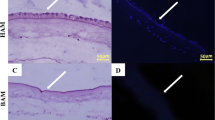
Abstract
Tissue engineering is an interdisciplinary field that applies the principles of engineering and life sciences toward the development of biological substitutes that restore, maintain, or improve tissue function. The aims of this work were to compare chemically and physically processed human Amniotic Membranes (hAM) and analyze the cytocompatibility and proliferation rate (PR) of two primary human mesenchymal stromal cell lines, from different sources and donor conditions seeded over these scaffolds. The evaluated hAM processes were: cold shock to obtain a frozen amniotic membrane (FEAM) with remaining dead epithelial cells, denudation of hAM with trypsin for 20/10 min (DEAM20/10) or treatment with sodium dodecyl sulfate to decellularized hAM (DAM). All samples were sterilized with gamma radiation. The selection of the treated hAM to then generate composites was performed by scanning and transmission electron microscopy and characterization by X-ray diffraction, selecting DEAM10 and FEAM as scaffolds for cell seeding. Two sources of primary human stromal cells were used, both developed by our researchers, human Dental Pulp Stem Cells (hDPSC) from living donors and human Mesenchymal Stromal Cells (hMSC) from bone marrow isolated from brain dead donors. This last line of cells conveys a novel source of human cells that, to our knowledge, have not been tested as part of this type of construct. We developed four in vitro constructs without cytotoxicity signs and with different PR depending on the scaffolds and cells. hDPSC and hMSC grew over both FEAM and DEAM10, but DEAM10 allowed higher PR.






Similar content being viewed by others
Abbreviations
- hAM:
-
Human Amniotic Membranes
- PR:
-
Proliferation rate
- hAM control:
-
Fresh hAM
- FEAM:
-
Freezed Amniotic Membrane
- DEAM20/DEAM10:
-
De-epithelialized hAM with trypsin for 20/10 min
- DAM:
-
Decellularized hAM
- SEM:
-
Scanning Electron Microscopy
- TEM:
-
Transmission Electron Microscopy
- ECM:
-
Extracellular matrix
- XRD:
-
X-ray diffraction
- CBO:
-
Cross Beam Optics
- hDPSC:
-
Human Dental Pulp Stem Cells
- hMSC:
-
Human Mesenchymal Stromal Cells
- TE:
-
Tissue Engineering
- INDT:
-
National Institute of Donation and Transplant
- BDD:
-
Brain dead donor
- BSC:
-
Biological safety cabinet
- PMMA:
-
Polymethylmethacrylate
- SD:
-
Standard deviation
- D-PBS:
-
Dulbecco's phosphate-buffered saline
- CFSE:
-
Carboxyfluorescein succinimidyl ester
- DAPI:
-
4,6-Diamidino- 2-phenylindole
References
Ab Hamid SS, Zahari NK, Yusof N, Hassan A (2014) Scanning electron microscopic assessment on surface morphology of preserved human amniotic membrane after gamma sterilisation. Cell Tissue Bank 15(1):15–24. https://doi.org/10.1007/s10561-012-9353-x
Aggarwal S, Pittenger MF (2005) Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood 105(4):1815–1822. https://doi.org/10.1182/blood-2004-04-1559
Alge DL, Zhou D, Adams LL, Wyss BK, Shadday MD, Woods EJ, Gabriel Chu TM, Goebel WS (2010) Donor-matched comparison of dental pulp stem cells and bone marrow-derived mesenchymal stem cells in a rat model. J Tissue Eng Regen Med 4(1):73–81. https://doi.org/10.1002/term.220
Álvarez I, Bengochea M, Mizraji R, Toledo R, Saldías MC, Carretto E, Pérez H, Castro A, García C (2009a) Three decades of the history of donation and transplantation in Uruguay. Transpl Proc 41(8):3495–3499. https://doi.org/10.1016/j.transproceed.2009.09.005
Álvarez I, Morales Pedraza J, Saldías MC, Pérez Campos H, Wodowóz O, Acosta M, Vicentino W, Silva W, Rodríguez G, Machín D, Álvarez O (2009b) The impact of the International Atomic Energy Agency (Iaea) program on radiation and tissue banking in Uruguay: development of tissues quality control and quality management system in the National Multi-Tissue Bank of Uruguay. Cell Tissue Ban 10(2):173–181. https://doi.org/10.1007/s10561-008-9094-z
Awad HA, Butler DL, Boivin GP, Smith FNL, Malaviya P, Huibregtse B, Caplan AI (1999) Autologous mesenchymal stem cell-mediated repair of tendon. Tissue Eng 5(3):267–277. https://doi.org/10.1089/ten.1999.5.267
Banik BL, Bowers DT, Fattahi P, Brown JL (2017) Bio-instructive scaffolds for musculoskeletal interfaces. Bio-instructive scaffolds for musculoskeletal tissue engineering and regenerative medicine. Elsevier, pp 203–233. https://doi.org/10.1016/B978-0-12-803394-4.00009-4
Castellanos G, Bernabé-García Á, Moraleda JM, Nicolás FJ (2017) Amniotic membrane application for the healing of chronic wounds and ulcers. Placenta 59:146–153. https://doi.org/10.1016/j.placenta.2017.04.005
Cavallo C, Cuomo C, Fantini S, Ricci F, Tazzari PL, Lucarelli E, Donati D, Facchini A, Lisignoli G, Fornasari PM, Grigolo B, Moroni L (2011) Comparison of alternative mesenchymal stem cell sources for cell banking and musculoskeletal advanced therapies. J Cell Biochem 112(5):1418–1430. https://doi.org/10.1002/jcb.23058
Cirman T, Beltram M, Schollmayer P, Rožman P, Kreft ME (2014) Amniotic membrane properties and current practice of amniotic membrane use in ophthalmology in Slovenia. Cell Tissue Bank 15(2):177–192. https://doi.org/10.1007/s10561-013-9417-6
Conde MCM, Chisini LA, Grazioli G, Francia A, de Carvalho RV, Alcázar JCB, Tarquinio SBC, Demarco FF (2016) Does cryopreservation affect the biological properties of stem cells from dental tissues? A systematic review. Braz Dental J 27(6):633–640. https://doi.org/10.1590/0103-6440201600980
Corradetti B, Taraballi F, Minardi S, Van Eps J, Cabrera F, Francis LW, Gazze SA, Ferrari M, Weiner BK, Tasciotti E (2016) Chondroitin sulfate immobilized on a biomimetic scaffold modulates inflammation while driving chondrogenesis. Stem Cells Transl Med 5(5):670–682. https://doi.org/10.5966/sctm.2015-0233
Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini FC, Krause DS, Deans RJ, Keating A, Prockop DJ, Horwitz EM (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8(4):315–317. https://doi.org/10.1080/14653240600855905
Dua HS, Gomes JAP, King AJ, Maharajan VS (2004) The amniotic membrane in ophthalmology. Surv Ophthalmol 49(1):51–77. https://doi.org/10.1016/j.survophthal.2003.10.004
Echarte L, Zunino J, Machin D, Sujanov A, Marquisa N, Lorenzo M, Alvarez I, Bengochea M, Touriño C (2019) Brain-death donors as an alternative source of human stromal mesenchymal cells for cell-based therapy. Cytotherapy 21(5):S86–S87. https://doi.org/10.1016/j.jcyt.2019.03.512
Farhadihosseinabadi B, Farahani M, Tayebi T, Jafari A, Biniazan F, Modaresifar K, Moravvej H, Bahrami S, Redl H, Tayebi L, Niknejad H (2018) Amniotic membrane and its epithelial and mesenchymal stem cells as an appropriate source for skin tissue engineering and regenerative medicine. Artif Cells Nanomed Biotechnol 46(sup2):431–440. https://doi.org/10.1080/21691401.2018.1458730
Francia A, Grazioli G, Echarte L, Maglia Á, Touriño C, Álvarez I (2021) Establecimiento e implementación de un protocolo simplificado de expansión y cultivo de Células Madre de Pulpa Dental Humana (DPSCh). Odontoestomatología. https://doi.org/10.22592/ode2021n37e207
Friedenstein A, Deriglasova U, Kulagina N, Panasuk A, Rudakowa S, Luriá E, Ruadkow IA (1974) Precursors for fibroblasts in different populations of hematopoietic cells as detected by the in vitro colony assay method. Exp Hematol 2(2):83–92
Galipeau J, Krampera M, Barrett J, Dazzi F, Deans RJ, DeBruijn J, Dominici M, Fibbe WE, Gee AP, Gimble JM, Hematti P, Koh MBC, LeBlanc K, Martin I, McNiece IK, Mendicino M, Oh S, Ortiz L, Phinney DG, Sensebe L (2016) International Society for Cellular Therapy perspective on immune functional assays for mesenchymal stromal cells as potency release criterion for advanced phase clinical trials. Cytotherapy 18(2):151–159. https://doi.org/10.1016/j.jcyt.2015.11.008
Gholipourmalekabadi M, Sameni M, Radenkovic D, Mozafari M, Mossahebi-Mohammadi M, Seifalian A (2016) Decellularized human amniotic membrane: How viable is it as a delivery system for human adipose tissue-derived stromal cells? Cell Prolif 49(1):115–121. https://doi.org/10.1111/cpr.12240
Gronthos S, Mankani M, Brahim J, Robey PG, Shi S (2000) Postnatal human dental pulp stem cells (Dpscs) in vitro and invivo. Proc Natl Acad Sci 97(25):13625–13630. https://doi.org/10.1073/pnas.240309797
Hopkinson A, Shanmuganathan VA, Gray T, Yeung AM, Lowe J, James DK, Dua HS (2008) Optimization of amniotic membrane (Am) denuding for tissue engineering. Tissue Eng Part C Methods 14(4):371–381. https://doi.org/10.1089/ten.tec.2008.0315
Proyectos de Investigación Aprobados. (xxxx). Instituto Nacional de Donación y Trasplante. https://www.indt.gub.uy/?S=Proyectos_Investigacion
Johnstone BH, Miller HM, Beck MR, Gu D, Thirumala S, LaFontaine M, Brandacher G, Woods EJ (2020) Identification and characterization of a large source of primary mesenchymal stem cells tightly adhered to bone surfaces of human vertebral body marrow cavities. Cytotherapy 22(11):617–628. https://doi.org/10.1016/j.jcyt.2020.07.003
Keating A (2006) Mesenchymal stromal cells. Curr Opin Hematol 13(6):419–425. https://doi.org/10.1097/01.moh.0000245697.54887.6f
Kesting MR, Wolff K-D, Nobis CP, Rohleder NH (2014) Amniotic membrane in oral and maxillofacial surgery. Oral Maxillofac Surg 18(2):153–164. https://doi.org/10.1007/s10006-012-0382-1
Koizumi N, Fullwood NJ, Bairaktaris G, Inatomi T, Kinoshita S, Quantock AJ (2000) Cultivation of corneal epithelial cells on intact and denuded human amniotic membrane. Invest Ophthalmol Vis Sci 41(9):2506–2513
Langer R, Vacanti JP (1993) Tissue engineering. Science 260(5110):920–926. https://doi.org/10.1126/science.8493529
Laranjo M (2015) Preservation of amniotic membrane. In: Mamede Ana Catarina, Botelho Maria Filomena (eds) Amniotic membrane. Springer, Dordrecht, pp 209–230. https://doi.org/10.1007/978-94-017-9975-1_13
Leal-Marin S, Kern T, Hofmann N, Pogozhykh O, Framme C, Börgel M, Figueiredo C, Glasmacher B, Gryshkov O (2021) Human amniotic membrane: a review on tissue engineering, application, and storage. J Biomed Mater Res B Appl Biomater 109(8):1198–1215. https://doi.org/10.1002/jbm.b.34782
Lim R (2017) Concise review: fetal membranes in regenerative medicine: new tricks from an old dog? Stem Cells Transl Med 6(9):1767–1776. https://doi.org/10.1002/sctm.16-0447
Malhotra C, Jain AK (2014) Human amniotic membrane transplantation: different modalities of its use in ophthalmology. World J Transpl 4(2):111. https://doi.org/10.5500/wjt.v4.i2.111
Mansilla E, Marín GH, Berges M, Scafatti S, Rivas J, Núñez A, Menvielle M, Lamonega R, Gardiner C, Drago H, Sturla F, Portas M, Bossi S, Castuma MV, Peña Luengas S, Roque G, Martire K, Tau JM, Orlandi G, Tarditti A (2015) Cadaveric bone marrow mesenchymal stem cells: first experience treating a patient with large severe burns. Burns Trauma. https://doi.org/10.1186/s41038-015-0018-4
Maraldi T, Riccio M, Pisciotta A, Zavatti M, Carnevale G, Beretti F, La Sala GB, Motta A, De Pol A (2013) Human amniotic fluid-derived and dental pulp-derived stem cells seeded into collagen scaffold repair critical-size bone defects promoting vascularization. Stem Cell Res Ther 4(3):53. https://doi.org/10.1186/scrt203
Monteiro BG, Loureiro RR, Cristovam PC, Covre JL, Gomes JÁP, Kerkis I (2019) Amniotic membrane as a biological scaffold for dental pulp stem cell transplantation in ocular surface reconstruction. Arq Bras de Oftalmol. https://doi.org/10.5935/0004-2749.20190009
Instituto Nacional de Donación y Trasplante. (xxxx). INDT. https://www.indt.gub.uy
Navarro M, Michiardi A, Castaño O, Planell JA (2008) Biomaterials in orthopaedics. J R Soc Interface 5(27):1137–1158. https://doi.org/10.1098/rsif.2008.0151
Perez H, . Saldias, . Sanchez, . Martucci, . Acosta, . Alvarez, . Faccio, . Suescun, . Romero, . Mombru, McGPMcIRLMA (2012) X ray diffraction: an approach to structural quality of biological preserved tissues in tissue banks. In: Katkov Igor (ed) Current frontiers in cryopreservation. InTech. https://doi.org/10.5772/33725
Pfeiffer E, Vickers SM, Frank E, Grodzinsky AJ, Spector M (2008) The effects of glycosaminoglycan content on the compressive modulus of cartilage engineered in type II collagen scaffolds. Osteoarthr Cartil 16(10):1237–1244. https://doi.org/10.1016/j.joca.2008.02.014
Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, Moorman MA, Simonetti DW, Craig S, Marshak DR (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284(5411):143–147. https://doi.org/10.1126/science.284.5411.143
Pittenger MF, Discher DE, Péault BM, Phinney DG, Hare JM, Caplan AI (2019) Mesenchymal stem cell perspective: cell biology to clinical progress. NPJ Regener Med 4(1):22. https://doi.org/10.1038/s41536-019-0083-6
Porzionato A, Stocco E, Barbon S, Grandi F, Macchi V, De Caro R (2018) Tissue-engineered grafts from human decellularized extracellular matrices: a systematic review and future perspectives. Int J Mol Sci 19(12):4117. https://doi.org/10.3390/ijms19124117
Riau AK, Beuerman RW, Lim LS, Mehta JS (2010) Preservation, sterilization and de-epithelialization of human amniotic membrane for use in ocular surface reconstruction. Biomaterials 31(2):216–225. https://doi.org/10.1016/j.biomaterials.2009.09.034
Riccio M, Maraldi T, Pisciotta A, La Sala GB, Ferrari A, Bruzzesi G, Motta A, Migliaresi C, De Pol A (2012) Fibroin scaffold repairs critical-size bone defects in vivo supported by human amniotic fluid and dental pulp stem cells. Tissue Eng Part A 18(9–10):1006–1013. https://doi.org/10.1089/ten.tea.2011.0542
Salah RA, Mohamed IK, El-Badri N (2018) Development of decellularized amniotic membrane as a bioscaffold for bone marrow-derived mesenchymal stem cells: ultrastructural study. J Mol Histol 49(3):289–301. https://doi.org/10.1007/s10735-018-9768-1
Silini AR, Cargnoni A, Magatti M, Pianta S, Parolini O (2015) The long path of human placenta, and its derivatives, in regenerative medicine. Front Bioeng Biotechnol. https://doi.org/10.3389/fbioe.2015.00162
Tao H, Han Z, Han ZC, Li Z (2016) Proangiogenic features of mesenchymal stem cells and their therapeutic applications. Stem Cells Inter 2016:1–11. https://doi.org/10.1155/2016/1314709
Verma K, Bains R, Bains VK, Rawtiya M, Loomba K, Srivastava SC (2014) Therapeutic potential of dental pulp stem cells in regenerative medicine: an overview. Dent Res J 11(3):302–308
Wagner M, Walter P, Salla S, Johnen S, Plange N, Rütten S, Goecke TW, Fuest M (2018) Cryopreservation of amniotic membrane with and without glycerol additive. Graefe’s Arch Clinic Exp Ophthalmol 256(6):1117–1126. https://doi.org/10.1007/s00417-018-3973-1
Young RG, Butler DL, Weber W, Caplan AI, Gordon SL, Fink DJ (1998) Use of mesenchymal stem cells in a collagen matrix for achilles tendon repair. J Orthop Res 16(4):406–413. https://doi.org/10.1002/jor.1100160403
Zhang T, Yam GH-F, Riau AK, Poh R, Allen JC, Peh GS, Beuerman RW, Tan DT, Mehta JS (2013) The effect of amniotic membrane de-epithelialization method on its biological properties and ability to promote limbal epithelial cell culture. Investig Opthalmol Vis Sci 54(4):3072. https://doi.org/10.1167/iovs.12-10805
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Echarte, L., Grazioli, G., Pereira, L. et al. Processing methods for human amniotic membrane as scaffold for tissue engineering with mesenchymal stromal human cells. Cell Tissue Bank 25, 269–283 (2024). https://doi.org/10.1007/s10561-022-10014-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-022-10014-8