Abstract
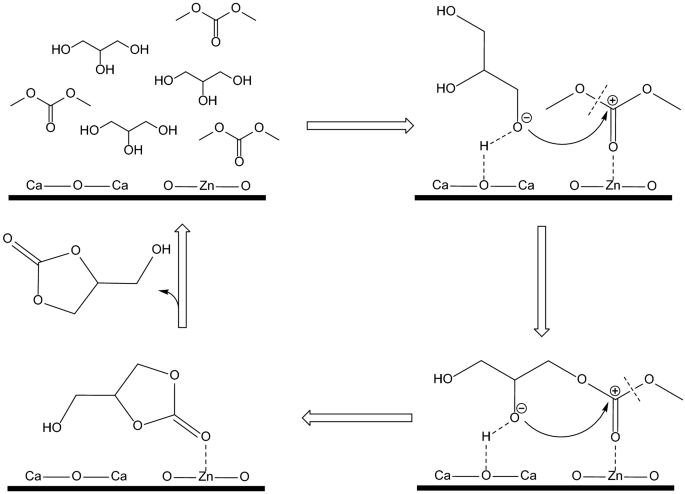
Magnetic mesoporous CoFe2O4@(CaO–ZnO) exhibited good catalytic activity in the transesterification of glycerol and dimethyl carbonate to yield glycerol carbonate. The glycerol conversion of 97.7% and glycerol carbonate yield of 96.9% could be obtained in this reaction system under the optimized reaction conditions. CoFe2O4@(CaO–ZnO) was characterized by a series of techniques including TPD, XRD, SEM, TEM, EDS, VSM and BET to evaluate the physico-chemical properties of the catalyst. It was demonstrated that the interaction of acid–base sites improved the catalysis performance of CoFe2O4@(CaO–ZnO): the strong basic sites were beneficial to the activation of glycerol to glyceroxide anion which could increase glycerol conversion, and the acid site contributes to the carbonyl activation of dimethyl carbonate, converts glyceroxide anion to glycerol carbonate, and improves the selectivity of glycerol carbonate. Meanwhile, the solid–liquid separation process after reaction would be simplified significantly considering CoFe2O4@(CaO–ZnO) could be easily separated from the reaction mixture under magnetic action.
Graphic Abstract
Acidic–basic bifunctional magnetic mesoporous CoFe2O4@(CaO–ZnO) was used as a solid base catalyst for the glycerol transesterification. Basic sites were beneficial to the activation of glycerol to glycerol oxygen anion to increase glycerol conversion and acidic sites were beneficial to carbonyl activation of DMC to increase glycerol carbonate selectivity. With magnetism, the solid base catalyst was easily recovered.










Similar content being viewed by others
References
Steven C, Arun M (2012) Nature 488:294–303
Shan R, Lu LL, Shi YY, Yuan HR, Shi JF (2018) Energy Covers Manag 178:277–289
Prashant VK (2007) J Phys Chem C 111:2834–2860
Jassinnee M, Hwai CO, Masjuki HH, Silitonga AS, Chen W, Kusumo F, Dharma S, Sebayang AH (2018) Energy Convers Manag 158:400–415
Chang JM, Guan XY, Pan SY, Jia ML, Chen Y, Fan HJ (2018) New J Chem 42:13074–13080
Norhasyimi R, Ahmad ZA, Abdul RM (2010) Renew Sustain Energy Rev 14:987–1000
Chun-Hui CZ, Jorge NB, Fan YX, Lu GQM (2008) Chem Soc Rev 37:527–549
Lozano FJ, Lozano R (2018) J Clean Prod 172:4162–4169
Zhang JG, Loris L, Gökalp G, Paul JD, Yan N (2018) Chin J Catal 39:1445–1452
Mario P, Rosaria C, Hiroshi K, Michele R, Cristina DP (2007) Angew Chem Int Ed 46:4434–4440
Georgios D, Stefan S, Harun T (2018) ACS Catal 8:6301–6333
Teng WK, Gek CN, Rozita Y, Mohamed KA (2014) Energy Convers Manag 88:484–497
Christy S, Noschese A, Lomelí-Rodriguez M, Greeves N, Lopez-Sanchez JA (2018) Curr Opin Green Sustain Chem 14:99–107
Liu P, Derchi M, Hensen EJM (2013) Appl Catal A 467:124–131
Huy N-P, Eun WS (2018) Appl Catal A 561:28–40
Chaves DM, Silva MJ (2019) New J Chem 43:3698–3706
Castro-Osma JA, Lamb KJ, North M (2016) ACS Catal 6:5012–5025
Wu YF, Song XH, Zhang JH, Li S, Yang XH, Wang HZ, Wei RP, Gao LJ, Zhang J, Xiao GM (2018) J Taiwan Inst Chem E 87:131–139
Zhou HX, Wang BW, Wang SP, Huang SY, Ma XB (2018) Chem Lett 47:1075–1078
Matthieu OS, Sonia A, Elisabeth PTG, Thierry D, Olivier C, Frederic G (2013) Green Chem 15:283–306
Khayoon MS, Hameed BH (2013) Appl Catal A 466:272–281
Song XH, Wu YF, Cai FF, Pan DH, Xiao GM (2017) Appl Catal A 532:77–85
Liu P, Margherita D, Emiel JMH (2014) Appl Catal B 144:135–143
Chiappe C, Rajamani S (2011) Pure Appl Chem 84:755–762
Sang CK, Yong HK, Hyuk L (2007) J Mol Catal B 49:75–78
Okoye PU, Abdullah AZ, Hameed BH (2016) J Taiwan Inst Chem Eng 68:51–58
Doyle AM, Alismaeel ZT, Albayati TM, Abbas AS (2017) Fuel 199:394–402
Shibasaki-Kitakawa N, Hiromori K, Ihara T, Nakashima K, Yonemoto T (2015) Fuel 139:11–17
Fidelis SHS, Tae KK, Sang DL (2011) Appl Catal A 401:220–225
Sasipim L, Thikumporn N, Kunchana B, Chawalit N (2013) Chem Eng J 225:616–624
Yu XH, Wen ZZ, Li HL, Tu ST, Yan JY (2011) Fuel 90:1868–1874
Yun HTY, Siow HT, Umer R, Aminul I, Mohd ZH, Keat TL (2014) Energy Convers Manag 88:1290–1296
Xie WL, Zhao LL (2013) Energy Convers Manag 76:55–62
Ana CAR, José SG, Josefa MMR, Ramón MT, David MA, Atonio JL, Pedro MT (2010) Catal Today 149:281–287
Xu Y, Wei J, Yao JL, Fu JL, Xue DS (2008) Mater Lett 62:1403–1405
Liu LH, Fan MM, Zhang PB, Jiang PP (2015) Fenxi Shiyanshi 34:1339–1342
Zheng LP, Xia SX, Lu XY (2015) Chin J Catal 36:1759–1765
Meng YL, Wang BY, Li SF, Tian SJ, Zhang MH (2013) Bioresour Technol 128:305–309
Wu YF, Song XH, Cai FF (2017) J Alloys Compd 720:360–368
José ROG, Olga GJA, Belén MM, Amaia PR, Camilo RL, Leire LL, Jesús TS, María CVV (2009) Appl Catal A 366:315–324
Corro G, Bañuelos F, Vidal E, Cebada S (2019) Catal Today. https://doi.org/10.1016/j.cattod.2019.03.007
Acknowledgements
The financial supports from the National Natural Science Foundation of China (NSFC) (No. 21978112), International Joint Research Laboratory for Biomass Conversion Technology at Jiangnan University and MOE & SAFEA for the 111 Project (B13025) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Zhang, P., Chen, Y., Zhu, M. et al. Acidic–Basic Bifunctional Magnetic Mesoporous CoFe2O4@(CaO–ZnO) for the Synthesis of Glycerol Carbonate. Catal Lett 150, 2863–2872 (2020). https://doi.org/10.1007/s10562-020-03191-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03191-2