Abstract
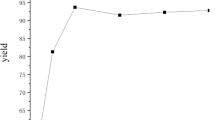
Pepsin was found to catalyze asymmetric cross aldol reactions of aromatic and polycyclicaromatic aldehydes with cyclic ketones in ionic liquids and deep eutectic solvents for the first time. Pepsin exhibited high catalytic activity and excellent stereoselectivity in ionic liquids and deep eutectic solvents in the presence of moderate water. High yields of up to 93.1%, excellent enantioselectivities of up to 96% ee, and good diastereoselectivities of up to 5:95 dr were achieved.
Graphic Abstract


Similar content being viewed by others
References
Guarneri A, van Berkel WJ, Paul CE (2019) Alternative coenzymes for biocatalysis. Curr Opin Biotechnol 60:63–71
Turner NJ, Kumar R (2018) Editorial overview: biocatalysis and biotransformation: the golden age of biocatalysis. Curr Opin Chem Biol 43:A1–A3
Dwivedee BP, Soni S, Sharma M, Bhaumik J, Laha JK, Banerjee UC (2018) Promiscuity of lipase catalyzed reactions for organic synthesis: a recent update. ChemistrySelect 3(9):2441–2466
He W-X, Xing X, Yang Z-J, Yu Y, Wang N, Yu X-Q (2019) Biocatalytic one-pot three-component synthesis of indoloquinolizines with high diastereoselectivity. Catal Lett 249(2):638–643
Koszelewski D, Ostaszewski R (2019) Biocatalytic promiscuity of lipases in carbon-phosphorus bond formation. ChemCatChem 11(10):2554–2558
Cai J-F, Guan Z, He Y-H (2011) The lipase-catalyzed asymmetric C-C Michael addition. J Mol Catal B: Enzym 68(3–4):240–244
Rizzo PVS, Boarin LA, Freitas IOM, Gomes RS, Beatriz A, Rinaldi AW, Domingues NLC (2014) The study of biocatalyzed thio-Michael reaction: a greener and multi-gram protocol. Tetrahedron Lett 55(2):430–434
Jiang L, Wang B, Li R-R, Shen S, Yu H-W, Ye L-D (2014) Catalytic promiscuity of Escherichia coli BioH esterase: application in the synthesis of 3,4-dihydropyran derivatives. Process Biochem 49(7):1135–1138
Wu L-L, Xiang Y, Yang D-C, Guan Z, He Y-H (2016) Biocatalytic asymmetric Mannich reaction of ketimines using wheat germ lipase. Catal Sci Technol 6(11):3963–3970
Eremeev NL, Zaitsev SY (2016) Porcine pancreatic lipase as a catalyst in organic synthesis. Mini-Rev Org Chem 13(1):78–85
Xu F, Xu J, Hu YJ, Lin XF, Wu Q (2016) One-pot bienzymatic cascade combining decarboxylative aldol reaction and kinetic resolution to synthesize chiral β-hydroxy ketone derivatives. RSC Adv 6(80):76829–76837
Li W, Liu DN, Geng X, Li ZQ, Gao RJ (2019) Real-time regulation of catalysis by remotecontrolled enzyme-conjugated gold nanorod composites for aldol reaction-based applications. Catal Sci Technol 9:2221–2230
Tian X-M, Zhang S-Q, Zheng L-Y (2016) Enzyme-catalyzed henry reaction in choline chloride-based deep eutectic solvents. J Microbiol Biotechnol 26(1):80–88
Foley M, Gavin DP, Deasy RE, Milner SE, Moody TS, Eccles KS, Lawrence SE, Maguire AR (2018) Dynamic kinetic resolution of 2-methyl-2-nitrocyclohexanol: combining the intramolecular nitroaldol (Henry) reaction & lipase catalysed resolution. Tetrahedron 74(13):1435–1443
Ranganathan S, Gärtner T, Wiemann LO, Sieber V (2015) A one pot reaction cascade of in situ hydrogen peroxide production and lipase mediated in situ production of peracids for the epoxidation of monoterpenes. J Mol Catal B 114:72–76
Branneby C, Carlqvist P, Magnusson A, Hult K, Brinck T, Berglund P (2003) Carbon-carbon bonds by hydrolytic enzymes. J Am Chem Soc 125(4):874–875
He Y-H, Li H-H, Chen Y-L, Xue Y, Yuan Y, Guan Z (2012) Chymopapain-catalyzed direct asymmetric aldol reaction. Adv Synth Catal 354(4):712–719
Chen X, Liu B-K, Kang H, Lin X-F (2011) A tandem Aldol condensation/dehydration co-catalyzed by acylase and N-heterocyclic compounds in organic media. J Mol Catal B: Enzym 68(1):71–76
Li C, Feng X-W, Wang N, Zhou Y-J, Yu X-Q (2008) Biocatalytic promiscuity: the first lipase-catalysed asymmetric aldol reaction. Green Chem 10(6):616–618
Carrea G, Ottolina G, Riva S (1995) Role of solvents in the control of enzyme selectivity in organic media. Trends Biotechnol 13(2):63–70
Klibanov AM (2001) Improving enzymes by using them in organic solvents. Nature 409(6817):241–246
Li H-H, He Y-H, Yuan Y, Guan Z (2011) Nuclease p1: a new biocatalyst for direct asymmetric aldol reaction under solvent-free conditions. Green Chem 13(1):185–189
Xie Z-B, Wang N, Zhou L-H, Wan F, He T, Le Z-G, Yu XQ (2013) Lipase-catalyzed stereoselective cross-aldol reaction promoted by water. ChemCatChem 5(7):1935–1940
Xie Z-B, Wang N, Jiang G-F, Yu X-Q (2013) Biocatalytic asymmetric aldol reaction in buffer solution. Tetrahedron Lett 54(8):945–948
Ksenia SE, Valentine PA (2018) Ionic liquids in whole-cell biocatalysis: a compromise between toxicity and efficiency. Biophys Rev 10(3):881–900
Zhang Y, Wang N, Xie Z-B, Zhou L-H, Yu X-Q (2014) Ionic liquid as a recyclable and efficient medium for lipase-catalyzed asymmetric cross aldol reaction. J Mol Catal B 110:100–110
Abbott AP, Capper G, Davies DL, Munro HL, Rasheed RK, Tambyrajah V (2001) Preparation of novel, moisture-stable, Lewis-acidic ionic liquids containing quaternary ammonium salts with functional side chains. Chem Commun 19:2010–2011
Smith L, Abbott AP, Ryder KS (2014) Deep eutectic solvents (DESs) and their applications. Chem Rev 114(21):11060–11082
Zhang M, Liu Y-H, Shang Z-R, Hu H-C, Zhang Z-H (2017) Supported molybdenum on graphene oxide/Fe3O4: an efficient, magnetically separable catalyst for one-pot construction of spiro-oxindole dihydropyridines in deep eutectic solvent under microwave irradiation. Catal Commun 88:39–44
Liu P, Hao J-W, Mo L-P, Zhang Z-H (2015) Recent advances in the application of deep eutectic solvents as sustainable media as well as catalysts in organic reactions. RSC Adv 5:48675–48704
Gao G, Wang P, Liu P, Zhang W-H, Mo L-P, Zhang Z-H (2018) Deep Eutectic solvent catalyzed one-pot synthesis of 4,7-dihydro-1H-pyrazolo[3,4-b]pyridine-5-carbonitriles. Chin J Org Chem 38:846–854
González-Martínez D, Gotor V, Gotor-Fernández V (2016) Application of deep eutectic solvents in promiscuous lipase catalysed aldol reactions. Eur J Org Chem 8:1513–1519
Choi YS, Yoo YJ (2012) A hydrophilic and hydrophobic organic solvent mixture enhances enzyme stability in organic media. Biotechnol Lett 34(6):1131–1135
Yang Z, Pan W (2005) Ionic liquids: Green solvents for nonaqueous biocatalysis. Enzyme Microb Technol 37(1):19–28
Zhang W, Zhao Z-Y, Wang Z, Guo C, Wang C-Y, Zhao R, Wang L (2017) Lipase-catalyzed synthesis of Indolyl 4H-chromenes via a multicomponent reaction in ionic liquid. Catalysts 7:185–194
Acknowledgements
We gratefully acknowledge the Natural Science Foundation of Guangdong Province (Grant No. 2018A030307022), Special Innovation Projects of Common Universities in Guangdong Province (Grant No. 2018KTSCX126).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors of this paper are aware of the submission and agree to its publication and declare no conflicts of interest.
Research Involving Human and Animal Participants
This study does not cover human participants and/or animal studies.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, Y., Chen, XY., Liang, XY. et al. Pepsin-Catalyzed Asymmetric Cross Aldol Reaction Promoted by Ionic Liquids and Deep Eutectic Solvents. Catal Lett 150, 2549–2557 (2020). https://doi.org/10.1007/s10562-020-03176-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03176-1