Abstract
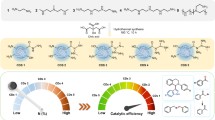
Four oxygen-sensitive meso-phenyl group with p-azo function Boron-dipyrromethene (BODIPY) dyes were synthesized, and their structures and photophysical properties were studied by 1H NMR, cyclic voltammetry and emission spectroscopy. Oxygen-sensing properties of BODIPYs are relevant to their catalytic activity under visible light. They were used to photocatalize oxidation of thioanisole. These metal-free visible-light catalytic agents could complete 100 % conversation from thioanisole to methylphenyl sulfoxide.
Graphical Abstract
Four oxygen-sensitive meso-phenyl group with p-azo function BODIPY dyes were synthesized, and their structures and photophysical properties were studied by 1H NMR, cyclic voltammetry and emission spectroscopy. Oxygen-sensing properties of BODIPYs are relevant to their catalytic activity under visible light. They were used to photocatalize oxidation of thioanisole. These metal-free visible-light catalytic agents could complete 100 % conversation from thioanisole to methylphenyl sulfoxide

.






Similar content being viewed by others
References
Yoon TP, Ischay MA, Du J (2010) Nat Chem 2:527
Narayanam JMR, Tucker JW, Stephenson CRJ (2009) J Am Chem Soc 131:8756
Condie AG, González-Gómez JC, Stephenson CRJ (2010) J Am Chem Soc 132:1464
Ischay MA, Lu Z, Yoon TP (2010) J Am Chem Soc 132:8572
Tucker JW, Nguyen JD, Narayanam JMR, Krabbeand SW, Stephenson CRJ (2010) Chem Commun 46:4985
Zen J, Liou S, Kumar AS, Hsia MS (2003) Angew Chem Int Ed 42:577
Neumann M, Füldner S, König B, Zeitler K (2011) Angew Chem Int Ed 50:951
Zhao Q, Huang C, Li F (2011) Chem Soc Rev 40:2508
Su F, Mathew SC, Möhlmann L, Antonietti M, Wang X, Blechert S (2011) Angew Chem Int Ed 50:65
Liu H, Feng W, Kee CW, Zhao Y, Leow D, Pan Y, Tan C (2010) Green Chem 12:953
Ulrich G, Ziessel R, Harriman A (2008) Angew Chem Int Ed 47:1184
Ziessel R, Harriman A (2011) Chem Commun 47:611
Baruah M, Qin W, Vallée RAL, Beljonne D, Rohand T, Dehaen W, Boens N (2005) Org Lett 7:4377
Cheng T, Xu Y, Zhang S, Zhu W, Qian X, Duan L (2008) J Am Chem Soc 130:16160
Bozdemir OA, Cakmak SE, Ekiz OO, Dana A, Akkaya EU (2011) Angew Chem Int Ed 50:10907
Descalzo AB, Xu HJ, Xue ZL, Hoffmann K, Shen Z, Weller MG, You XZ, Rurack K (2008) Org Lett 10:1581
Li WL, Xie ZG, Jing XB (2011) Catal Commun 16:94
Yang P, Zhao JZ, Wu WH, Yu XR, Liu Y (2012) J Org Chem 77:6166
Ishiguro SK, Hayashi M, Sawaki Y (1996) J Am Chem Soc 118:7265
Urano Y, Asanuma D, Hama Y, Koyama Y, Barrett T, Kamiya M, Nagano T, Watanabe T, Hasegawa A, Choyke PL, Kobayashi H (2009) Nat Med 15:104
Hoogendoorn S, Blom AEM, Willems LI, van der Marel GA, Overkleeft HS (2011) Org Lett 13:5656
Kowada T, Kikuta J, Kubo A, Ishii M, Maeda H, Mizukami S, Kikuchi K (2011) J Am Chem Soc 133:17772
Gabe Y, Urano Y, Kikuchi K, Kojima H, Nagano T (2004) J Am Chem Soc 126:3357
Jiao L, Yu C, Li J, Wang Z, Wu M, Hao E (2009) J Org Chem 74:7525
Burghart A, Kim H, Welch MB, Thoresen LH, Reibenspies J, Burgess K (1999) J Org Chem 64:7813
Lazarides T, Charalambidis G, Vuillamy A, Reglier M, Klontzas E, Froudakis G, Kuhri S, Guldi D, Coutsolelos AG (2011) Inorg Chem 50:8926
Werner T, Huber C, Heinl S, Kollmannsberger M, Daub J, Wolfbeis O, Fresenius S (1997) J Anal Chem 359:150
Kollmannsberger M, Gareis T, Heinl S, Breu J, Daub J (1997) Angew Chem Int Ed Engl 36:1333
Baruah M, Qin W, Flors C, Hofkens J, Vallee RAL, Beljonne D, Van der Auweraer M, De Borggraeve WM, Boens N (2006) J Phys Chem A 110:5998
Zhang GQ, Palmer GM, Dewhirst M, Fraser CL (2009) Nat Mater 8:747
Zhang GQ, Chen JB, Payne SJ (2007) J Am Chem Soc 129:8942
Galletta M, Puntoriero F, Campagna S, Chiorboli C, Quesada M, Goeb S, Ziessel R (2006) J Phys Chem A 110:434
Zhang GQ, Chen JB, Payne SJ, Kooi SE, Demas JN, Fraser CL (2007) J Am Chem Soc 129:8942
Fister JC III, Rank D, Harris JM (1995) Anal Chem 67:4269
Liu Q, Li YN, Zhang HH, Chen B, Tung CH, Wu LZ (2012) Chem Eur J 18:620
Acquaye JH, Muller JG, Takeuchi KJ (1993) Inorg Chem 32:160
Foote CS, Wexler S (1964) J Am Chem Soc 86:3880
Corey EJ, Taylor C (1964) J Am Chem Soc 105:3881
Yogo T, Urano Y, Ishitsuka Y, Maniwa F, Nagano T (2005) J Am Chem Soc 127:12162
Atilgan S, Ekmekci Z, Dogan AL, Guc D, Akkaya EU (2006) Chem Commun 42:4398
Acknowledgments
Financial support was provided by the National Natural Science Foundation of china (Project No. 21104075).
Author information
Authors and Affiliations
Corresponding author
Electronic Supplementary Material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Quan, L., Lin, W., Sun, T. et al. Green Photocatalysis with Oxygen Sensitive BODIPYs under Visible Light. Catal Lett 144, 308–313 (2014). https://doi.org/10.1007/s10562-013-1122-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10562-013-1122-3