Abstract
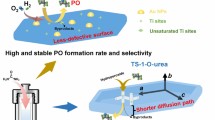
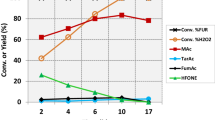
Na+ ions in TS-1 influence the results of liquid-phase oxidations of hydrocarbons seriously, and the avoiding of Na+ ion impurity in TS-1 synthesis is crucial for its catalytic application. In this paper, however, the solvent-free gas-phase epoxidation of propylene with H2O2 vapor (G-HPPO) was investigated over TS-1 zeolites with different Na+ ion content. Significant improvement in the performance of G-HPPO process was observed with NaOH solution hydrothermally modified TS-1 which had a Na/Ti ratio of 0.68. The performance of G-HPPO process was further enhanced when the Na/Ti ratio of hydrothermally modified TS-1 was increased to 1.0 via subsequent Na+ ion impregnation. The catalyst showed 16.9% propylene conversion, 97.5% PO selectivity and 79.3% H2O2 utility at a propylene to H2O2 ratio of around 5. On the other hand, when the Na+ ion content of the hydrothermally modified TS-1 was reduced via subsequent NH4+-exchange, the resulted catalyst exhibited a remarkably deteriorated G-HPPO process performance. By Combining the characterizations of UV–Raman, UV–vis and FT-IR with DFT calculation, it is concluded that in the NaOH solution hydrothermally modified TS-1 the Na+ ions served as counter cations of the silicon hydroxyls adjacent to “open” tetra-coordinated framework Ti sites. As a result, the local environment of the “open” Ti sites (with titanium hydroxyls) was adjusted and the Ti sites were properly activated. Whereas, in the case of excess Na+ ions were introduced into the TS-1 (for example Na/Ti ratio more than 1.0), the titanium hydroxyl of the “open” Ti sites would be occupied, to which the deteriorated G-HPPO process performance was ascribed.
Graphic Abstract








Similar content being viewed by others
References
Nijhuis TA, Makkee M, Moulijn JA, Weckhuysen BM (2006) Ind Eng Chem Res 45:3447–3459
Lin M, Xia CJ, Zhu B, Li H, Shu XT (2016) Chem Eng J 295:370–375
Zhao JL, Zhou JC, Su J, Guo HC, Wang XS, Gong WM (2007) AIChE J 53:3204–3209
Su J, Zhou JC, Liu CY, Wang XS, Guo HC (2010) Chin J Catal 31:1195–1199
Klemm E, Dietzsch E, Schwarz T, Kruppa T, Oliveira ALD, Becker F, Markowz G, Schirrmeister S, Schutte R, Caspary KJ, Schuth F, Honicke D (2008) Ind Eng Chem Res 47:2086–2090
Su J, Xiong G, Zhou J, Liu WH, Zhou DH, Wang GR, Wang XS, Guo HC (2012) J Catal 288:1–7
Perez Ferrandez DM, de Croon MHJM, Schouten JC, Nijhuis TA (2013) Ind Eng Chem Res 52:10126–10132
Perego C, Carati A, Ingallina P, Mantegazza MA, Bellussi G (2001) Appl Catal A 221:63–72
Fan WB, Duan R-G, Yokoi T, Wu P, Kubota Y, Tatsumi T (2008) J Am Chem Soc 130:10150–10164
Guo Q, Feng ZC, Li G, Fan FT, Li C (2013) J Phys Chem C 117:2844–2848
Zuo Y, Liu M, Zhang T, Hong LW, Guo XW, Song WC, Chen YS, Zhu PY, Jaye C, Fischer D (2015) RSC Adv 5:17897–17904
Signorile M, Crocellà V, Damin A, Rossi B, Lamberti C, Bonino F, Bordig S (2018) J Phys Chem C 122:9021–9034
Clerici MG, Ingallina P (1993) J Catal 140:71–83
Lamberti C, Bordig S, Arduino D, Zecchina A (1998) J Phys Chem B 102:6382–6390
Wells DH Jr, Delgass WN, Thomson KT (2004) J Am Chem Soc 126:2956–2962
Nie XW, Ji XJ, Chen YG, Guo XW, Song CS (2017) Mol Catal 441:150–167
Khouw CB, Davis ME (1995) J Catal 151:77–86
Tatsumi T, Koyano KA, Shimizu Y (2000) Appl Catal A 200:125–134
Li G, Wang XS, Yan HS, Chen YY, Su QS (2001) Appl Catal A 218:31–38
Capel-Sanchez MC, Campos-Martin JM, Fierro JLG (2003) Appl Catal A 246:69–77
Jin SQ, Feng ZC, Fan FT, Li C (2014) Catal Lett 145:468–481
Wang XS, Guo XW, Li G (2002) Catal Today 74:65–75
Yi YH, Zhou JC, Guo HC, Zhao JL, Su J, Wang L, Wang XS, Gong WM (2013) Angew Chem Int Ed 52:8446–8449
Xiong G, Cao YY, Guo ZD, Jia QY, Tian FP, Liu LP (2016) Phys Chem Chem Phys 18:190–196
Wang LL, Xiong G, Su J, Li P, Guo HC (2012) J Phys Chem C 116:9122–9131
Ricchiardi G, Damin A, Bordiga S, Lamberti C, Spano G, Rivetti F, Zecchina A (2001) J Am Chem Soc 123:11409–11419
Hijar CA, Jacubinas RM, Eckert J, Henson NJ, Hay PJ, Ott KC (2000) J Phys Chem B 104:12157–12164
Dong JC, Zhu HL, Xiang YJ, Wang Y, An PF, Gong Y, Liang YX, Qiu LM, Zheng A, Peng XX, Lin M, Xu GT, Guo ZY, Chen DL (2016) J Phys Chem C 120:20114–20124
Li MZ, Yan XY, Zhu MY, Wang MQ, Zhou DH (2018) Catal Sci Technol 8:4975–4984
Acknowledgements
This work is financially supported by the National Natural Science Foundation of China (No. 21603023).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Miao, C., He, N., Zhu, Q. et al. Effect of Sodium Ions on Catalytic Performance of TS-1 in Gas-Phase Epoxidation of Propylene with Hydrogen Peroxide Vapor. Catal Lett 150, 281–290 (2020). https://doi.org/10.1007/s10562-019-02948-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-019-02948-8