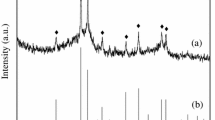
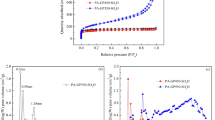
Abstract
The investigation describes detailed kinetic modelling of biodiesel synthesis catalysed by waste derived carbonaceous catalyst in a batch reactor. The modelling is conducted via three approaches i.e. power law equation, Eley–Rideal (E–R) and Langmuir–Hinshelwood (L–H) mechanism. Reversible and irreversible pathways with respect to triglycerides (TG) and fatty acid methyl esters (FAME) for first and second order reactions were considered for developing model equations. The kinetics of heterogeneous catalysis is studied using E–R and L–H mechanism to assess the exact rate controlling step by considering all the possible resistances offered by solid catalyst. The rate expressions for adsorption and desorption of individual reactant and product and surface chemical reaction were also developed. The influence of diffusional resistance offered by solid catalyst on conversion is also determined. The best fitted model is identified from calculated regression coefficients. The results revealed that glycerol desorption from catalyst surface given by E–R and L–H mechanisms is controlling the biodiesel synthesis process. The best suited model equation is considered for evaluating the kinetic parameters of the transesterification process. Thus, the study gives exact rate controlling step for heterogeneous catalysis to be used for reactor design and cost-effective production of biodiesel.
Graphic Abstract







Similar content being viewed by others
Abbreviations
- %x:
-
Percentage conversion
- TG:
-
Triglyceride (rubber seed oil)
- MA:
-
Methanol
- DG:
-
Diglyceride
- MG:
-
Monoglyceride
- Deff :
-
Effective diffusivity
- CA :
-
Concentration of component A (mol L−1)
- DAB :
-
Diffusivity of component A in component B
- r:
-
Rate (mol L−1 h−1)
- t:
-
Time (min)
- K:
-
Rate constant at constant methanol concentration
- \({{\text{K}^{\prime}}},\,{{\text{K}^{\prime\prime}}}\,{\text{and}}\,{{\text{K}^{\prime\prime}}}_{{\text{i}}}\) :
-
Constants
- (k1):
-
Rate constant for conversion of TG → DG
- (k2):
-
Rate constant for conversion of DG → TG
- (k3):
-
Rate constant for conversion of DG → MG
- (k4):
-
Rate constant for conversion of MG → DG
- (k5):
-
Rate constant for conversion of MG → G
- (k6):
-
Rate constant for conversion of G → MG
- (kt):
-
Rate constant for conversion of TG → G (for overall reaction)
- (kt−):
-
Rate constant for conversion of G → TG (for overall reaction)
- K1, K2, K3, K4 :
-
Constants
- (kad):
-
Rate constant for methanol adsorption in forward reaction
- (kad−):
-
Rate constant for methanol adsorption in backward reaction (ER)
- (ks):
-
Rate constant for forward surface reaction (ER)
- (ks−):
-
Rate constant for backward surface reaction (ER)
- (kd):
-
Rate constant for desorption of glycerol in forward reaction (ER)
- (kd−):
-
Rate constant for desorption of glycerol in backward reaction (ER)
- S:
-
Active sites
- \(\left({{{\text{K}^{\prime}}}_{{{\text{ad}}}} } \right)\) :
-
Equilibrium constant methanol adsorption(ER)
- \(\left({{{\text{K}^{\prime}}}_{{{\text{d}}}} } \right)\) :
-
Equilibrium constant for glycerol desorption (ER)
- Ct :
-
Concentration of total active sites
- Cv :
-
Concentration of vacant active sites
- K1, K2, K3, K4 :
-
Constants
- E:
-
Activation energy
- R:
-
Universal gas constant
- Ao :
-
Pre-exponential factor
References
Singh SP, Singh D (2010) Biodiesel production through the use of different sources and characterization of oils and their esters as the substitute of diesel: a review. Renew Sustain Energy Rev 14:200–216
Dhawane S, Kumar T, Halder G (2018) Recent advancement and prospective of heterogeneous carbonaceous catalysts in chemical and enzymatic transformation of biodiesel. Energy Convers Manage 167:176–202
Komers K, Skopal F, Stloukal R, Machek J (2002) Kinetics and mechanism of the KOH catalyzed methanolysis of rapeseed oil for biodiesel production. Eur J Lipid Sci Technol 104:728–737
Guan G, Kusakabe K, Sakurai N, Moriyama K (2009) Transesterification of vegetable oil to biodiesel fuel using acid catalysts in the presence of dimethyl ether. Fuel 88:81–86
Aniya VK, Muktham RK, Alka K, Satyavathi B (2015) Modeling and simulation of batch kinetics of non-edible karanja oil for biodiesel production: a mass transfer study. Fuel 161:137–145
Darnoko D, Cherya M (2000) Kinetics of palm oil transesterification in a batch reactor. J Am Oil Chem Soc 77:1263–1267
Freedman B, Buttefield RO, Pryde EH (1986) Transesterification kinetics of soybean oil. J Am Oil Chem Soc 63:1375–1380
Al-Sakkari EG, El-Sheltawy ST, Attiab NK, Mostafa SR (2017) Kinetic study of soybean oil methanolysis using cement kiln dust as a heterogeneous catalyst for biodiesel production. Appl Catal B 206:146–157
Hattori H, Shima M, Kabashima H (2000) Alcoholysis of ester and epoxide catalysed by solid bases. Stud Surf Sci Catal 130:3507–3512
Xiao Y, Gao L, Xiao G, Lv J (2010) Kinetics of the transesterification reaction catalysed by solid base in a fixed-bed reactor. Energy Fuels 24:5829–5833
Eze VC (2014) The use of meso-scale oscillatory baffled reactors for rapid screening of heterogeneously catalysed biodiesel production reactions. PhD Thesis, School of Chemical Engineering and Advanced Materials, Newcastle University, United Kingdom
Macleod C (2008) Evaluation of heterogeneous catalysts for biodiesel production. Ph.D. Thesis, School of Chemical Engineering and Advanced Materials, Newcastle University, United Kingdom, 2008
Mucino GEG, Romero RA, Ramos MJ, Jiménez RB, Natividad R (2016) Kinetics of transesterification of safflower oil to obtain biodiesel using heterogeneous catalysis. Int J Chem Reactor Eng 14:929–938
Dhawane S, Kumar T, Halder G (2015) Central composite design approach towards optimization of flamboyant pods derived steam activated carbon for its use as heterogeneous catalyst in transesterification of Hevea brasiliensis oil. Energy Convers Manage 100:277–287
Dhawane S, Kumar T, Halder G (2016) Biodiesel synthesis from Hevea brasiliensis oil employing carbon supported heterogeneous catalyst: optimization by Taguchi method. Renew Energ. 89:506–514
Dhawane S, Kumar T, Halder G (2016) Parametric effects and optimization on synthesis of iron (II) doped carbonaceous catalyst for the production of biodiesel. Energy Convers Manage 122:310–320
Dhawane S, Bora A, Kumar T, Halder G (2017) Parametric optimization of biodiesel synthesis from rubber seed oil using iron doped carbon catalyst by Taguchi approach. Renew Energ. 105:616–624
Talib NB, Triwahyono S, Jalil AA, Mamat CR, Salamun N, Fatah NA et al (2016) The Utilization of a cost effective Lapindo mud catalyst derivedfrom eruption waste for transesterification of waste oils. Energy Convers Manage 108:411–421
Vujicic DJ, Comic D, Zarubica A, Micic R, Boskovi G (2010) Kinetics of biodiesel synthesis from sunflower oil over CaO heterogeneous catalyst. Fuel 89:2054–2061
Dossin TF, Reyniers MF, Marin GB (2006) Kinetics of heterogeneously MgO-catalyzed transesterification. Appl. Catal. B: Environ. 62:35–45
Fogler HS (2005) Elements of Chemical Reaction Engineering, 3rd edn. Prentice-Hall, New Delhi
Fadhil Abdelrahman B, Aziz Akram M, Altamer Marwa H (2016) Potassium acetate supported on activated carbon for transesterification of new non-edible oil, bitter almond oil. Fuel 170:130–140
Lee T, Zubir ZA, Jamil FM, Matsumoto A, Yeoh FY (2014) Combustion and pyrolysis of activated carbon fibre from oil palm empty fruit bunch fibre assisted through chemical activation with acid treatment. J Anal Appl Pyrol 110:408–418
Halder G, Khan AA, Dhawane S (2015) Fluoride Sorption onto a Steam-Activated Biochar Derived from Cocos nucifera Shell. Clean-Soil Air Water 43(9999):1–10
Dai YM, Chen KT, Chen CC (2014) Study of the microwave lipid extraction from microalgae for biodiesel production. Chem Eng J 250:267–273
Lukić I, Kesić Ž, Maksimović S, Zdujić M, Krstić J, Skala D (2014) Kinetics of heterogeneous methanolysis of sunflower oil with CaO·ZnO catalyst: influence of different hydrodynamic conditions. Chemical Industry & Chemical Engineering Quarterly 20:425–439
. Kapil A., Wilson K., Lee A. F and Sadhukhan J., Kinetic Modeling Studies of Heterogeneously Catalyzed Biodiesel Synthesis Reactions, Ind. Eng. Chem. Res. 2011, 50, 4818-4830
Marinković DM, Miladinović MR, Avramović JM, Krstić IB, Stanković MV, Stamenković OS, Jovanović DM, Veljković VB (2018) Kinetic modeling and optimization of sunflower oil methanolysis catalyzed by spherically-shaped CaO/γ-Al2O3 catalyst. Energy Convers Manage 163:122–133
Acknowledgements
The authors would like to thank Rashmi Dhurandhar and Sumona Show for their technical support in characterisation of catalyst.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
Authors declare there is no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Dhawane, S.H., Al-Sakkari, E.G. & Halder, G. Kinetic Modelling of Heterogeneous Methanolysis Catalysed by Iron Induced on Microporous Carbon Supported Catalyst. Catal Lett 149, 3508–3524 (2019). https://doi.org/10.1007/s10562-019-02905-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-019-02905-5