Abstract
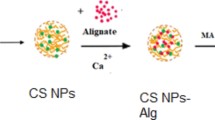
This work aimed to synthesize new heterogeneous iron-based catalysts supported on biopolymers (alginate (Alg), carboxymethylcellulose (CMC), xanthan gum (GX) and chitosan (Qui)) and their mixtures—and their mixtures—to evaluate their degradation Bisphenol A (BPA) in aqueous solutions. The Alg-Fe, QuiGa-Fe and CMC-Fe, CMCGX-Fe, AlgCMC-Fe, AlgGX-Fe catalysts were prepared and characterized by SEM, FTIR and FAAS. For the degradation of BPA was evaluated and AlgCMC-Fe was considered the best catalyst for having high catalytic activity and less leaching of iron ions in the reaction medium. Then, the kinetics of BPA degradation by Fenton oxidation process were investigated varying: H2O2 concentration, AlgCMC-Fe content, pH, reaction time and initial BPA concentrations. The degradation of BPA was monitored by HPLC–DAD and the results showed that AlgCMC-Fe were able to promote total degradation of BPA in aqueous solutions a (2.00 mg L−1 BPA) at a pH close to neutral, with a recycling capacity of up to three times with the same efficiency. The pseudo-second-order was the most appropriate model to describe the kinetic mechanism.
Graphic Abstract







Similar content being viewed by others
References
Elena A, Orbeci C, Lazau C et al (2013) Waste water treatment methods. In: Water treatment. InTech, London
Zheng C, Zhao L, Zhou X et al (2013) Treatment technologies for organic wastewater. In: Water treatment. InTech, London
Babuponnusami A, Muthukumar K (2014) A review on Fenton and improvements to the Fenton process for wastewater treatment. J Environ Chem Eng 2:557–572. https://doi.org/10.1016/j.jece.2013.10.011
Reddy PVL, Kim KH, Kavitha B et al (2018) Photocatalytic degradation of bisphenol A in aqueous media: a review. J Environ Manag 213:189–205
Olmez-Hanci T, Arslan-Alaton I, Genc B (2013) Bisphenol A treatment by the hot persulfate process: oxidation products and acute toxicity. J Hazard Mater 263:283–290. https://doi.org/10.1016/j.jhazmat.2013.01.032
Dietrich M, Franke M, Stelter M, Braeutigam P (2017) Degradation of endocrine disruptor bisphenol A by ultrasound-assisted electrochemical oxidation in water. Ultrason Sonochem. https://doi.org/10.1016/j.ultsonch.2017.05.038
Wang N, Zheng T, Zhang G, Wang P (2016) A review on Fenton-like processes for organic wastewater treatment. J Environ Chem Eng 4:762–787. https://doi.org/10.1016/j.jece.2015.12.016
Lee Y, Lee W (2010) Degradation of trichloroethylene by Fe(II) chelated with cross-linked chitosan in a modified Fenton reaction. J Hazard Mater 178:187–193. https://doi.org/10.1016/j.jhazmat.2010.01.062
Cruz A, Couto L, Esplugas S, Sans C (2017) Study of the contribution of homogeneous catalysis on heterogeneous Fe(III)/alginate mediated photo-Fenton process. Chem Eng J 318:272–280. https://doi.org/10.1016/j.cej.2016.09.014
Munoz M, de Pedro ZM, Casas JA, Rodriguez JJ (2015) Preparation of magnetite-based catalysts and their application in heterogeneous Fenton oxidation—a review. Appl Catal B 176–177:249–265. https://doi.org/10.1016/j.apcatb.2015.04.003
Dong Y, Dong W, Cao Y et al (2011) Preparation and catalytic activity of Fe alginate gel beads for oxidative degradation of azo dyes under visible light irradiation. Catal Today 175:346–355. https://doi.org/10.1016/j.cattod.2011.03.035
Rosales E, Iglesias O, Pazos M, Sanromán MA (2012) Decolourisation of dyes under electro-Fenton process using Fe alginate gel beads. J Hazard Mater 213–214:369–377. https://doi.org/10.1016/j.jhazmat.2012.02.005
Iglesias O, Gómez J, Pazos M, Sanromán MÁ (2014) Electro-Fenton oxidation of imidacloprid by Fe alginate gel beads. Appl Catal B 144:416–424. https://doi.org/10.1016/j.apcatb.2013.07.046
Nidheesh PV (2015) Heterogeneous Fenton catalysts for the abatement of organic pollutants from aqueous solution: a review. RSC Adv 5:40552–40577. https://doi.org/10.1039/C5RA02023A
Ben HS, Adhoum N, Monser L (2015) Synthesis of magnetic alginate beads based on Fe3O4 nanoparticles for the removal of 3-methylindole from aqueous solution using Fenton process. J Hazard Mater 294:128–136. https://doi.org/10.1016/j.jhazmat.2015.03.068
He J, Yang X, Men B, Wang D (2016) Interfacial mechanisms of heterogeneous Fenton reactions catalyzed by iron-based materials: a review. J Environ Sci 39:97–109. https://doi.org/10.1016/j.jes.2015.12.003
Ben HS, Fourcade F, Assadi A et al (2016) Effective heterogeneous electro-Fenton process for the degradation of a malodorous compound, indole, using iron loaded alginate beads as a reusable catalyst. Appl Catal B 182:47–58. https://doi.org/10.1016/j.apcatb.2015.09.007
Ben Hammouda S, Adhoum N, Monser L (2016) Chemical oxidation of a malodorous compound, indole, using iron entrapped in calcium alginate beads. J Hazard Mater 301:350–361. https://doi.org/10.1016/j.jhazmat.2015.09.012
de Jesús Ruíz-Baltazar Á, Reyes-López SY, de Lourdes Mondragón-Sánchez M et al (2019) Eco-friendly synthesis of Fe3O4 nanoparticles: evaluation of their catalytic activity in methylene blue degradation by kinetic adsorption models. Results Phys 12:989–995. https://doi.org/10.1016/j.rinp.2018.12.037
Shriver DF, Atkins PW, Overton TL et al (2008) Química inorgânica, 4th edn. Bookman, Porto Alegre
Papageorgiou SK, Kouvelos EP, Favvas EP et al (2010) Metal–carboxylate interactions in metal–alginate complexes studied with FTIR spectroscopy. Carbohydr Res 345:469–473. https://doi.org/10.1016/j.carres.2009.12.010
Silvertein RM, Webster FX, Kiemle DJ (2007) Identificação espectrométrica de compostos orgânicos, 7th edn. LTC, Rio de Janeiro
Lee KY, Mooney DJ (2012) Alginate: properties and biomedical applications. Prog Polym Sci 37:106–126. https://doi.org/10.1016/j.progpolymsci.2011.06.003
Rinaudo M (2014) Biomaterials based on a natural polysaccharide: alginate. TIP 17:92–96. https://doi.org/10.1016/S1405-888X(14)70322-5
Mirzaei A, Chen Z, Haghighat F, Yerushalmi L (2017) Removal of pharmaceuticals from water by homo/heterogonous Fenton-type processes—a review. Chemosphere 174:665–688. https://doi.org/10.1016/j.chemosphere.2017.02.019
Luo Y, Guo W, Ngo HH et al (2014) A review on the occurrence of micropollutants in the aquatic environment and their fate and removal during wastewater treatment. Sci Total Environ 473–474:619–641. https://doi.org/10.1016/j.scitotenv.2013.12.065
Petrie B, Barden R, Kasprzyk-Hordern B (2015) A review on emerging contaminants in wastewaters and the environment: current knowledge, understudied areas and recommendations for future monitoring. Water Res 72:3–27. https://doi.org/10.1016/j.watres.2014.08.053
Alves T, Girardi R, Pinheiro A (2017) Micropoluentes orgânicos: ocorrência, remoção e regulamentação. Rev Gestão Água da América Lat. https://doi.org/10.21168/rega.v14n0.p0-1
Vettorello G, Brandt VV, Dallazen MC et al (2017) Micropoluentes em água—O novo desafio emergente. Rev Cad Pedagógico. https://doi.org/10.22410/issn.1983-0882.v14i1a2017.1410
Zhang D, Gersberg RM, Ng WJ, Tan SK (2014) Removal of pharmaceuticals and personal care products in aquatic plant-based systems: a review. Environ Pollut 184:620–639. https://doi.org/10.1016/j.envpol.2013.09.009
Ho Y-S (2006) Review of second-order models for adsorption systems. J Hazard Mater 136:681–689. https://doi.org/10.1016/j.jhazmat.2005.12.043
Sun S-P, Li C-J, Sun J-H et al (2009) Decolorization of an azo dye orange G in aqueous solution by Fenton oxidation process: effect of system parameters and kinetic study. J Hazard Mater 161:1052–1057. https://doi.org/10.1016/j.jhazmat.2008.04.080
Acknowledgements
This study was financed in part by the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior—Brasil (CAPES)—Finance Code 001. We also are grateful for the financial support provided by CNPq and FAPERJ; the Scanning Electron Microscopy Laboratory (PPGQ-UERJ) for SEM analysis; and the Laboratory of Instrumental Characterization II (PPGQ-UERJ) for FTIR and FAAS analysis.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
da Silva Bezerra, D., França, R.J. & da Costa Marques, M.R. A Novel Catalytic Process for Degradation of Bisphenol A in Aqueous Solutions Using Fe Supported on Alginate/Carboxymethylcellulose. Catal Lett 151, 1477–1487 (2021). https://doi.org/10.1007/s10562-020-03403-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03403-9