Abstract
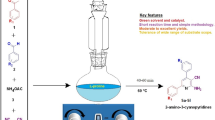
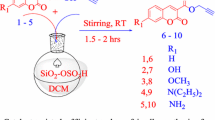
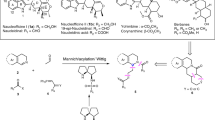
Although l-proline derived aminothioureas are being used as catalyst in asymmetric synthesis, their applications in multicomponent reactions are not yet reported in the literature. We report herein a l-proline derived secondary aminothiourea as a multifunctional catalyst in multicomponent reaction for synthesis of coumarin-based unsymmetrical trisubstituted methanes whose prototype has been found to be acetylcholinesterase inhibitor. The method requires low catalyst loading (5 mol%), very short reaction time (15–30 min) to give excellent yields. For the first time, it is observed that our bifunctional catalyst significantly increases the rate of conversion in comparison to that of two cooperative organocatalysts having similar catalytic sites.
Graphical Abstract
l-Proline derived secondary aminothiourea organocatalyst in multicomponent reaction: synthesis of coumarin derived trisubstituted methanes






Similar content being viewed by others
References
Mukherjee S, Yang JW, Hoffmann S, List B (2007) Chem Rev 107:5471
Saito S, Nakadai M, Yamamoto H (2001) Synlett 1245
Mase N, Tanaka F, Barbas CF III (2003) Org Lett 5:4369
Mase N, Tanaka F, Barbas CF III (2004) Angew Chem Int Ed 43:2420
Tang Z, Jiang F, Yu L-T, Cui X, Gong L-Z, Mi A-Q, Jiang Y-Z, Wu Y-D (2003) J Am Chem Soc 125:5262
Tang Z, Jiang F, Cui X, Gong L-Z, Mi A-Q, Jiang Y-Z, Wu Y-D (2004) Proc Natl Acad Sci USA 101:5755
Torii H, Nakadai M, Ishihara K, Saito S, Yamamoto H (2004) Angew Chem Int Ed 43:1983
Berkessel A, Koch B, Lex J (2004) Adv Synth Catal 346:1141
Silva F, Sawicki M, Gouverneur V (2006) Org Lett 8:5417
Obregón-Zúñiga A, Milán M, Juaristi E (2017) Org Lett 19:1108
Hernández JG, Juaristi E (2012) Chem Commun 48:5396
Serdyuk OV, Heckel CM, Tsogoeva SB (2013) Org Biomol Chem 11:7051
Siau W-Y, Wang J (2011) Catal Sci Technol 1:1298
Fu J-Y, Huang Q-C, Wang Q-W, Wang L-X, Xu X-Y (2010) Tetrahedron Lett 51:4870
Demir AS, Basceken S (2013) Tetrahedron Asymm 24:515
Kokotos C (2012) J Org Chem 77:1131
Fotaras S, Kokotos CG, Kokotos G (2012) Org Biomol Chem 10:5613
Khandelwal S, Tailor YK, Kumar M (2016) Current Organocatal 3:176
Clarke CJ, Tu W-C, Levers O, Bröhl A, Hallett JP (2018) Chem Rev 118:747
Peng XM, Damu GLV, Zhou C-H (2013) Curr Pharm Des 19:3884
Craig LA, Hong NS, McDonald RJ (2011) Neurosci Biobehav Rev 35:1397
Francis PT, Palmer AM, Snape M, Wilcock GK (1999) J Neurol Neurosurg Psychiatry 66:137
Peters J, Trovaslet M, Trapp M, Nachon F, Hill F, Royer E, Tehei M (2012) Phys Chem Chem Phys 14:6764
Baruah P, Basumatary G, Yesylevskyy SO, Aguan K, Bez G, Mitra S (2018) J Bimol Struct Dyn. https://doi.org/10.1080/07391102.2018.1465853
Bharti R, Parvin T (2015) Synth Commun 45:1442
Bharti R, Parvin T (2015) RSC Adv 5:66833
Bentley TW (2011) Org Biomol Chem 9:6685
Wang W-H, Abe T, Wang X-B, Kodama K, Hirose T, Zhang G-Y (2010) Tetrahedron 21:2925
Pedrosa R, Andrés JM, Gamarra A, Manzano R, Pérez-López C (2013) Tetrahedron 69:10811
Appel R, Chelli S, Tokuyasu T, Troshin K, Mayr H (2013) J Am Chem Soc 135:6579
Acknowledgements
The analytical services provided by Sophisticated Analytical Instrumentation Facility (SAIF), North Eastern Hill University, Shillong, India are acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Basumatary, G., Mohanta, R. & Bez, G. l-Proline Derived Secondary Aminothiourea Organocatalyst for Synthesis of Coumarin Derived Trisubstituted Methanes: Rate Enhancement by Bifunctional Catalyst over Cooperative Catalysis. Catal Lett 149, 2776–2786 (2019). https://doi.org/10.1007/s10562-019-02809-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-019-02809-4