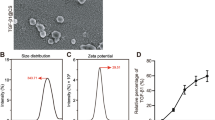
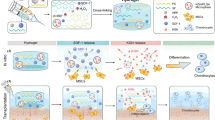
Abstract
The treatment of articular cartilage defects has become a major clinical concern. Currently, additional efforts are necessary to develop effective methods to cure this disease. In this work, we combined gene therapy with tissue engineering methods to test their effect on cartilage repair. In in vitro experiments, we obtained C-type natriuretic peptide (CNP) gene-modified bone marrow-derived mesenchymal stem cells (BMSCs) by transfection with recombinant adenovirus containing the CNP gene and revealed that CNP gene-modified BMSCs had good chondrogenic differentiation ability. By the freeze-drying method, we successfully synthesized a chitosan/silk fibroin (CS/SF) porous scaffold, which had a suitable aperture size for chondrogenesis. Then, we loaded CNP gene-modified BMSCs onto CS/SF scaffolds and tested their effect on repairing full-thickness cartilage defects in rat joints. The gross morphology and histology examination results showed that the composite of the CNP gene-modified BMSCs and CS/SF scaffolds had better repair effects than those of the other three groups at each time point. Additionally, compared to the group with BMSCs and scaffolds, we found that there was more cartilage matrix in the CNP gene-modified BMSCs and CS/SF scaffolds group. Data obtained in the present study suggest that the composite of CNP gene-modified BMSCs and CS/SF scaffolds represent promising strategies for repairing focal cartilage lesions.





Similar content being viewed by others
References
Abarrategi A, Lopiz-Morales Y, Ramos V, Civantos A, Lopez-Duran L, Marco F, Lopez-Lacomba JL (2010) Chitosan scaffolds for osteochondral tissue regeneration. J Biomed Mater Res Part A 95:1132–1141. https://doi.org/10.1002/jbm.a.32912
Agoston H, Khan S, James CG, Gillespie JR, Serra R, Stanton LA, Beier F (2007) C-type natriuretic peptide regulates endochondral bone growth through p38 MAP kinase-dependent and -independent pathways. BMC Dev Biol 7:18. https://doi.org/10.1186/1471-213x-7-18
Augst A et al (2008) Effects of chondrogenic and osteogenic regulatory factors on composite constructs grown using human mesenchymal stem cells, silk scaffolds and bioreactors. J R Soc Interface 5:929–939. https://doi.org/10.1098/rsif.2007.1302
Bhardwaj N, Kundu SC (2012) Chondrogenic differentiation of rat MSCs on porous scaffolds of silk fibroin/chitosan blends. Biomaterials 33:2848–2857. https://doi.org/10.1016/j.biomaterials.2011.12.028
Bhardwaj N, Nguyen QT, Chen AC, Kaplan DL, Sah RL, Kundu SC (2011) Potential of 3-D tissue constructs engineered from bovine chondrocytes/silk fibroin-chitosan for in vitro cartilage tissue engineering. Biomaterials 32:5773–5781. https://doi.org/10.1016/j.biomaterials.2011.04.061
Camarero-Espinosa S, Rothen-Rutishauser B, Foster EJ, Weder C (2016) Articular cartilage: from formation to tissue engineering. Biomater Sci. https://doi.org/10.1039/c6bm00068a
Cao H, Kuboyama N (2010) A biodegradable porous composite scaffold of PGA/beta-TCP for bone tissue engineering. Bone 46:386–395. https://doi.org/10.1016/j.bone.2009.09.031
Chlapanidas T et al (2011) Regenerated silk fibroin scaffold and infrapatellar adipose stromal vascular fraction as feeder-layer: a new product for cartilage advanced therapy. Tissue Eng Part A 17:1725–1733. https://doi.org/10.1089/ten.TEA.2010.0636
Cotter EJ, Wang KC, Yanke AB, Chubinskaya S (2018) Bone marrow aspirate concentrate for cartilage defects of the knee: from bench to bedside evidence. Cartilage 9:161–170. https://doi.org/10.1177/1947603517741169
Cucchiarini M, Madry H (2014) Use of tissue engineering strategies to repair joint tissues in osteoarthritis: viral gene transfer approaches. Curr Rheumatol Rep 16:449. https://doi.org/10.1007/s11926-014-0449-0
Cucchiarini M, Madry H, Guilak F, Saris DB, Stoddart MJ, Koon Wong M, Roughley P (2014) A vision on the future of articular cartilage repair. Eur Cells Mater 27:12–16
Frisch J, Venkatesan JK, Rey-Rico A, Madry H, Cucchiarini M (2015) Current progress in stem cell-based gene therapy for articular cartilage repair. Curr Stem Cell Res Ther 10:121–131
Gigante A, Cecconi S, Calcagno S, Busilacchi A, Enea D (2012) Arthroscopic knee cartilage repair with covered microfracture and bone marrow concentrate. Arthrosc Tech 1:e175–e180. https://doi.org/10.1016/j.eats.2012.07.001
Jiang X et al (2017) TGF-beta1 is involved in vitamin D-induced chondrogenic differentiation of bone marrow-derived mesenchymal stem cells by regulating the ERK/JNK pathway. Cell Physiol Biochem Int J Exp Cell Physiol Biochem Pharmacol 42:2230–2241. https://doi.org/10.1159/000479997
Kim JD et al (2014) Clinical outcome of autologous bone marrow aspirates concentrate (BMAC) injection in degenerative arthritis of the knee. Eur J Orthop Surg Traumatol Orthop Traumatol 24:1505–1511. https://doi.org/10.1007/s00590-013-1393-9
Kocamaz E, Gok D, Cetinkaya A, Tufan AC (2012) Implication of C-type natriuretic peptide-3 signaling in glycosaminoglycan synthesis and chondrocyte hypertrophy during TGF-beta1 induced chondrogenic differentiation of chicken bone marrow-derived mesenchymal stem cells. J Mol Histol 43:497–508. https://doi.org/10.1007/s10735-012-9430-2
Kwon H, Sun L, Cairns DM, Rainbow RS, Preda RC, Kaplan DL, Zeng L (2013) The influence of scaffold material on chondrocytes under inflammatory conditions. Acta Biomater 9:6563–6575. https://doi.org/10.1016/j.actbio.2013.01.004
Melke J, Midha S, Ghosh S, Ito K, Hofmann S (2016) Silk fibroin as biomaterial for bone tissue engineering. Acta Biomater 31:1–16. https://doi.org/10.1016/j.actbio.2015.09.005
Nakao K et al (2015) The local CNP/GC-B system in growth plate is responsible for physiological endochondral bone growth. Sci Rep 5:10554. https://doi.org/10.1038/srep10554
O’Driscoll SW (1998) The healing and regeneration of articular cartilage. J Bone Jt Surg Am 80:1795–1812
Olney RC (2006) C-type natriuretic peptide in growth: a new paradigm. Growth Horm IGF Res 16 Suppl A:S6-14. https://doi.org/10.1016/j.ghir.2006.03.016
Orozco L et al (2013) Treatment of knee osteoarthritis with autologous mesenchymal stem cells: a pilot study. Transplantation 95:1535–1541. https://doi.org/10.1097/TP.0b013e318291a2da
Peake NJ, Bader DL, Vessillier S, Ramachandran M, Salter DM, Hobbs AJ, Chowdhury TT (2015) C-type natriuretic peptide signalling drives homeostatic effects in human chondrocytes. Biochem Biophys Res Commun 465:784–789. https://doi.org/10.1016/j.bbrc.2015.08.087
Safran MR, Seiber K (2010) The evidence for surgical repair of articular cartilage in the knee. J Am Acad Orthop Surg 18:259–266
Shi Q, Qian ZY, Liu C, Guo XM, Xu J (2015) Construction and identification of recombinant adenovirus vector carrying C-type natriuretic peptide gene. Chin J Prosthodont 16:325–329
Suh JK, Matthew HW (2000) Application of chitosan-based polysaccharide biomaterials in cartilage tissue engineering: a review. Biomaterials 21:2589–2598
Tezcan B, Serter S, Kiter E, Tufan AC (2010) Dose dependent effect of C-type natriuretic peptide signaling in glycosaminoglycan synthesis during TGF-beta1 induced chondrogenic differentiation of mesenchymal stem cells. J Mol Histol 41:247–258. https://doi.org/10.1007/s10735-010-9284-4
Vishwanath V, Pramanik K, Biswas A (2016) Optimization and evaluation of silk fibroin-chitosan freeze-dried porous scaffolds for cartilage tissue engineering application. J Biomater Sci Polym Ed 27:657–674. https://doi.org/10.1080/09205063.2016.1148303
Waldman SD, Usmani Y, Tse MY, Pang SC (2008) Differential effects of natriuretic peptide stimulation on tissue-engineered cartilage. Tissue Eng Part A 14:441–448. https://doi.org/10.1089/tea.2007.0035
Welton KL, Logterman S, Bartley JH, Vidal AF, McCarty EC (2018) knee cartilage repair and restoration: common problems and solutions. Clin Sports Med 37:307–330. https://doi.org/10.1016/j.csm.2017.12.008
Yang H et al (2013) Comparison of mesenchymal stem cells derived from gingival tissue and periodontal ligament in different incubation conditions. Biomaterials 34:7033–7047. https://doi.org/10.1016/j.biomaterials.2013.05.025
Ye K et al (2014) Chondrogenesis of infrapatellar fat pad derived adipose stem cells in 3D printed chitosan scaffold. PloS ONE 9:e99410. https://doi.org/10.1371/journal.pone.0099410
Zhang P, Hong Z, Yu T, Chen X, Jing X (2009) In vivo mineralization and osteogenesis of nanocomposite scaffold of poly(lactide-co-glycolide) and hydroxyapatite surface-grafted with poly(L-lactide). Biomaterials 30:58–70. https://doi.org/10.1016/j.biomaterials.2008.08.041
Zhang Q, Lu H, Kawazoe N, Chen G (2014) Pore size effect of collagen scaffolds on cartilage regeneration. Acta Biomater 10:2005–2013. https://doi.org/10.1016/j.actbio.2013.12.042
Zhao Y, Tan K, Zhou Y, Ye Z, Tan WS (2016) A combinatorial variation in surface chemistry and pore size of three-dimensional porous poly(epsilon-caprolactone) scaffolds modulates the behaviors of mesenchymal stem cells. Mater Sci Eng C Mater Biol Appl 59:193–202. https://doi.org/10.1016/j.msec.2015.10.017
Zhu S, Zhang B, Man C, Ma Y, Liu X, Hu J (2014) Combined effects of connective tissue growth factor-modified bone marrow-derived mesenchymal stem cells and NaOH-treated PLGA scaffolds on the repair of articular cartilage defect in rabbits. Cell Transpl 23:715–727. https://doi.org/10.3727/096368913x669770
Acknowledgements
This study was supported by Grant from the National Natural Science Foundation of China (No. 51472270).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest associated with this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, S., Qian, Z., Liu, D. et al. Integration of C-type natriuretic peptide gene-modified bone marrow mesenchymal stem cells with chitosan/silk fibroin scaffolds as a promising strategy for articular cartilage regeneration. Cell Tissue Bank 20, 209–220 (2019). https://doi.org/10.1007/s10561-019-09760-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-019-09760-z