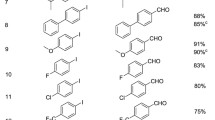
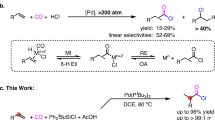
Abstract
The phosphine-free palladium-based catalytic system Pd(OAC)2/DABCO has been developed for the carbonylative transformations of aryl halides with formic acid. The DABCO acts as a base as well as N-donor ligand to coordinate with metal ions and stabilize Pd-catalytic sites during the reaction process. Using DCC as the activator of formic acid and PEG as the reaction media. The aryl halides have been conveniently transformed into the corresponding aldehydes in moderate to excellent yields.
Graphic Abstract
An efficient catalytic system Pd(OAc)2/ DABCO/ DCC for selective carbonylation of aryl halides with formic acid is described.




Similar content being viewed by others
References
Kollár L (2008) Modern carbonylation methods. Wiley, Hoboken
Wu X-F, Fang X, Wu L, Jackstell R, Neumann H, Beller M (2014) Transition-metal-catalyzed. Carbonylation reactions of olefins and alkynes: a personal account. Acc Chem Res 47(4):1041–1053
Dirksen A, Dawson PE (2008) Rapid oxime and hydrazone ligations with aromatic aldehydes for biomolecular labeling. Bioconjug Chem 19(12):2543–2548
Campos CA, Gianino JB, Pinkerton DM, Ashfeld BL (2011) Aryl aldehydes as traceless dielectrophiles in bifunctional titanocene-catalyzed propargylic C-X activations. Org Lett 13(20):5680–5683
Li Y, Hu Y, Wu X-F (2018) Non-noble metal-catalysed carbonylative transformations. Chem Soc Rev 47(1):172–194
Gautam P, Bhanage BM (2015) Recent advances in the transition metal catalyzed carbonylation of alkynes, arenes and aryl halides using CO surrogates. Catal Sci Technol 5(10):4663–4702
Shimomaki K, Murata K, Martin R, Iwasawa N (2017) Visible-light-driven carboxylation of aryl halides by the combined use of palladium and photoredox catalysts. J Am Chem Soc 139(28):9467–9470
Shao C, Lu A, Wang X, Zhou B, Guan X, Zhang Y (2017) Oxalic acid as the in situ carbon monoxide generator in palladium-catalyzed hydroxycarbonylation of arylhalides. Org Biomol Chem 15(23):5033–5040
Di Nicola A, Arcadi A, Rossi L (2016) BMIm HCO3: an ionic liquid with carboxylating properties. Synthesis of carbamate esters from amines. New J Chem 40(12):9895–9898
Sang R, Kucmierczyk P, Dong K, Franke R, Neumann H, Jackstell R, Beller M (2018) Palladium-catalyzed selective generation of CO from formic acid for carbonylation of alkenes. J Am Chem Soc 140(15):5217–5223
Brennführer A, Neumann H, Beller M (2009) Palladium-catalyzed carbonylation reactions of aryl halides and related compounds. Angew Chem Int Ed 48(23):4114–4133
Molla RA, Iqubal MA, Ghosh K, Roy AS, Islam SM (2014) Mesoporous poly-melamine-formaldehyde stabilized palladium nanoparticle (Pd@ mPMF) catalyzed mono and double carbonylation of aryl halides with amines. RSC Adv. 4(89):48177–48190
Ashfield L, Barnard CF (2007) Reductive carbonylation−an efficient and practical catalytic route for the conversion of aryl halides to aldehydes. Org Process Res Dev 11(1):39–43
Hajipour A-R, Tavangar-Rizi Z, Iranpoor N (2016) Palladium-catalyzed carbonylation of aryl halides: an efficient, heterogeneous and phosphine-free catalytic system for aminocarbonylation and alkoxycarbonylation employing Mo(CO)6 as a solid carbon monoxide source. RSC Adv 6(82):78468–78476
Ueda T, Konishi H, Manabe K (2013) Palladium-catalyzed reductive carbonylation of aryl halides with N-formylsaccharin as a CO source. Angew Chem Int Ed 52(33):8611–8615
Wu FP, Peng JB, Meng LS, Qi X, Wu XF (2017) Palladium-catalyzed ligand-controlled selective synthesis of aldehydes and acids from aryl halides and formic acid. ChemCatChem 9(16):3121–3124
Qi X, Jiang LB, Li HP, Wu XF (2015) A convenient palladium-catalyzed carbonylative suzuki coupling of aryl halides with formic acid as the carbon monoxide source. Chemistry 21(49):17650–17656
Wu F-P, Peng J-B, Qi X, Wu X-F (2017) Palladium-catalyzed carbonylative transformation of organic halides with formic acid as the coupling partner and CO source: synthesis of carboxylic acids. J Org Chem 82(18):9710–9714
Sun G, Lv X, Zhang Y, Lei M, Hu L (2017) Palladium-catalyzed formylation of aryl iodides with HCOOH as CO source. Org Lett 19(16):4235–4238
Neumann H, Kadyrov R, Wu XF, Beller M (2012) Palladium-catalyzed reductive carbonylation of aryl bromides with phosphinite ligands. Chem Asian J 7(10):2213–2216
Huang H, Li X, Yu C, Zhang Y, Mariano PS, Wang W (2017) Visible-Light-promoted nickel-and organic-dye-cocatalyzed formylation reaction of aryl halides and triflates and vinyl bromides with diethoxyacetic acid as a formyl equivalent. Angew Chem Int Ed 56(6):1500–1505
Cacchi S, Fabrizi G, Goggiamani A (2004) Palladium-catalyzed synthesis of aldehydes from aryl iodides and acetic formic anhydride. J Comb Chem 6(5):692–694
Iranpoor N, Firouzabadi H, Etemadi-Davan E, Rostami A, Rajabi Moghadam K (2015) Palladium-catalysed reductive carbonylation of aryl halides with iron pentacarbonyl for synthesis of aromatic aldehydes and deuterated aldehydes. Appl Organomet Chem 29(11):719–724
Acknowledgements
We are thankful to Persian Gulf University Research Council for partial support of this work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest associated with this publication. They also confirm that the manuscript has been read and approved by all 3 authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Molaei, E., Mohammadsaleh, F. & Niknam, K. Powerful and Phosphine-Free Palladium-Catalyzed Selective Formylation of Aryl Halides with Formic Acid as CO Source. Catal Lett 150, 1970–1975 (2020). https://doi.org/10.1007/s10562-020-03108-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03108-z