Abstract
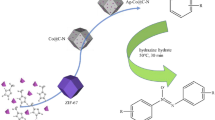
A Heterogeneous catalyst developed for selective reduction of nitroarenes to the analogous anilines using formic acid as hydrogen source. This catalytic procedure offers a simplistic path to prepare aromatic amines in good to excellent yields. Especially, even anilines functionalized with other potentially reducible moieties are obtained with high selectivity. Herein, we report convenient and stable bimetallic AgPd nanocatalyst supported on metal organic framework coated with polyaniline. Hydrogenation of nitroarenes gave analogues anilines with excellent yields at 90 °C in 6 h with no use of additives. Catalyst maintained stable performance in five repeated cycles.
Graphic Abstract



Similar content being viewed by others
References
Ergen S, Nişancı B, Metin Ö (2018) One-pot reductive amination of aldehydes with nitroarenes using formic acid as the hydrogen donor and mesoporous graphitic carbon nitride supported AgPd alloy nanoparticles as the heterogeneous catalyst. New J Chem 42(12):10000–10006
Gomez S, Peters JA, Maschmeyer T (2002) The reductive amination of aldehydes and ketones and the hydrogenation of nitriles: mechanistic aspects and selectivity control. Adv Synth Catal 344(10):1037–1057
Tripathi RP, Verma SS, Pandey J, Tiwari VK (2008) Recent development on catalytic reductive amination and applications. Curr Org Chem 12(13):1093–1115
Liang S, Monsen P, Hammond GB, Xu B (2016) Au/TiO2 catalyzed reductive amination of aldehydes and ketones using formic acid as reductant. Org Chem Front 3(4):505–509
Liu J-T, Yang S, Tang W, Yang Z, Xu J (2018) Iridium-catalyzed efficient reduction of ketones in water with formic acid as a hydride donor at low catalyst loading. Green Chem 20(9):2118–2124
Jiang L, Zhou P, Zhang Z, Chi Q, Jin S (2017) Environmentally friendly synthesis of secondary amines via one-pot reductive amination over a heterogeneous Co–Nx catalyst. New J Chem 41(20):11991–11997
Jeong G-Y, Singh AK, Kim M-G, Gyak K-W, Ryu U, Choi KM et al (2018) Metal-organic framework patterns and membranes with heterogeneous pores for flow-assisted switchable separations. Nat Commun 9(1):3968
Han X, Yang S, Schröder M (2019) Porous metal–organic frameworks as emerging sorbents for clean air. Nat Rev Chem 3(2):108–118
Zhao P, Fang H, Mukhopadhyay S, Li A, Rudić S, McPherson IJ et al (2019) Structural dynamics of a metal–organic framework induced by CO2 migration in its non-uniform porous structure. Nat Commun 10(1):999
Furukawa H, Cordova KE, O’Keeffe M, Yaghi OM (2013) The chemistry and applications of metal-organic frameworks. Science 341(6149):1230444
Shen K, Zhang L, Chen X, Liu L, Zhang D, Han Y et al (2018) Ordered macro-microporous metal-organic framework single crystals. Science 359(6372):206–210
Long JR, Yaghi OM (2009) The pervasive chemistry of metal–organic frameworks. Chem Soc Rev 38(5):1213–1214
Carrington EJ, McAnally CA, Fletcher AJ, Thompson SP, Warren M, Brammer L (2017) Solvent-switchable continuous-breathing behaviour in a diamondoid metal–organic framework and its influence on CO2 versus CH4 selectivity. Nat Chem 9:882
Eddaoudi M, Kim J, Rosi N, Vodak D, Wachter J, O'Keeffe M et al (2002) Systematic design of pore size and functionality in isoreticular MOFs and their application in methane storage. Science 295(5554):469–472
Cai G, Jiang H-L (2017) A modulator-induced defect-formation strategy to hierarchically porous metal-organic frameworks with high stability. Angew Chem Int Ed 56(2):563–567
Fang Z, Bueken B, De Vos DE, Fischer RA (2015) Defect-engineered metal-organic frameworks. Angew Chem Int Ed 54(25):7234–7254
Peng L, Zhang J, Xue Z, Han B, Sang X, Liu C et al (2014) Highly mesoporous metal–organic framework assembled in a switchable solvent. Nat Commun 5:4465
Zhang W, Liu Y, Lu G, Wang Y, Li S, Cui C et al (2015) Mesoporous Metal-organic frameworks with size-, shape-, and space-distribution-controlled pore structure. Adv Mater 27(18):2923–2929
Ramohlola KE, Monana GR, Hato MJ, Modibane KD, Molapo KM, Masikini M et al (2018) Polyaniline-metal organic framework nanocomposite as an efficient electrocatalyst for hydrogen evolution reaction. Composites B Eng 137:129–139
Sanchez F, Motta D, Roldan A, Hammond C, Villa A, Dimitratos N (2018) Hydrogen generation from additive-free formic acid decomposition under mild conditions by Pd/C: experimental and DFT studies. Top Catal 61(3):254–266
Nabid MR, Bide Y, Etemadi B (2017) Ag@Pd nanoparticles immobilized on a nitrogen-doped graphene carbon nanotube aerogel as a superb catalyst for the dehydrogenation of formic acid. New J Chem 41(19):10773–10779
Ma X, Feng Y, Li Y, Han Y, Lu G, Yang H et al (2015) Promoting effect of polyaniline on Pd catalysts for the formic acid electrooxidation reaction. Chin J Catal 36(7):943–951
Tabrizi NS, Xu Q, van der Pers NM, Lafont U, Schmidt-Ott A (2008) Synthesis of mixed metallic nanoparticles by spark discharge. J Nanopart Res 11(5):1209
Zhang X, Yang Y, Huang W, Yang Y, Wang Y, He C et al (2018) g-C3N4/UiO-66 nanohybrids with enhanced photocatalytic activities for the oxidation of dye under visible light irradiation. Mater Res Bull 99:349–358
Acknowledgements
The authors are thankful to CSIR-HRDG for their financial assistance [File no: 09/172(0084)/2017-EMR-1].
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Babel, V., Hiran, B.L. Heterogeneous AgPd Alloy Nanocatalyst for Selective Reduction of Aromatic Nitro Compounds Using Formic Acid as Hydrogen Source. Catal Lett 150, 1865–1869 (2020). https://doi.org/10.1007/s10562-020-03098-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-020-03098-y