Abstract
A high performance HZSM-5 catalyst for liquid phase dehydration of methanol to dimethyl ether (DME) was developed by optimizing Si/Al ratio and hydrothermal treatment temperature. During 6788 h long term test, DME selectivity kept above 99.96% and only slight deactivation was observed primarily due to coking and decline of HZSM-5 crystallinity.
Graphic Abstract




Similar content being viewed by others
References
Mondal U, Yadav GD (2019) Perspective of dimethyl ether as fuel: Part I. Catalysis. J CO2 Util 32:299–320. https://doi.org/10.1016/j.jcou.2019.02.003
Azizi Z, Rezaeimanesh M, Tohidian T, Rahimpour MR (2014) Dimethyl ether: a review of technologies and production challenges. Chem Eng Process Process Intensif 82:150–172. https://doi.org/10.1016/j.cep.2014.06.007
Arcoumanis C, Bae C, Crookes R, Kinoshita E (2008) The potential of di-methyl ether (DME) as an alternative fuel for compression-ignition engines: a review. Fuel 87:1014–1030. https://doi.org/10.1016/j.fuel.2007.06.007
Akarmazyan SS, Panagiotopoulou P, Kambolis A, Papadopoulou C, Kondarides DI (2014) Methanol dehydration to dimethylether over Al2O3 catalysts. Appl Catal B Environ 145:136–148. https://doi.org/10.1016/j.apcatb.2012.11.043
Lei Z, Zou Z, Dai C, Li Q, Chen B (2011) Synthesis of dimethyl ether (DME) by catalytic distillation. Chem Eng Science 66:3195–3203. https://doi.org/10.1016/j.ces.2011.02.034
Hänggi S, Elbert P, Bütler T, Cabalzar U, Teske S, Bach C, Onder C (2019) A review of synthetic fuels for passenger vehicles. Energy Rep 5:555–569. https://doi.org/10.1016/j.egyr.2019.04.007
Bahadori F, Oshnuie MN (2019) Exergy analysis of indirect dimethyl ether production process. Sustain Energy Technol Assess 31:142–145. https://doi.org/10.1016/j.seta.2018.12.025
Migliori M, Aloise A, Giordano G (2014) Methanol to dimethylether on H-MFI catalyst: the influence of the Si/Al ratio on kinetic parameters. Catal Today 227:138–143. https://doi.org/10.1016/j.cattod.2013.09.033
Spivey JJ (1991) Review: Dehydration catalysts for the methanol/dimethyl ether reaction. Chem Eng Commun 110:123–142. https://doi.org/10.1080/00986449108939946
Boon J, van Kampen J, Hoogendoorn R, Tanase S, van Berkel FPF, MvanS A (2019) Reversible deactivation of γ-alumina by steam in the gas-phase dehydration of methanol to dimethyl ether. Catal Commun 119:22–27. https://doi.org/10.1016/j.catcom.2018.10.008
Osman AI, Abu-Dahrieh JK, Abdelkader A, Hassan NM, Laffir F, McLaren M, Rooney D (2017) Silver-modified η-Al2O3 catalyst for DME production. J Phys Chem C 121:25018–25032. https://doi.org/10.1021/acs.jpcc.7b04697
Sierraalta A, Añez R, Coll DS, Alejos P (2020) Conversion of methanol to dimethyl ether over silicoaluminophosphates: isolated acid sites and the influence of silicon islands. A DFT-ONIOM study. Microporous Mesoporous Mater 292:109732–109740. https://doi.org/10.1016/j.micromeso.2019.109732
Migliori M, Catizzone E, Aloise A, Bonura G, Gómez-Hortigüela L, Frusteri L, Cannilla C, Frusteri F, Giordano G (2018) New insights about coke deposition in methanol-to-DME reaction over MOR- MFI- and FER-type zeolites. J Ind Eng Chem 68:196–208. https://doi.org/10.1016/j.jiec.2018.07.046
Wei Y, de Jongh PE, Bonati MLM, Law DJ, Sunley GJ, de Jong KP (2015) Enhanced catalytic performance of zeolite ZSM-5 for conversion of methanol to dimethyl ether by combining alkaline treatment and partial activation. Appl Catal A Gen 504:211–219. https://doi.org/10.1016/j.apcata.2014.12.027
Catizzone E, Aloise A, Migliori M, Giordano G (2015) Dimethyl ether synthesis via methanol dehydration: effect of zeolite structure. Appl Catal A Gen 502:215–220. https://doi.org/10.1016/j.apcata.2015.06.017
Yu Y, Sun D, Wang S, Xiao M, Sun L, Meng Y (2019) Heteropolyacid salt catalysts for methanol conversion to hydrocarbons and dimethyl ether: effect of reaction temperature. Catalysts 9(4):320. https://doi.org/10.3390/catal9040320
Alharbi W, Kozhevnikova EF, Kozhevnikov IV (2015) Dehydration of methanol to dimethyl ether over heteropoly acid catalysts: the relationship between reaction rate and catalyst acid strength. ACS Catal 5:7186–7193. https://doi.org/10.1021/acscatal.5b01911
Said AEAA, Goda MN, Kassem MA (2020) Promotional effect of B2O3 WO3 and ZrO2 on the structural textural and catalytic properties of FePO4. Catal Lett 150:1714–1728. https://doi.org/10.1007/s10562-019-03081-2
Mondal U, Yadav GD (2019) Perspective of dimethyl ether as fuel: Part II-analysis of reactor systems and industrial processes. J CO2 Util 32:321–338. https://doi.org/10.1016/j.jcou.2019.02.006
Hosseininejad S, Afacan A, Hayes RE (2012) Catalytic and kinetic study of methanol dehydration to dimethyl ether. Chem Eng Res Des 90:825–833. https://doi.org/10.1016/j.cherd.2011.10.007
Liu Z, Sun X, Zhu S, Xu L, Lv Z, Meng S (2007) Process for synthesis of dimethyl ether by catalytic distillation from methanol. WO Pat 2007014534 A1
Kiss AA, Suszwalak DJ-PC, Ignat RM (2013) Process intensification alternatives in the DME production. Chem Eng Trans 35:91–96. https://doi.org/10.3303/CET1335015
Khandan N, Kazemeini M, Aghaziarati M (2008) Determining an optimum catalyst for liquid-phase dehydration of methanol to dimethyl ether. Appl Catal A Gen 349:6–12. https://doi.org/10.1016/j.apcata.2008.07.029
Prodinger S, Shi H, Eckstein S, Hu JZ, Olarte MV, Camaioni DM, Derewinski MA, Lercher JA (2017) Stability of zeolites in aqueous phase reactions. Chem Mater 29:7255–7262. https://doi.org/10.1021/acs.chemmater.7b01847
Sun X, Wang Y, He Y, Yang Y, Xu S, Zhu S, Yang M, Liu Z (2019) Dissolution equilibrium and in situ growth of HMCM-49 in aqueous-phase reaction. Ind Eng Chem Res 58:9339–9342. https://doi.org/10.1021/acs.iecr.9b01417
Zhang L, Chen K, Chen B, White JL, Resasco DE (2015) Factors that determine zeolite stability in hot liquid water. J Am Chem Soc 137:11810–11819. https://doi.org/10.1021/jacs.5b07398
Ravenelle RM, Schüßler F, D’Amico A, Danilina N, van Bokhoven JA, Lercher JA, Jones CW, Sievers C (2010) Stability of zeolites in hot liquid water. J Phys Chem C 114:19582–19595. https://doi.org/10.1021/jp104639e
Lutz W, Toufar H, Kurzhals R, Suckow M (2005) Investigation and modeling of the hydrothermal stability of technically relevant zeolites. Adsorption 11:405–413. https://doi.org/10.1007/s10450-005-5406-9
Liu Z, Sun X, Xu L, Meng S, Zhu S, Lv Z (2007) Catalyst for preparing dimethyl ether by dewatering of methanol at liquid phase or mixed phase. WO Pat 2007006238 A1
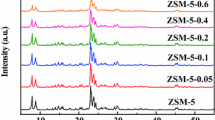
Zhang S, Gong Y, Zhang L, Liu Y, Dou T, Xu J, Deng F (2015) Hydrothermal treatment on ZSM-5 extrudates catalyst for methanol to propylene reaction: finely tuning the acidic property. Fuel Process Technol 129:130–138. https://doi.org/10.1016/j.fuproc.2014.09.006
Holzinger J, Beato P, Lundegaard LF, Skibsted J (2018) Distribution of aluminum over the tetrahedral sites in ZSM-5 zeolites and their evolution after steam treatment. J Phys Chem C 122:15595–15613. https://doi.org/10.1021/acs.jpcc.8b05277
Kubo K, Iida H, Namba S, Igarashi A (2014) Effect of steaming on acidity and catalytic performance of H-ZSM-5 and P/H-ZSM-5 as naphtha to olefin catalysts. Microporous Mesoporous Mater 188:23–29. https://doi.org/10.1016/j.micromeso.2014.01.002
Meng Q, Xin H, Zhang Y, Huang Y, Yi X, Sun Y, Zhong S, Li X (2015) Catalytic dehydration of ethanol to ethylene over steam-treated ZSM-5 zeolites. Sci Adv Mater 7:2343–2351. https://doi.org/10.1166/sam.2015.2388
Almutairi SMT, Mezari B, Pidko EA, Magusin PCMM, Hensen EJM (2013) Influence of steaming on the acidity and the methanol conversion reaction of HZSM-5 zeolite. J Catal 307:194–203. https://doi.org/10.1016/j.jcat.2013.07.021
Tian P, Wei Y, Ye M, Liu Z (2015) Methanol to olefins (MTO): from fundamentals to commercialization. ACS Catal 5:1922–1938. https://doi.org/10.1021/acscatal.5b00007
Qi L, Wei Y, Xu L, Liu Z (2015) Reaction behaviors and kinetics during induction period of methanol conversion on HZSM-5 zeolite. ACS Catal 5:3973–3982. https://doi.org/10.1021/acscatal.5b00654
Vjunov A, Fulton JL, Camaioni DM, Hu JZ, Burton SD, Arslan I, Lercher JA (2015) Impact of aqueous medium on zeolite framework integrity. Chem Mater 27:3533–3545. https://doi.org/10.1021/acs.chemmater.5b01238
Ennaert T, Geboers J, Gobechiya E, Courtin CM, Kurttepeli M, Houthoofd K, Kirschhock CEA, Magusin PCMM, Bals S, Jacobs PA, Sels BF (2015) Conceptual frame rationalizing the self-stabilization of H-USY zeolites in hot liquid water. ACS Catal 5:754–768. https://doi.org/10.1021/cs501559s
Shen SC, Kawi S (1999) Understanding of the effect of Al substitution on the hydrothermal stability of MCM-41. J Phys Chem B 103:8870–8876. https://doi.org/10.1021/jp991831y
Mokaya R (2000) Al content dependent hydrothermal stability of directly synthesized aluminosilicate MCM-41. J Phys Chem B 104:8279–8286. https://doi.org/10.1021/jp001494p
Zapata PA, Huang Y, Gonzalez-Borja MA, Resasco DE (2013) Silylated hydrophobic zeolites with enhanced tolerance to hot liquid water. J Catal 208:82–97. https://doi.org/10.1016/j.jcat.2013.05.024
Acknowledgements
We are grateful to Professor Chenglin Sun and Ms. Yefang Ruan for their beneficial discussion and help in experiments.
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
The manuscript was written through contributions of all authors. XS: Data curation, Formal analysis, Investigation, Writing. YY: Data curation, Investigation. YH: Formal analysis, Investigation. SZ: Supervision, Validation. ZL: Conceptualization, Project administration, Supervision.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no conflicts of interest to declare that are relevant to the content of this article.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Sun, X., Yang, Y., He, Y. et al. Stability of Zeolite HZSM-5 in Liquid Phase Dehydration of Methanol to Dimethyl Ether. Catal Lett 151, 2004–2010 (2021). https://doi.org/10.1007/s10562-020-03454-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03454-y