Abstract
Fe-doped mesoporous alumina (MA) was successfully synthesized via a facile one-pot synthesis method. The resulting Fe-doped MA samples possess typical mesoporous structure, relatively high BET surface area, and narrowed pore size diameter. Besides, the iron species are well-dispersed in the alumina matrix, and more importantly, some small oligonuclear iron oxide clusters are linked on the surface of alumina, leading to the elimination of strong acid sites. The reaction of phenol hydroxylation was preceded at room temperature for 2 h. The introduction of mesoporous structure in alumina would be favorable for the adsorption and diffusion process of reactant and product molecules, and also the well-dispersed iron species in alumina matrix and large amount of acid sites stimulate more active hydroxyl radicals which are greatly beneficial for the catalytic process, especially the elimination of strong acid sites would inhibit the over-oxidation reaction. In this case, the sample of 5Fe–MA displays the best catalytic performance especially with the extremely high dihydroxybenzene selectivity of 93.2%, and the good catalytic stability is also evidenced by the five times recycling tests.
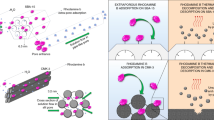
Graphic Abstract
In this work, the Fe-doped mesoporous alumina (MA) was successfully prepared. The iron species are well-dispersed in the alumina matrix, and some small oligonuclear iron oxide clusters are linked on the surface of alumina, leading to the elimination of strong acid sites. The advanced properties of mesoporous structure, well-dispersed metal active centers and lack of strong acid sites in Fe-doped MA are greatly beneficial for the catalytic process, especially for the dihydroxybenzene selectivity with extremely high value of 93.2%.











Similar content being viewed by others
References
Abbo HS, Titinchi SJJ (2012) Appl Catal A 435–436:148. https://doi.org/10.1016/j.apcata.2012.05.048
Pithakratanayothin S, Tongsri R, Chaisuwan T, Wongkasemjit S (2017) Catal Sci Technol 7:5413. https://doi.org/10.1039/C7CY00655A
Zhao Y, He G, Dai W, Chen H (2014) Ind Eng Chem Res 53:12566. https://doi.org/10.1021/ie501624u
Suresh S, Srivastava VC, Mishra IM (2012) Int J Energy Environ Eng 3:32. https://doi.org/10.1186/2251-6832-3-32
Shi F, Mu L, Yu P, Hu J, Zhang L (2014) J Mol Catal A 391:66. https://doi.org/10.1016/j.molcata.2014.04.006
Xu J, Ibrahim A-R, Hu X et al (2016) Microporous Mesoporous Mater 231:1. https://doi.org/10.1016/j.micromeso.2016.05.015
Abbo HS, Titinchi SJJ (2010) Top Catal 53:254. https://doi.org/10.1007/s11244-009-9408-9
Wang J, Park J-N, Jeong H-C et al (2004) Energy Fuels 18:470. https://doi.org/10.1021/ef0300904
Zuo Y, Song W, Dai C et al (2013) Appl Catal A 453:272. https://doi.org/10.1016/j.apcata.2012.12.027
Xia C, Long L, Zhu B, Lin M, Shu X (2016) Catal Commun 80:49. https://doi.org/10.1016/j.catcom.2016.03.011
He Z, Wu J, Gao B, He H (2015) ACS Appl Mater Interfaces 7:2424. https://doi.org/10.1021/am507134g
Zhou S, Shao B, Jin W et al (2019) Appl Catal A 575:159. https://doi.org/10.1016/j.apcata.2019.02.022
Li W, Cao C-Y, Wu L-Y, Ge M-F, Song W-G (2011) J Hazard Mater 198:143. https://doi.org/10.1016/j.jhazmat.2011.10.025
Badoga S, Sharma RV, Dalai AK, Adjaye J (2015) Appl Catal A 489:86. https://doi.org/10.1016/j.apcata.2014.10.008
Xu L, Wang F, Chen M et al (2016) ChemCatChem 8:2536. https://doi.org/10.1002/cctc.201600472
Ma Y, Zeng M, He J et al (2011) Appl Catal A 396:123. https://doi.org/10.1016/j.apcata.2011.02.001
Seki T, Ikeda S, Onaka M (2006) Microporous Mesoporous Mater 96:121. https://doi.org/10.1016/j.micromeso.2006.06.026
Abidli A, Hamoudi S, Belkacemi K (2015) Dalton Trans 44:9823. https://doi.org/10.1039/C4DT03630A
Morris SM, Fulvio PF, Jaroniec M (2008) J Am Chem Soc 130:15210. https://doi.org/10.1021/ja806429q
Fu L, Yang H (2014) J Phys Chem C 118:14299. https://doi.org/10.1021/jp502204h
Selli E, Rossetti I, Meloni D, Sini F, Forni L (2004) Appl Catal A 262:131. https://doi.org/10.1016/j.apcata.2003.11.016
Maurya MR, Titinchi SJJ, Chand S, Mishra IM (2002) J Mol Catal A 180:201. https://doi.org/10.1016/S1381-1169(01)00435-6
Cai W, Yu J, Anand C, Vinu A, Jaroniec M (2011) Chem Mater 23:1147. https://doi.org/10.1021/cm102512v
Zhou S, Jin W, Ding Y et al (2018) Dalton Trans 47:16862. https://doi.org/10.1039/C8DT03054E
Li Y, Feng Z, Lian Y et al (2005) Microporous Mesoporous Mater 84:41. https://doi.org/10.1016/j.micromeso.2005.05.021
Hensen EJM, Zhu Q, Hendrix MMRM et al (2004) J Catal 221:560. https://doi.org/10.1016/j.jcat.2003.09.024
Gao S, Zhou S, Yang F, Long S, Kong Y (2016) ChemistrySelect 1:1305. https://doi.org/10.1002/slct.201600184
Long S, Zhou S, Yang F, Lu K, Xi T, Kong Y (2016) RSC Adv 6:76064. https://doi.org/10.1039/C6RA13146H
He S, Sun C, Bai Z, Dai X, Wang B (2009) Appl Catal A 356:88. https://doi.org/10.1016/j.apcata.2008.12.024
Zou J-J, Liu Y, Pan L, Wang L, Zhang X (2010) Appl Catal B 95:439. https://doi.org/10.1016/j.apcatb.2010.01.024
Yamashita T, Hayes P (2008) Appl Surf Sci 254:2441. https://doi.org/10.1016/j.apsusc.2007.09.063
Grosvenor AP, Kobe BA, Biesinger MC, McIntyre NS (2004) Surf Interface Anal 36:1564. https://doi.org/10.1002/sia.1984
Liu C, Shan Y, Yang X, Ye X, Wu Y (1997) J Catal 168:35. https://doi.org/10.1006/jcat.1997.1612
Xin H, Liu J, Fan F et al (2008) Microporous Mesoporous Mater 113:231. https://doi.org/10.1016/j.micromeso.2007.11.022
Acknowledgements
This work was funded by the National Nature Science Foundation of China (51673040), the Natural Science Foundation of Jiangsu Province (BK20180366, BK20171357), the Fundamental Research Funds for the Central Universities (No. 3207048418), the Fundamental Research Funds for Central Universities (2242018k30008), the Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions (1107047002), the Fund Project for Transformation of Scientific and Technological Achievements of Jiangsu (BA2018045), the Prospective Joint Research Project of Jiangsu Province (BY2016076-01), the Opening Project of Guangxi Key Laboratory of Clean Pulp & Papermaking and Pollution Control (KF201605), and the Scientific Innovation Research Foundation of College Graduate in Jiangsu Province (KYLX16_0266).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Wang, Y., Zhou, Y., He, M. et al. Fe-Doped Mesoporous Alumina: Facile One-Pot Synthesis, Modified Surface-Acidity and Its Enhanced Catalytic Performance in Phenol Hydroxylation. Catal Lett 150, 2273–2282 (2020). https://doi.org/10.1007/s10562-020-03153-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03153-8