Abstract
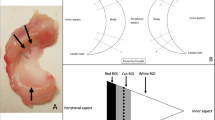
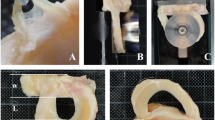
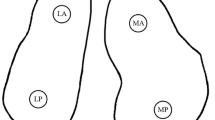
The common practice of freezing meniscal allograft tissue is limited due to the formation of damaging ice crystals. Vitrification, which eliminates the formation of damaging ice crystals, may allow the mechanical properties of meniscal allograft tissue to be maintained during storage and long-term preservation. The primary objective of this study was to investigate the differences between fresh, frozen, and vitrified porcine lateral menisci examining compressive mechanical properties in the axial direction. Unconfined compressive stress-relaxation testing was conducted to quantify the mechanical properties of fresh, frozen and vitrified porcine lateral menisci. The compressive mechanical properties investigated were peak and equilibrium stress, secant, instantaneous and equilibrium modulus, percent stress-relaxation, and relaxation time constants from three-term Prony series. Frozen menisci exhibited inferior compressive mechanical properties in comparison with fresh menisci (significant differences in peak and equilibrium stress, and secant, instantaneous and equilibrium modulus) and vitrified menisci (significant differences in peak stress, and secant and instantaneous modulus). Interestingly, fresh and vitrified menisci exhibited comparable compressive mechanical properties (stress, modulus and relaxation parameters). These findings are significant because (1) vitrification was successful in maintaining mechanical properties at values similar to fresh menisci, (2) compressive mechanical properties of fresh menisci were characterized providing a baseline for future research, and (3) freezing affected mechanical properties confirming that freezing should be used with caution in future investigations of meniscal mechanical properties. Vitrification was superior to freezing for preserving compressive mechanical properties of menisci which is an important advance for vitrification as a preservation option for meniscal allograft transplantation.



Similar content being viewed by others
Data availability
Data will be made available on reasonable request.
References
Abdelgaied A, Stanley M, Galfe M, Berry H, Ingham E, Fisher J (2015) Comparison of the biomechanical tensile and compressive properties of decellularised and natural porcine meniscus. J Biomech 48(8):1389–1396. https://doi.org/10.1016/j.jbiomech.2015.02.044
Andrews SH, Rattner JB, Shrive NG, Ronsky JL (2015) Swelling significantly affects the material properties of the menisci in compression. J Biomech 48(8):1485–1489. https://doi.org/10.1016/j.jbiomech.2015.02.001
Binnet MS, Akan B, Kaya A (2012) Lyophilised medial meniscus transplantations in ACL-deficient knees: a 19-year follow-up. Knee Surg Sports Traumatol Arthrosc 20(1):109–113. https://doi.org/10.1007/s00167-011-1556-3
Brockbank KG (1992) U.S. Patent No. US5131850A, U.S. patent and trademark office, Washington
Fabbriciani C, Lucania L, Milano G, Schiavone Panni A, Evangelisti M (1997) Meniscal allografts: cryopreservation vs deep-frozen technique. An experimental study in goats. Knee Surg Sports Traumatol Arthrosc 5(2):124–134. https://doi.org/10.1007/s001670050038
Fahy GM, Wowk B (2015) Principles of cryopreservation by vitrification. In: Wolkers W, Oldenhof H (eds) Cryopreservation and freeze-drying protocols, 3rd edn. Springer, New York, pp 21–82
Fahy GM, MacFarlane DR, Angell CA, Meryman HT (1984) Vitrification as an approach to cryopreservation. Cryobiology 21(4):407–426. https://doi.org/10.1016/0011-2240(84)90079-8
Finger EB, Bischof JC (2018) Cryopreservation by vitrification. Curr Opin Organ Transplant 23(3):353–360. https://doi.org/10.1097/mot.0000000000000534
Gabrion A, Aimedieu P, Laya Z, Havet E, Mertl P, Grebe R, Laude M (2005) Relationship between ultrastructure and biomechanical properties of the knee meniscus. Surg Radiol Anat 27(6):507–510. https://doi.org/10.1007/s00276-005-0031-6
Gelber PE, Gonzalez G, Lloreta JL, Reina F, Caceres E, Monllau JC (2008) Freezing causes changes in the meniscus collagen net: a new ultrastructural meniscus disarray scale. Knee Surg Sports Traumatol Arthrosc 16(4):353–359. https://doi.org/10.1007/s00167-007-0457-y
Gelber PE, Gonzalez G, Torres R, Giralt NG, Caceres E, Monllau JC (2009) Cryopreservation does not alter the ultrastructure of the meniscus. Knee Surg Sports Traumatol Arthrosc 17(6):639–644. https://doi.org/10.1007/s00167-009-0736-x
Jacquet C, Erivan R, Argenson JN, Parratte S, Ollivier M (2018) Effect of 3 preservation methods (freezing, cryopreservation, and freezing irradiation) on human menisci ultrastructure: an ex vivo comparative study with fresh tissue as a gold standard. Am J Sports Med 46(12):2899–2904. https://doi.org/10.1177/0363546518790504
Jomha NM, Anoop PC, Bagnall K, McGann LE (2002) Effects of increasing concentrations of dimethyl sulfoxide during cryopreservation of porcine articular cartilage. Cell Preserv Technol 1(2):111–120. https://doi.org/10.1089/15383440232088261
Jomha NM, Anoop PC, McGann LE (2004) Intramatrix events during cryopreservation of porcine articular cartilage using rapid cooling. J Orthop Res 22(1):152–157. https://doi.org/10.1016/s0736-0266(03)00158-x
Jomha NM, Elliott JAW, Law GK, Maghdoori B, Forbes JF, Abazari A, Adesida AB, Laouar L, Zhou X, McGann LE (2012) Vitrification of intact human articular cartilage. Biomaterials 33(26):6061–6068. https://doi.org/10.1016/j.biomaterials.2012.05.007
Junkin DM, Patel JK, Johnson DL (2009) Meniscal allograft transplantation. In: McKeon BP, Bono JV, Richmond JC (eds) Knee arthroscopy. Springer, New York, pp 41–65
Lakes EH, Matuska AM, McFetridge PS, Allen KD (2016) Mechanical integrity of a decellularized and laser drilled medial meniscus. J Biomech Eng 138(3):1–12. https://doi.org/10.1115/1.4032381
Lewis PB, Williams JM, Hallab N, Virdi A, Yanke A, Cole BJ (2008) Multiple freeze-thaw cycled meniscal allograft tissue: a biomechanical, biochemical, and histologic analysis. J Orthop Res 26(1):49–55. https://doi.org/10.1002/jor.20473
McGann L, Farrant J (1976) Survival of tissue culture cells frozen by a two-step procedure to −196 °C. II. Warming rate and concentration of dimethyl sulphoxide. Cryobiology 13:269–273. https://doi.org/10.1016/0011-2240(76)90107-3
Mickiewicz P, Binkowski M, Bursig H, Wróbel Z (2014) Preservation and sterilization methods of the meniscal allografts: literature review. Cell Tissue Bank 15(3):307–317. https://doi.org/10.1007/s10561-013-9396-7
Miller RH, Azar FM (2008) Knee injuries. In: Canale ST, Beaty JH (eds) Campbell’s operative orthopaedics, 11th edn. Mosby, Philadelphia, pp 2395–2600
Muldrew K, Novak K, Yang H, Zernicke R, Schachar NS, McGann LE (2000) Cryobiology of articular cartilage: ice morphology and recovery of chondrocytes. Cryobiology 40(2):102–109. https://doi.org/10.1006/cryo.2000.2236
Pegg DE, Diaper MP (1990) Freezing versus vitrification: basic principles. In: Smit Sibinga CT, Das PC, Meryman HT (ed) Cryopreservation and low temperature biology in blood transfusion, In Proceedings of the fourteenth international symposium on blood transfusion, Groningen 1989, organised by the Red Cross Blood Bank Groningen-Drenthe. Kluwer Academic Publishers, Boston, pp. 55–69
Sakai A, Engelmann F (2007) Vitrification, encapsulation-vitrification and droplet-vitrification: a review. CryoLetters 28(3):151–172
Takroni TA, Yu H, Laouar L, Adesida AB, Elliott J, Jomha NM (2017) Ethylene glycol and glycerol loading and unloading in porcine meniscal tissue. Cryobiology 74:50–60. https://doi.org/10.1016/j.cryobiol.2016.12.003
Verdonk PC, Demurie A, Almqvist KF, Veys EM, Verbruggen G, Verdonk R (2005) Transplantation of viable meniscal allograft: Survivorship analysis and clinical outcome of one hundred cases. J Bone Jt Surg Am 87(4):715–724. https://doi.org/10.2106/jbjs.c.01344
Verdonk PC, Demurie A, Almqvist KF, Veys EM, Verbruggen G, Verdonk R (2006) Transplantation of viable meniscal allograft: surgical technique. J Bone Jt Surg Am 88(1):109–118. https://doi.org/10.2106/jbjs.e.00875
Willett TL, Whiteside R, Wild PM, Wyss UP, Anastassiades T (2005) Artefacts in the mechanical characterization of porcine articular cartilage due to freezing. Proc Inst Mech Eng H 219(1):23–29. https://doi.org/10.1243/095441105X9200
Wu K, Laouar L, Shardt N, Chen Z, Elliott JAW, McGann LE, Jomha NM (2018) Comparison of three multi-cryoprotectant loading protocols for vitrification of articular cartilage. Cryobiology 85:153. https://doi.org/10.1016/j.cryobiol.2018.10.132
Wu K, Shardt N, Laouar L, Elliott JA, Jomha NM (2021) Vitrification of particulated articular cartilage via calculated protocols. NPJ Regen Med 6:15. https://doi.org/10.1038/s41536-021-00123-5
Acknowledgements
This work was supported by the Edmonton Orthopaedic Research Committee (NMJ) and the Natural Sciences and Engineering Research Council of Canada (GMT: 04560).
Author information
Authors and Affiliations
Contributions
JS, LW, NMJ, SA and GMT contributed to the conception and design of the study, JS, KW, and LL contributed to the acquisition of data, all authors contributed to the analysis and interpretation of data, JS and GMT contributed to drafting the article, and all authors contributed to reviewing, revising and approving the article.
Corresponding author
Ethics declarations
Conflict of interest
Dr. N.M. Jomha is a co-inventor on US and Canadian patents for articular cartilage preservation: Jomha NM, McGann LE, Elliott JAW, Law G, Forbes F, Torgabeh AA, Maghdoori B, Weiss A, University of Alberta (2014) “Cryopreservation of articular cartilage”. US Patent 8,758,988; Canada 2,788,202.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, J., Westover, L., Wu, K. et al. Compressive mechanical properties of vitrified porcine menisci are superior to frozen and similar to fresh porcine menisci. Cell Tissue Bank 24, 737–745 (2023). https://doi.org/10.1007/s10561-022-10065-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-022-10065-x