-4-(methylthio) benzene with horseradish peroxidase compounds I and II. Biochemistry 29(11):2757–2763
Doerge DR, Cooray NM, Brewster ME (1991) Peroxidase-catalyzed S-oxygenation: mechanism of oxygen transfer for lactoperoxidase. Biochemistry 30(37):8960–8964
Kobayashi S, Nakano M, Kimura T, Schaap AP (1987) On the mechanism of the peroxidase-catalyzed oxygen-transfer reaction. Biochemistry 26(16):5019–5022
Newmyer SL, de Montellano PRO (1995) Horseradish Peroxidase His-42→ Ala, His-42→ Val, and Phe-41→ Ala Mutants histidine catalysis and control of substrate access to the heme iron. J Biol Chem 270(33):19430–19438
Hashimoto S, Tatsuno Y, Kitagawa T (1986) Resonance Raman evidence for oxygen exchange between the FeIV= O heme and bulk water during enzymic catalysis of horseradish peroxidase and its relation with the heme-linked ionization. Proc Natl Acad Sci 83(8):2417–2421
Kamble MP, Shinde SD, Yadav GD (2016) Kinetic resolution of (R, S)-α-tetralol catalyzed by crosslinked Candida antarctica lipase B enzyme supported on mesocellular foam: a nanoscale enzyme reactor approach. J Mol Catal B Enzym 132:61–66
Mathpati AC, Badgujar KC, Bhanage BM (2016) Kinetic modeling and docking study of immobilized lipase catalyzed synthesis of furfuryl acetate. Enzyme Microb Technol 84:1–10
Zhang Y, Zhao Y, Jiang W, Yao Q, Li Z, Gao X, Liu T, Yang F, Wang F, Liu J (2019) Lipase-catalyzed oxidation of cyclohexanone to form ε-caprolactone and kinetic modeling. ACS Sustainable Chemistry & Engineering 7(15):13294–13306
Zhang Y, Zhao Y, Gao X, Jiang W, Li Z, Yao Q, Yang F, Wang F, Liu J (2019) Kinetic model of the enzymatic Michael addition for synthesis of mitomycin analogs catalyzed by immobilized lipase from T. laibacchii. Molecular Catalysis 466:146–156
Dai L, Klibanov AM (2000) Peroxidase-catalyzed asymmetric sulfoxidation in organic solvents versus in water. Biotechnol Bioeng 70(3):353–357
Colonna S, Gaggero N, Richelmi C, Pasta P (1999) Recent biotechnological developments in the use of peroxidases. Trends Biotechnol 17(4):163–168
Zhang Y, Kuiying L, Deng Y, Li H, Wang Z, Li D, Gao X, Wang F (2021) Asymmetric Biooxidation using resting cells of Rhodococcus rhodochrous ATCC 4276 mutant QZ-3 for preparation of (S)-omeprazole in a chloroform–water biphasic system using response surface methodology. Catal Lett. https://doi.org/10.1007/s10562-021-03531-w
Acknowledgements
This work was supported by the National Natural Science Foundation of China [Grant Numbers 21546004 and 21576145], China Scholarship Council (Grant Number: 201908370079) and Shandong Provincial Key R&D Program [Grant Numbers 2019GSF107027, 2019GNC106028 and 2019GSF107033].
Author information
Authors and Affiliations
Contributions
HT and YYZ designed and wrote the manuscript; YSD, DPL, ZYW, HLL, XG, FYW revised the manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Tang, H., Zhang, Y., Deng, Y. et al. Optimization of Synthesis of (S)-Omeprazole Catalyzed by Soybean Pod Peroxidase in Water-in-Oil Microemulsions Using RSM. Catal Lett 152, 720–731 (2022). https://doi.org/10.1007/s10562-021-03681-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-021-03681-x