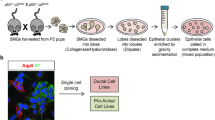
Abstract
Serous Acinar Cells (ACs) are mature and functional secretory epithelial cells that develop and complete through other stem cells at the end of the ductal system. So, the regeneration of the salivary gland damaged by radiation does not occur without cell therapy. Todays, an accessible tissue like the Periodontal Ligament (PDL) of the tooth was considered to easily extract the Mesenchymal Stem Cells (MSCs). In-vitro differentiation of stem cells before transplantation to damaged tissue reduces the risk of tumorigenesis. This study was conducted to evaluate the feasibility of differentiation of PDLSCs into salivary acinar cells by a co-culture system. PDLSCs were isolated from adult human PDL tissue and co-cultured with rat parotid ACs using an indirect co-culture system. The transdifferentiation of PDLSCs was evaluated by PCR of Aquaporin 5 (AQP5) and Carbonic anhydrase 6 (CA6) genes, then quantitative real-time PCR was used to measure the gene expression levels. The data were analyzed by ANOVA. Specific bond with the correct size on 6% acrylamide gel and TBE5X buffer showed the expression of AQP5 and CA6 in PDLSCs co-cultured with acinar cells. RT-PCR revealed co-cultured PDLSCs with or without KGF (Keratinocyte Growth Factor) showed significantly increased expression of AQP5 genes in compared to the initial PDLSCs. Expression of AQP5 and CA6, indicating successful transdifferentiation of PDLSCs into ACs, in co-culture system for 3 weeks.
Graphical abstract






Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
References
Chang Y-H, Chu T-Y, Ding D-C (2017) WNT/β-Catenin signaling pathway regulates non-tumorigenesis of human embryonic stem cells co-cultured with human umbilical cord mesenchymal stem cells. Sci Rep 7(1):1–10
Chen M-H, Chen R-S, Hsu Y-H et al (2005) Proliferation and phenotypic preservation of rat parotid acinar cells. Tissue Eng 11(3–4):526–534
Diomede F, Rajan TS, D’Aurora M et al (2017a) Stemness characteristics of periodontal ligament stem cells from donors and multiple sclerosis patients: a comparative study. Stem Cells Int 2017:1606125
Diomede F, Thangavelu SR, Merciaro I et al (2017b) Porphyromonas gingivalis lipopolysaccharide stimulation in human periodontal ligament stem cells: role of epigenetic modifications to the inflammation. Eur J Histochem 61(3):2826. Published 2017b Aug 25.
Feng J, van der Zwaag M, Stokman MA et al (2009) Isolation and characterization of human salivary gland cells for stem cell transplantation to reduce radiation-induced hyposalivation. Radiother Oncol 92(3):466–471
François S, Bensidhoum M, Mouiseddine M et al (2006) Local irradiation not only induces homing of human mesenchymal stem cells at exposed sites but promotes their widespread engraftment to multiple organs: a study of their quantitative distribution after irradiation damage. Stem Cells 24(4):1020–1029
Goers L, Freemont P, Polizzi KM (2014) Co-culture systems and technologies: taking synthetic biology to the next level. J R Soc Interface 11(96):20140065
Gronthos S, Mankani M, Brahim J et al (2000) Postnatal human dental pulp stem cells (DPSCs) in vitro and in vivo. Proc Natl Acad Sci 97(25):13625–13630
Hossain A, Gumin J, Gao F et al (2015) Mesenchymal stem cells isolated from human gliomas increase proliferation and maintain stemness of glioma stem cells through the IL-6/gp130/STAT3 pathway. Stem Cells 33(8):2400–2415
Hsieh MS, Jeng YM, Jhuang YL, Chou YH, Lin CY (2016) Carbonic anhydrase VI: a novel marker for salivary serous acinar differentiation and its application to discriminate acinic cell carcinoma from mammary analogue secretory carcinoma of the salivary gland. Histopathology 68(5):641–647
Huang W, Chang M, Tsai K et al (2013) Mesenchymal stem cells promote growth and angiogenesis of tumors in mice. Oncogene 32(37):4343–4354
Hussein AM, Darwish ZE, Raslan HS et al (2021) Dental stem cells (concepts and applications. Alexandria Dental Journal, 46(1) Section (A): Oral and Maxillofacial Surgery, Oral Medicine, Periodontology, Oral Radiology and Pathology, Oral Biology 66–71
Jasmer KJ, Gilman KE, Muñoz Forti K et al (2020) Radiation-induced salivary gland dysfunction: mechanisms, therapeutics and future directions. J Clin Med 9(12):4095
Kuehnle I, Goodell MA (2002) The therapeutic potential of stem cells from adults. BMJ 325(7360):372–376
Lee I-C, Wang J-H, Lee Y-T et al (2007) The differentiation of mesenchymal stem cells by mechanical stress or/and co-culture system. Biochem Biophys Res Commun 352(1):147–152
Li C, Zhao H, Wang B (2021) Mesenchymal stem/stromal cells: developmental origin, tumorigenesis and translational cancer therapeutics. Transl Oncol 14(1):100948
Lim J-Y, Ra J, C, Shin I. S, et al (2013a) Systemic transplantation of human adipose tissue-derived mesenchymal stem cells for the regeneration of irradiation-induced salivary gland damage. PLoS ONE 8(8):e71167
Lim J-Y, Yi T, Choi J-S et al (2013b) Intraglandular transplantation of bone marrow-derived clonal mesenchymal stem cells for amelioration of post-irradiation salivary gland damage. Oral Oncol 49(2):136–143
Lin C-Y, Lee B-S, Liao C-C et al (2007) Transdifferentiation of bone marrow stem cells into acinar cells using a double chamber system. J. Formosan Med. Assoc. 106(1):1–7
Lin C-Y, Chang F-H, Chen C-Y et al (2011) Cell therapy for salivary gland regeneration. J Dent Res 90(3):341–346
Lombaert IM, Wierenga PK, Kok T et al (2006) Mobilization of bone marrow stem cells by granulocyte colony-stimulating factor ameliorates radiation-induced damage to salivary glands. Clin Cancer Res 12(6):1804–1812
Lombaert IM, Brunsting JF, Wierenga PK et al (2008) Rescue of salivary gland function after stem cell transplantation in irradiated glands. PLoS ONE 3(4):e2063
Lombaert I, Movahednia MM, Adine C et al (2017) Concise review: salivary gland regeneration: therapeutic approaches from stem cells to tissue organoids. Stem Cells 35(1):97–105
Ma T, Song Y, Gillespie A, Carlson EJ et al (1999) Defective secretion of saliva in transgenic mice lacking aquaporin-5 water channels. J Biol Chem 274(29):20071–20074
Maimets M, Rocchi C, Bron R et al (2016) Long-term in vitro expansion of salivary gland stem cells driven by Wnt signals. Stem Cell Reports 6(1):150–162
Maria OM, Tran SD (2011) Human mesenchymal stem cells cultured with salivary gland biopsies adopt an epithelial phenotype. Stem Cells Dev 20(6):959–967
McLean K, Gong Y, Choi Y et al (2011) Human ovarian carcinoma–associated mesenchymal stem cells regulate cancer stem cells and tumorigenesis via altered BMP production. J Clin Investig 121(8):3206–3219
Miura M, Gronthos S, Zhao M et al (2003) SHED: stem cells from human exfoliated deciduous teeth. Proc Natl Acad Sci 100(10):5807–5812
Nanduri LS, Maimets M, Pringle SA et al (2011) Regeneration of irradiated salivary glands with stem cell marker expressing cells. Radiotherapy Oncolog 99(3):367–372
Nanduri LS, Lombaert IM, van der Zwaag M et al (2013) Salisphere derived c-Kit+ cell transplantation restores tissue homeostasis in irradiated salivary gland. Radiother Oncol 108(3):458–463
Nishiwaki S, Nakayama T, Saito S et al (2012) Efficacy and safety of human adipose tissue-derived mesenchymal stem cells for supporting hematopoiesis. Int J Hematol 96(3):295–300
Nusse R, Fuerer C, Ching W et al (2008) Wnt signaling and stem cell control. Paper presented at the Cold Spring Harbor Symposia on Quantitative Biology
Prockop DJ (2009) Repair of tissues by adult stem/progenitor cells (MSCs): controversies, myths, and changing paradigms. Mol Ther 17(6):939–946
Roato I, Chinigò G, Genova T et al (2021) Oral cavity as a source of mesenchymal stem cells useful for regenerative medicine in dentistry. Biomedicines 9(9):1085
Rompianesi G, Hann A, Komolafe O et al (2017) Serum amylase and lipase and urinary trypsinogen and amylase for diagnosis of acute pancreatitis. Cochrane Database of Systematic Reviews (4)
Rubio R, Gutierrez-Aranda I, Sáez-Castillo A et al (2013) The differentiation stage of p53-Rb-deficient bone marrow mesenchymal stem cells imposes the phenotype of in vivo sarcoma development. Oncogene 32(41):4970–4980
Seo B-M, Miura M, Gronthos S et al (2004) Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet 364(9429):149–155
Soundara Rajan T, Giacoppo S, Diomede F et al (2017) Human periodontal ligament stem cells secretome from multiple sclerosis patients suppresses NALP3 inflammasome activation in experimental autoimmune encephalomyelitis. Int J Immunopathol Pharmacol 30(3):238–252
Stephens LC, Schultheiss TE, Price RE et al (1991) Radiation apoptosis of serous acinar cells of salivary and lacrimal glands. Cancer 67(6):1539–1543
Sugito T, Kagami H, Hata K et al (2004) Transplantation of cultured salivary gland cells into an atrophic salivary gland. Cell Transplant 13(6):691–700
Sumita Y, Liu Y, Khalili S et al (2011) Bone marrow-derived cells rescue salivary gland function in mice with head and neck irradiation. Int J Biochem Cell Biol 43(1):80–87
Tran S, Wang J, Bandyopadhyay, et al (2005) Primary culture of polarized human salivary epithelial cells for use in developing an artificial salivary gland. Tissue Eng 11(1–2):172–181
Tran SD, Liu Y, Xia D et al (2013) Paracrine effects of bone marrow soup restore organ function, regeneration, and repair in salivary glands damaged by irradiation. PLoS ONE 8(4):e61632
Zhang J, An Y, Gao L-N et al (2012) The effect of aging on the pluripotential capacity and regenerative potential of human periodontal ligament stem cells. Biomaterials 33(29):6974–6986
Zhu W, Liang M (2015) Periodontal ligament stem cells: current status, concerns, and future prospects. Stem Cells Int 2015
Acknowledgements
This research has been supported and funded by Vice-Chancellor in Research Affaire, School of dentistry, Tehran University of Medical Science, Tehran, Iran. Grant number: 98-02-69-42401.
Funding
This study funded by Vice-Chancellor in Research Affairs, School of Dentistry, Tehran University of Medical Science, Tehran, Iran.
Author information
Authors and Affiliations
Contributions
NS Substantial contribution to the conception and design, Drafting the manuscript, critically revising the manuscript for important intellectual content, Approval of the final version submitted for publication. “Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.” FB Substantial contribution to the acquisition, analysis of data, critically revising the manuscript for important intellectual content, Approval of the final version submitted for publication. “Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.” MK Substantial contribution to the analysis and interpretation of data, Drafting the manuscript, Approval of the final version submitted for publication. “Agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.”
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interests related to this study.The authors have no conflict of interest to declare.
Ethics approval and consent to participate
The protocol of this study was approved by the ethics committee of Tehran University of Medical Science (IR.TUMS.VCR.REC.1399.482). Informed consents were obtained from all individual participants who took part in this study. Also, informed consent were taken from a parent and/or legal guardian for human participants that are minors. All procedures which were involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Human or animal rights
The protocol of this study was approved by the ethics committee of Tehran University of Medical Science (IR.TUMS.VCR.REC.1399.482) and this study was conducted in accordance with the Declaration of Helsinki.
Consent to publish
In this study there is no details, images, or videos relating to an individual person that needs to be agreed to publish.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sheykhbahaei, N., Bayramzadeh, F. & Koopaie, M. Transdifferentiation of periodontal ligament stem cells into acinar cells using an indirect co-culture system. Cell Tissue Bank 24, 241–251 (2023). https://doi.org/10.1007/s10561-022-10029-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-022-10029-1